
CHEMISTRY:MOLECULAR NATURE (LL)W/ACCESS
7th Edition
ISBN: 9781119497325
Author: JESPERSEN
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 11, Problem 92RQ
Intermolecular Forces and Physical Properties
What kinds of intermolecular attractive forces are present in the following substances?
(a)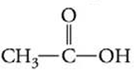 (b)
(b)
(c)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(ME EX2) Can you please explain problems to me in detail, step by step? Thank you so much! If needed color code them for me.
(SE EX 2) Problems 1-3, can you please explain them to me in detail and color code anything if needed.
(SE EX 2) Problems can you please explain them to me in detail and color-code anything if necessary?
Chapter 11 Solutions
CHEMISTRY:MOLECULAR NATURE (LL)W/ACCESS
Ch. 11 - Prob. 1PECh. 11 - List the following in order of their boiling...Ch. 11 - Propylamine and trimethylamine have the same...Ch. 11 - People living in arid, dry, regions can cool their...Ch. 11 - Use the kinetic molecular theory to explain why...Ch. 11 - Considering Figure 11.24, in which direction...Ch. 11 - Suppose a liquid is in equilibrium with its vapor...Ch. 11 - The Dead Sea is approximately 1300 ft below sea...Ch. 11 - The atmospheric pressure at the summit of Mt....Ch. 11 - Benzene has a boiling point of 80.1C, and a...
Ch. 11 - Steam can cause more severe bums than water, even...Ch. 11 - The equilibrium line from point B to D in Figure...Ch. 11 - What phase changes will occur if water at 20C and...Ch. 11 - Prob. 14PECh. 11 - Use Le Chtelier's principle to predict how a...Ch. 11 - Prob. 16PECh. 11 - At 0.00C, hexane, C6H14, has a vapor pressure of...Ch. 11 - Prob. 18PECh. 11 - Chromium crystallizes in a body-centered cubic...Ch. 11 - What is the ratio of the ions in the unit cell of...Ch. 11 - Polonium is the only metal known to crystallize in...Ch. 11 - Use the data in the previous Practice Exercise to...Ch. 11 - Stearic acid is an organic acid that has a chain...Ch. 11 - Boron nitride, which has the empirical formula BN,...Ch. 11 - Crystals of elemental sulfur are easily crushed...Ch. 11 - 11.1 Why are the intermolecular attractive forces...Ch. 11 - Compare the behavior of gases, liquids, and solids...Ch. 11 - Prob. 3RQCh. 11 - Why do intermolecular attractions weaken as the...Ch. 11 - Prob. 5RQCh. 11 - Define polarizability. How does this property...Ch. 11 - Prob. 7RQCh. 11 - 11.8 Which nonmetals, besides hydrogen, are...Ch. 11 - Prob. 9RQCh. 11 - Which would give a stronger iondipole interaction...Ch. 11 - Prob. 11RQCh. 11 - Prob. 12RQCh. 11 - Intermolecular Forces and Physical...Ch. 11 - Prob. 14RQCh. 11 - Intermolecular Forces and Physical Properties Name...Ch. 11 - Prob. 16RQCh. 11 - Prob. 17RQCh. 11 - Prob. 18RQCh. 11 - Prob. 19RQCh. 11 - Prob. 20RQCh. 11 - Intermolecular Forces and Physical...Ch. 11 - Prob. 22RQCh. 11 - Prob. 23RQCh. 11 - Prob. 24RQCh. 11 - Prob. 25RQCh. 11 - Prob. 26RQCh. 11 - Prob. 27RQCh. 11 - Prob. 28RQCh. 11 - Prob. 29RQCh. 11 - Changes of State and Dynamic Equilibrium What...Ch. 11 - Prob. 31RQCh. 11 - Changes of State and Dynamic Equilibrium
11.32 Why...Ch. 11 - Changes of State and Dynamic Equilibrium
11.33...Ch. 11 - Changes of State and Dynamic Equilibrium
11.34....Ch. 11 - Prob. 35RQCh. 11 - Prob. 36RQCh. 11 - Vapor Pressures of Liquids and Solids
11.37...Ch. 11 - Prob. 38RQCh. 11 - Vapor Pressures of Liquids and Solids 11.39 What...Ch. 11 - Vapor Pressures of Liquids and Solids Why does...Ch. 11 - Vapor Pressures of Liquids and Solids Why do we...Ch. 11 - Prob. 42RQCh. 11 - Boiling Points of Liquids Why does the boiling...Ch. 11 - Boiling Points of Liquids Mt. Kilimanjaro in...Ch. 11 - Boiling Points of Liquids
11.45. When liquid...Ch. 11 - Prob. 46RQCh. 11 - Boiling Points of Liquids Butane, C4H10, has a...Ch. 11 - Boiling Points of Liquids
11.48. Why does have a...Ch. 11 - Boiling Points of Liquids An HF bond is more polar...Ch. 11 - Energy and Changes of State The following is a...Ch. 11 - Energy and Changes of State
