
Study Guide for Chemistry: The Central Science
13th Edition
ISBN: 9780321949288
Author: Theodore E. Brown, James C. Hill
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 70E
Write the balanced equation for the reaction that occurs when ethylene glycol, C2H4(OH)2 Burns in air ?
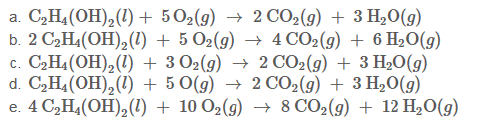
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Can someone help me whats the issue?
a. The change in the Gibbs energy of a certain constant pressure process is found to fit the expression:
AG-85.1 J mol −1 +36.5 J mol ¹K-1 × T
A. Calculate the value of AS for the process.
B. Next, use the Gibbs-Helmholtz equation:
(a(AG/T))
ΔΗ
-
T2
to calculate the value of AH for the process.
None
Chapter 11 Solutions
Study Guide for Chemistry: The Central Science
Ch. 11.2 - Only two isotopes of copper occur naturally:63Cu...Ch. 11.2 - 2.36 Rubidium has two naturally occurring...Ch. 11.3 - a. Thomson’s cathode-ray tube (Figure 2.49) and...Ch. 11.3 -
2.38 Consider the mass spectrometer shown in...Ch. 11.4 - Naturally occurring magnesium has the following...Ch. 11.4 - Mass spectrometry is more often applied to...Ch. 11.5 - 2-41 For each of the following elements, write its...Ch. 11.5 - Locate each of the following elements in the...Ch. 11.6 - 2-43 For each of the following elements, write its...Ch. 11.6 - 2.44 The elements of group 4A show an interesting...
Ch. 11.7 - 2.45 The structural formulas of the compounds...Ch. 11.7 - 2.46 Ball-and-stick representations of benzene, a...Ch. 11 - Prob. 1DECh. 11 - 2.59 Using the periodic table to guide you,...Ch. 11 -
2.71 Name the following ionic compounds:
a....Ch. 11 -
2.83
What is a functional group?
What functional...Ch. 11 - The element lead (Pb) consists of four naturally...Ch. 11 - Prob. 5ECh. 11 - The molecules have the same molecular formula...Ch. 11 - A sample of an ionic compound containing iron and...Ch. 11 -
The compound dioxane, which is used as a solvent...Ch. 11 - If 3.00 g of titanium metal is reacted with 6.00 g...Ch. 11 -
2.48 Two substances have the same molecular and...Ch. 11 - 2.49 Write the empirical formula corresponding to...Ch. 11 - Determine the molecular and empirical formulas of...Ch. 11 - 251 How many hydrogen atoms are un each of the...Ch. 11 - Prob. 14ECh. 11 - 253 Write the molecular and structural formulas...Ch. 11 - 2-54 Write the molecular and structural formulas...Ch. 11 - Fill in the gaps in the following table’Ch. 11 - 2.56 Fill in the gaps in the following...Ch. 11 - Prob. 19ECh. 11 - Prob. 20ECh. 11 - Predict the chemical formulas of the compounds...Ch. 11 - Prob. 22ECh. 11 - Prob. 23ECh. 11 - Predict whether each of the following compounds is...Ch. 11 - 2.66 Which of the following are ionic, and which...Ch. 11 - Prob. 26ECh. 11 - Prob. 27ECh. 11 -
2.69 Give the names and charges of the cation and...Ch. 11 - Give the names and charges of the cation and anion...Ch. 11 - Prob. 30ECh. 11 -
Give the chemical formula for each of the...Ch. 11 -
2.75 Give the name or chemical formula, as...Ch. 11 - Prob. 33ECh. 11 -
2.T Give the name or Chemical formula, as...Ch. 11 - Prob. 35ECh. 11 - Prob. 36ECh. 11 - Assume that you encounter the following sentences...Ch. 11 - a. What is a hydrocarbon? b. Pentane is the alkane...Ch. 11 - Prob. 39ECh. 11 -
2.85 Chloropropane is derived from propane by...Ch. 11 - Prob. 41ECh. 11 - Suppose a scientist repeats the Millikan oil-drop...Ch. 11 -
2.88 The natural abundance of 3He is...Ch. 11 - A cube of gold that is 1.00 cm on a side has a...Ch. 11 -
2.90 The diameter of a rubidium atom is 4.95 A....Ch. 11 -
2.91
Assuming the dimensions of the nucleus and...Ch. 11 - (a) What is the significance of the critical...Ch. 11 -
2.93 The nucleus of 6Li is a powerful absorber of...Ch. 11 - The element oxygen has three naturally occurring...Ch. 11 - Using a suitable reference such as the CRC...Ch. 11 - There are two different isotopes of bromine atoms....Ch. 11 -
