
Interpretation:
The resonance structure of the radical should be identified for the given molecule.
Concept introduction:
Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as radical
Answer to Problem 22PP
Answer
An unpaired electron occupies the allylic position and in is involving in resonance. The arrow pattern of radical resonance is given below (a).

An unpaired electron occupies the allylic position and in is involving in resonance. The arrow pattern of radical resonance is given below (b).
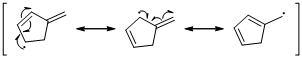
An unpaired electron occupies the allylic position and in is involving in resonance. The arrow pattern of radical resonance is given below (c).
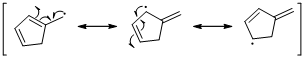
Unpaired electrons resonance with allylic double bonds. The arrow pattern of radical resonance is given below (d).
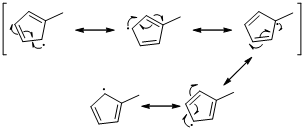
Unpaired electrons resonance with allylic double bonds. The arrow pattern of radical resonance is given below (f)
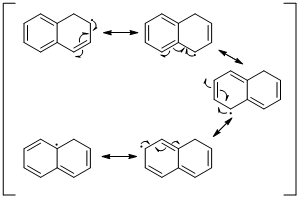
Explanation of Solution
The radical resonance structure of the given molecule
The radical resonance structure of the molecule is given below.

An unpaired electron occupies the allylic position and in is involving in resonance. Here three fishhook arrows were used to show the resonance.
The radical resonance structure of the molecule is given below.

An unpaired electron occupies the allylic position and in is involving in resonance. Here three fishhook arrows were used to show the resonance. The resulting resonance structure exhibits an unpaired electrons, the unpaired electrons allylic to another π bond that also involving resonance. Again three fishhook arrows were used for the resonance.
The radical resonance structure of the molecule is given below.
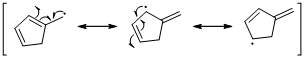
An unpaired electron occupies the allylic position and in is involving in resonance. Here three fishhook arrows were used to show the resonance. The resulting resonance structure exhibits an unpaired electrons, the unpaired electrons allylic to another π bond that also involving resonance. Again three fishhook arrows were used for the resonance.
The radical resonance structure of the molecule is given below.
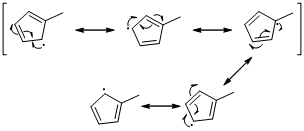
An unpaired electron occupies the allylic position and in is involving in resonance. Here three fishhook arrows were used to show the resonance. The resulting resonance structure exhibits an unpaired electrons, the unpaired electrons allylic to another π bond that also involving resonance. Again three fishhook arrows were used for the resonance. Similarly the five resonances structure is exist in same manner as shown above structure.
The radical resonance structure of the molecule is given below.
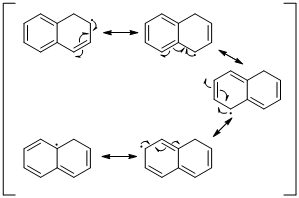
An unpaired electron occupies the allylic position and in is involving in resonance. Here three fishhook arrows were used to show the resonance. The resulting resonance structure exhibits an unpaired electrons, the unpaired electrons allylic to another π bond that also involving resonance. Again three fishhook arrows were used for the resonance. Similarly the five resonances structure is exist in same manner as shown above structure.
Conclusion
The resonance structure of the radical for the given molecule is found.
Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry
- Draw the complete structural formula from each condensed structure. Include all hydrogen atoms.arrow_forwardIndicate how H2O2 intervenes in the synthesis of K4[Co2(C2O4)4(OH)2]. Write the reactions.arrow_forwardExplain how, based on physical gas adsorption isotherms, we can determine whether multi-walled C nanotubes are open at their ends. Explain this.arrow_forward
- can somone answer pleasearrow_forwardConstruct a molecular orbital energy-level diagram for BeH2. Sketch the MO pictures (schematic representation) for the HOMO and LUMO of BeH2 [Orbital Potential Energies, H (1s): -13.6 eV; Be (2s): -9.3 eV, Be (2p): -6.0 eV]arrow_forwardIndicate the isomers of the A(H2O)6Cl3 complex. State the type of isomerism they exhibit and explain it briefly.arrow_forward
- State the formula of the compound potassium μ-dihydroxydicobaltate (III) tetraoxalate.arrow_forwardConsider the reaction of the cyclopentanone derivative shown below. i) NaOCH2CH3 CH3CH2OH, 25°C ii) CH3!arrow_forwardWhat constitutes a 'reference material', and why does its utilization play a critical role in the chemical analysis of food products? Provide examples.arrow_forward
- Explain what calibration is and why it is essential in relation to food analysis. Provide examples.arrow_forwardThe cobalt mu-hydroxide complex cobaltate(III) of potassium is a dinuclear complex. Correct?arrow_forwardThe cobalt mi-hydroxide complex cobaltate(III) of potassium is a dinuclear complex. Correct?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