11.51 Why is larger...Ch. 11 - Energy and Changes of State Would the heat of...Ch. 11 - Energy and Changes of State Hurricanes can travel...Ch. 11 - Energy and Changes of State Ethanol (grain...Ch. 11 - Energy and Changes of State A burn caused by steam...Ch. 11 - Energy and Changes of State
11.56 Arrange the...Ch. 11 - Prob. 57RQCh. 11 - Phase Diagrams
11.58 Define critical temperature...Ch. 11 - Phase Diagrams What is a supercritical fluid? Why...Ch. 11 - Phase Diagrams
11.60 What phases of a substance...Ch. 11 - Prob. 61RQCh. 11 - Prob. 62RQCh. 11 - Phase Diagrams Sketch a generic phase diagram that...Ch. 11 - Phase Diagrams
11.64 What is the significance of...Ch. 11 - Prob. 65RQCh. 11 - Le Chtelier's Principle and Changes of State State...Ch. 11 - Le Châtelier's Principle and Changes of...Ch. 11 - Le Chtelier's Principle and Changes of State Use...Ch. 11 - Le Chtelier's Principle and Changes of State Use...Ch. 11 - Le Châtelier's Principle and Changes of...Ch. 11 - Determining Heats of Vaporization According to the...Ch. 11 - Determining Heats of Vaporization Why can't...Ch. 11 - Determining Heats of Vaporization Why can any...Ch. 11 - Prob. 74RQCh. 11 - Prob. 75RQCh. 11 - Prob. 76RQCh. 11 - Determining the Structure of Solids What...Ch. 11 - Determining the Structure of Solids
11.78 The...Ch. 11 - The figure below illustrates the way the atoms of...Ch. 11 - Make a sketch of a layer of sodium ions and...Ch. 11 - 11.81 How do the crystal structures of copper and...Ch. 11 - Determining the Structure of Solids
11.82 What...Ch. 11 - Determining the Structure of Solids Only 14...Ch. 11 - Determining the Structure of Solids Write the...Ch. 11 - Determining the Structure of Solids Why cant...Ch. 11 - Prob. 86RQCh. 11 - Crystal Types and Physical Properties
11.87 What...Ch. 11 - Prob. 88RQCh. 11 - Prob. 89RQCh. 11 - Prob. 90RQCh. 11 - Intermolecular Forces and Physical Properties What...Ch. 11 - Intermolecular Forces and Physical Properties What...Ch. 11 - Intermolecular Forces and Physical Properties...Ch. 11 - Prob. 94RQCh. 11 - 11.95 Consider the compounds (chloroform, an...Ch. 11 - 11.96 Carbon dioxide does not liquefy at...Ch. 11 - Prob. 97RQCh. 11 - Prob. 98RQCh. 11 - Prob. 99RQCh. 11 - Prob. 100RQCh. 11 - 11.101 The following are the vapor pressures of...Ch. 11 - 11.102 The boiling points of some common...Ch. 11 - 11.103 Using the information in Problem 11.101,...Ch. 11 - 11.104 Using the information in Problem 11.102,...Ch. 11 - 11.105 What intermolecular forces must the...Ch. 11 - 11.106 What intermolecular attractions will be...Ch. 11 - Energy and Changes of State The molar heat of...Ch. 11 - Energy and Changes of State The molar heat of...Ch. 11 - *11.109 Suppose 45.0 g of water at is added to...Ch. 11 - A cube of solid benzene (C6H6) at its melting...Ch. 11 - Prob. 111RQCh. 11 - Prob. 112RQCh. 11 - Prob. 113RQCh. 11 - Prob. 114RQCh. 11 - Prob. 115RQCh. 11 - Prob. 116RQCh. 11 - Determining Heats of Vaporization
*11.117 Mercury...Ch. 11 - Prob. 118RQCh. 11 - Prob. 119RQCh. 11 - *11.120 If the vapor pressure of ethylene glycol...Ch. 11 - Determining the Structure of Solids
11.121 How...Ch. 11 - 11.122 How many copper atoms are within the...Ch. 11 - The atomic radius of nickel is 1.24 . Nickel...Ch. 11 - 11.124 Silver forms face-centered cubic crystals....Ch. 11 - Potassium ions have a radius of 133 pm, and...Ch. 11 - 11.126 The unit cell edge in sodium chloride has a...Ch. 11 - Prob. 127RQCh. 11 - Prob. 128RQCh. 11 - *11.129 Cesium chloride forms a simple cubic...Ch. 11 - 11.130 Rubidium chloride has the rock salt...Ch. 11 - Prob. 131RQCh. 11 - Crystal Types and Physical Properties Elemental...Ch. 11 - Prob. 133RQCh. 11 - Prob. 134RQCh. 11 - Prob. 135RQCh. 11 - Crystal Types and Physical Properties