2.99 It is common in mass spectrometry to assume...Ch. 11 - From the following list of elements—Ar, H, Ga, Al,...Ch. 11 - Prob. 54ECh. 11 -
2.102 The explosion of an atomic bomb releases...Ch. 11 - Prob. 56ECh. 11 - Prob. 57ECh. 11 -
2.105 From the molecular structures shown here,...Ch. 11 -
2.106 Name each of the following oxides. Assuming...Ch. 11 - Prob. 60ECh. 11 - Prob. 61ECh. 11 - Give the chemical names of each of the following...Ch. 11 -
2.112 Many familiar substances have common,...Ch. 11 -
2.113 Because many ions and compounds have very...Ch. 11 -
2.114 In what part of the atom does the strong...Ch. 11 - In the following diagram, the white spheres...Ch. 11 - In the following digram, the white spheres...Ch. 11 - Prob. 68ECh. 11 - Balance these equations by providing the missing...Ch. 11 - Write the balanced equation for the reaction that...Ch. 11 - Prob. 71ECh. 11 - Which of the following is the correct formula...Ch. 11 - Prob. 73AECh. 11 - Prob. 74AECh. 11 - Calculate the percentage of potassium by mass in...Ch. 11 - Which of the following samples contains the fewest...Ch. 11 - In dichloromethane, CH2Cl2 (= 1.60D)), the...Ch. 11 - Prob. 78AECh. 11 -
How many oxygen atoms are in (a) 0.25 mol...Ch. 11 - Prob. 80AECh. 11 - Prob. 81AECh. 11 - What is the mass, in grams, of 6.33 mol of NaHC03...Ch. 11 - What is the mass, in grams, of (a) 0.50 mol of...Ch. 11 - How many chlorine atoms are in 12.2 g of CCL4? a....Ch. 11 -
a. How many nitric acid molecules are in 4.20 g...Ch. 11 - A 2.144-g sample of phosgene, a compound used as a...Ch. 11 - A 5.325-g sample of methyl benzoate, a compound...Ch. 11 -
Cyclohexane a commonly used organic solvent, is...Ch. 11 - Prob. 89AECh. 11 - Prob. 90IECh. 11 - Decomposition of KCIO3 is sometimes used to...Ch. 11 - Propane, C3 H8 (Figure 3.8), is a common fuel used...Ch. 11 -
Methanol, CH3OH, reacts with oxygen from air in a...Ch. 11 - When 24 mol of methanol and 15 mol of oxygen...Ch. 11 - a. When 1.50 mol of Al and 3.00 mol of Cl2 combine...Ch. 11 - Molten gallium reacts with arsenic to form the...Ch. 11 -
When a 2.00-g strip of zinc metal is placed in...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- ASP please....arrow_forwardNonearrow_forwardConsider the structure of 1-bromo-2-fluoroethane. Part 1 of 2 Draw the Newman projection for the anti conformation of 1-bromo-2-fluoroethane, viewed down the C1-C2 bond. ✡ ぬ Part 2 of 2 H H F Br H H ☑ Draw the Newman projection for the gauche conformation of 1-bromo-2-fluoroethane, viewed down the C1-C2 bond. H F Br H Harrow_forward
- Please help me answer this question. I don't understand how or where the different reagents will attach and it's mostly due to the wedge bond because I haven't seen a problem like this before. Please provide a detailed explanation and a drawing showing how it can happen and what the final product will look like.arrow_forwardWhich of the following compounds is the most acidic in the gas phase? Group of answer choices H2O SiH4 HBr H2Sarrow_forwardWhich of the following is the most acidic transition metal cation? Group of answer choices Fe3+ Sc3+ Mn4+ Zn2+arrow_forward
- Based on the thermodynamics of acetic acid dissociation discussed in Lecture 2-5, what can you conclude about the standard enthalpy change (ΔHo) of acid dissociation for HCl? Group of answer choices You cannot arrive at any of the other three conclusions It is a positive value It is more negative than −0.4 kJ/mol It equals −0.4 kJ/molarrow_forwardPLEASE HELP URGENT!arrow_forwardDraw the skeletal structure corresponding to the following IUPAC name: 7-isopropyl-3-methyldecanearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
ENVIRONMENTAL POLLUTION; Author: 7activestudio;https://www.youtube.com/watch?v=oxtMFmDTv3Q;License: Standard YouTube License, CC-BY