
Concept explainers
(a)
Interpretation: The IUPAC name for the given cycloalkane needs to be determined.
Concept Introduction: According to the IUPAC rule,
(a)
Explanation of Solution
The given structure is as follows:
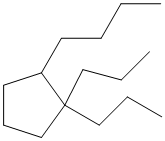
The above structure contains a cycloalkane ring with 5 carbon atoms thus, it is cyclopentane.
Here, numbering will be done according to the number of substituents present of cyclopentane ring. There are 2 substituent at one position and 1 substituent at other.
The numbering can be done as follows:
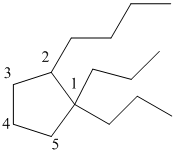
From the above structure, there are 2 propyl groups at 1st position and 1 butyl group at 2nd position of cyclopentane ring.
Thus, the IUPAC name of the molecule will be:
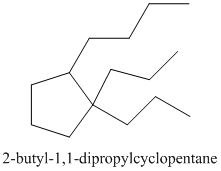
(b)
Interpretation: The IUPAC name for the given
Concept Introduction: According to the IUPAC rule, numbering of atom is done to find the longest continuous carbon chain in the molecule. After that name of groups attached to the chain are identified. The location of substituent groups is designated by numbers and name. The last step is to assemble the name by listing the groups in alphabetical order. Prefixes such as di, tri, tetra etc. are used to represent groups of same kind.
(b)
Explanation of Solution
The given structure is as follows:
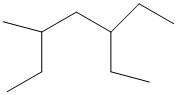
The numbering will be done according to the longest carbon chain in the molecule.
The numbering is done as follows:
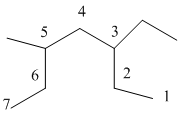
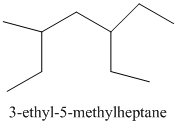
From the above structure, there is 1 ethyl group at 3rd position and 1 methyl group at 5th position of the main chain containing 7 carbon atoms.
Therefore, the IUPAC name of the structure will be:
Thus,
(c)
Interpretation: The IUPAC name for the given alkane needs to be determined.
Concept Introduction: According to the IUPAC rule, numbering of atom is done to find the longest continuous carbon chain in the molecule. After that name of groups attached to the chain are identified. The location of substituent groups is designated by numbers and name. The last step is to assemble the name by listing the groups in alphabetical order. Prefixes such as di, tri, tetra etc. are used to represent groups of same kind.
(c)
Explanation of Solution
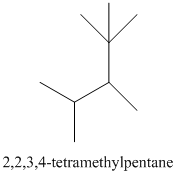
The given structure is as follows:
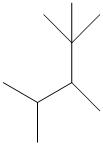
The numbering will be done according to the longest carbon chain in the molecule.
The numbering can be done as follows:
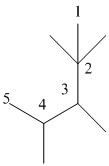
Thus, there are 5 carbon atoms in the parent chain. Also, there are 2 methyl groups at 2nd position, 1 methyl group at 3rd position and 1 methyl group at 4th position. There are total 4 methyl substituents.
Therefore, the IUPAC name of the molecule will be:
(d)
Interpretation: The IUPAC name for the given cycloalkane needs to be determined.
Concept Introduction: According to the IUPAC rule, numbering of atom is done to find the longest continuous carbon chain in the molecule. After that name of groups attached to the chain are identified. The location of substituent groups is designated by numbers and name. The last step is to assemble the name by listing the groups in alphabetical order. Prefixes such as di, tri, tetra etc. are used to represent groups of same kind.
(d)
Explanation of Solution
The given structure is as follows:
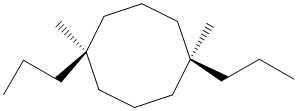
It contains a cycloalkane with 8 carbon atoms. Two substituents are same thus, numbering will start from any one of the substituent side.
The numbering can be represented as follows:
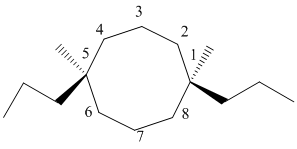
From the above structure, there is 1 methyl and 1 propyl group at 1st and 6th position.
Thus, IUPAC name will be as follows:
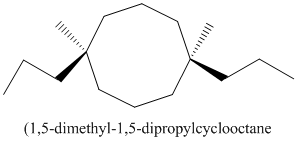
Stereochemistry can also be determined by giving priority to the groups attached.
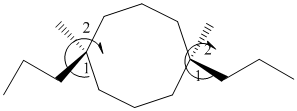
The 1-2 movement is clockwise but the lowest priority group is in front of the curved arrow so assignment will be reversed. Here, clockwise is S and anti or counterclockwise is R. Since, in the above structure the movement is clockwise so the configuration will be S for both sides. The IUPAC name with configuration will be:
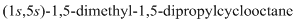
(e)
Interpretation: The IUPAC name for the given cycloalkane needs to be determined.
Concept Introduction: According to the IUPAC rule, numbering of atom is done to find the longest continuous carbon chain in the molecule. After that name of groups attached to the chain are identified. The location of substituent groups is designated by numbers and name. The last step is to assemble the name by listing the groups in alphabetical order. Prefixes such as di, tri, tetra etc. are used to represent groups of same kind.