11.1 36...Ch. 11 - List all of the attractive forces that exist in...Ch. 11 - 11.138 Calculate the mass of water vapor present...Ch. 11 - 11.139 Should acetone molecules be attracted to...Ch. 11 - The following thermochemical equations apply to...Ch. 11 - Melting point is sometimes used as an indication...Ch. 11 - When warm, moist air sweeps in from the ocean and...Ch. 11 - *11.143 Gold crystallizes in a face-centered cubic...Ch. 11 - Gold crystallizes with a face-centered cubic unit...Ch. 11 - Identify the type of unit cell belonging to the...Ch. 11 - Calculate the amount of empty space (in pm3) in...Ch. 11 - Silver has an atomic radius of 144 pm. What would...Ch. 11 - Potassium chloride crystallizes with the rock salt...Ch. 11 - Prob. 149RQCh. 11 - There are 270 Calories in a Hersheys* Milk...Ch. 11 - Prob. 151RQCh. 11 - *11.152 Frecze-drying is a process used to...Ch. 11 - When reporting the vapor pressure for a substance...Ch. 11 - 11.154 Supercritical is used to decaffeinate...Ch. 11 - 11.155 Freshly precipitated crystals are usually...Ch. 11 - 11.156 What are three “everyday” applications of...Ch. 11 - Prob. 157RQCh. 11 - 11.158 Galileo's thermometer is a tube of liquid...Ch. 11 - Use the Clausius-Clapeyron equation to plot the...Ch. 11 - Prob. 160RQCh. 11 - Earlier in this chapter it was noted that the...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Chlorine has two isotopes, 35Cl and 37Cl; 75.77% of chlorine is 35Cl, and 24.23% is 37Cl. The atomic mass of 35...
Organic Chemistry (8th Edition)
2.81 In which of the fo1losing pairs do both numbers contain the same number of significant figures? (2.2)
a....
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
DRAW IT The diagram shows a cell in meiosis. (a) Label the appropriate structures with these terms: chromosome ...
Campbell Biology (11th Edition)
Define histology.
Fundamentals of Anatomy & Physiology (11th Edition)
57. Which buffer system is the best choice to create a buffer with pH = 7.20? For the best system, calculate th...
Chemistry: A Molecular Approach (4th Edition)
What is the reducing agent in the following reaction?
2 Br –– (aq) + H2 O2 (aq) + 2 H+ (aq) → Br2 (aq) + 2 H2 ...
Chemistry: The Central Science (14th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (SE EX 2) Problems can you please explain them to me in detail and color-code anything if necessary?arrow_forward(ME EX2) Prblms 5-7 Can you please explain problems 5-7 to me in detail, step by step? Thank you so much! If needed color code them for me.arrow_forwardPlease help me understand how to predict 4-8, using the chemicals and formula equations, and ions. Is seems like a lot but that basically what I need help understanding, please! Thank you in advance. P.S - keep seeing people to reach out in the comments if I had additional questions, but I don't see one???arrow_forward
- (ME EX2) Problems Could you please explain problems to me in detail, step by step? Thank you so much! If necessary, please color-code them for me.arrow_forward(SE EX 2) Problems can you please explain them to me in detail and color-code anything if necessary?arrow_forward(SE EX 2) Problems 4-7, can you please explain them to me in detail and color-code anything if necessary?arrow_forward
- Can you explain problems 20 - 25 on here (step by step) and in detail for me pleasearrow_forward(ME EX2) Can you please explain problems to me in detail, step by step? Thank you so much! If needed color code them for me.arrow_forward(ME EX2) Problems Could you please explain problems to me in detail, step by step? Thank you so much! If necessary, please color-code them for me.arrow_forward
- (SE EX2) Prblms #1-3: Can you please explain in detail if needed color code them for the three different problems.arrow_forward(ME EX2) Problems Could you please explain problems to me in detail, step by step? Thank you so much! If necessary, please color-code them for me.arrow_forward(ME EX2) Prblms 15-16 Can you please explain problems 15-16 to me in detail, step by step? Thank you so much! If needed color code them for me.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Chemical Equilibria and Reaction Quotients; Author: Professor Dave Explains;https://www.youtube.com/watch?v=1GiZzCzmO5Q;License: Standard YouTube License, CC-BY