(e)
Explanation of Solution
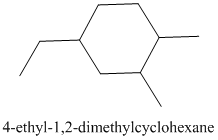
The given structure is as follows:
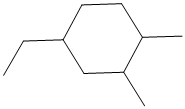
It contains a cycloalkane ring with 6 carbon atoms.
The numbering is represented as follows:
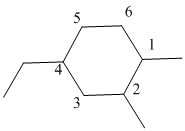
From the above structure, there is 1 methyl group at 1st position, 1 methyl group at 2nd position and 1 ethyl group at 4th position.
Thus, IUPAC name of the molecule will be:
(f)
Interpretation: The IUPAC name for the given alkane needs to be determined.
Concept Introduction: According to the IUPAC rule, numbering of atom is done to find the longest continuous carbon chain in the molecule. After that name of groups attached to the chain are identified. The location of substituent groups is designated by numbers and name. The last step is to assemble the name by listing the groups in alphabetical order. Prefixes such as di, tri, tetra etc. are used to represent groups of same kind.
(f)
Explanation of Solution
The given structure is as follows:
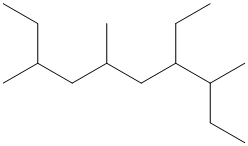
The numbering will be done according to the longest carbon chain in the molecule.
The numbering can be done as follows:
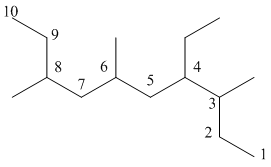
From the above structure, there is 1 ethyl group at 4th position and 1 methyl group each at 3rd, 6th and 8th carbon atom in the chain.
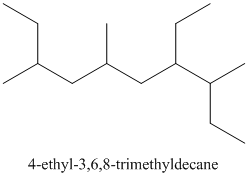
Thus, the IUPAC name of the molecule will be:
Want to see more full solutions like this?
Chapter 11 Solutions
Introduction To General, Organic, And Biochemistry
- can someone answer this pleasearrow_forwardcan someone do the reaction mechanism for this reaction and draw the molecules for Q2 and q3arrow_forwardIn this question, the product of the aldol condensation is shown. What would be the reactants for this product? Please provide a detailed explanation, as well as a drawing showing how the reactants will react to produce the product.arrow_forward
- 7. Propene undergoes a hydration reaction with water in the presence of an acid. a. There are two possible products for this reaction, both with the formula C,H,O. Show their structural formulas and names. (A1, B2) SCH4UR Name: (answer for part a. here!) VER 3 2021-2022 b. Which of the two products do you predict will form. Explain your choice using details from your learning. (B3)arrow_forwardWhat are the major products of the following organic reaction? Please include a detailed explanation as well as a drawing as to how the reaction proceeds.arrow_forwardWhat are the major products of the following reaction? Please provide a detailed explanation and a drawing to show how the reaction proceeds.arrow_forward
- What are the major products of the following organic reaction? Please include a detailed explanation as well as a drawing as to how the reaction proceeds.arrow_forwardPredict the organic product that forms in the reaction below: H + гон OH H+ H+ ☑ O Note: You may assume you have an excess of either reactant if the reaction requires more than one of those molecules to form the product. In the drawing area below, draw the skeletal ("line") structure of the missing organic product X. Explanation Check Click and drag to start drawing a structure. S 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centearrow_forwardIn the analysis of Mg content in a 25 mL sample, a titration volume of 5 mL was obtained using 0.01 M EDTA. Calculate the Mg content in the sample if the Ca content is 20 ppmarrow_forward
- Predict the organic products that form in the reaction below: H. H+ + OH H+ Y Note: You may assume you have an excess of either reactant if the reaction requires more than one of those molecules to form the products. In the drawing area below, draw the skeletal ("line") structures of the missing organic products X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Explanation Check Click and drag to start drawing a structure. G X C © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Access +arrow_forward111 Carbonyl Chem Choosing reagants for a Wittig reaction What would be the best choices for the missing reagents 1 and 3 in this synthesis? 1. PPh3 3 1 2 2. n-BuLi • Draw the missing reagents in the drawing area below. You can draw them in any arrangement you like. Do not draw the missing reagent 2. If you draw 1 correctly, we'll know what it is. • Note: if one of your reagents needs to contain a halogen, use bromine. Explanation Check Click and drag to start drawing a structure. × ©2025 McGraw Hill LLC. All Rights Reserved. Terms of Usearrow_forwardA student proposes the transformation below in one step of an organic synthesis. There may be one or more reactants missing from the left-hand side, but there are no products missing from the right-hand side. There may also be catalysts, small inorganic reagents, and other important reaction conditions missing from the arrow. • Is the student's transformation possible? If not, check the box under the drawing area. . If the student's transformation is possible, then complete the reaction by adding any missing reactants to the left-hand side, and adding required catalysts, inorganic reagents, or other important reaction conditions above and below the arrow. • You do not need to balance the reaction, but be sure every important organic reactant or product is shown. + T X O O лет-ле HO OH HO OH This transformation can't be done in one step.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





