
(a)
Interpretation:
Whether the following molecule has a liquid crystalline phase or not should be determined.
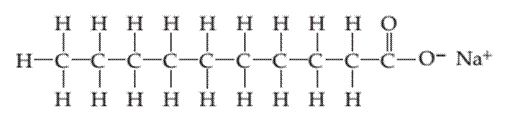
Concept introduction:
Liquid crystals are defined as a phase in which substance exhibits properties of both liquids and solids. Liquid crystal flow like a liquid but their arrangement of the molecule as well as intermolecular forces is like solid.
Liquid crystal molecules are made up of six-membered rings with on terminal polar group, a linkage group and a side chain of carbon atoms. Each carbon atom in liquid crystal molecules has trigonal planar geometry.
The molecules are rigid. The rigidity is increased due to the presence of double-bonded linkage groups such as
The terminal polar groups exhibit strong intermolecular forces such as strong dipole-dipole interaction or dipole−induced dipole interaction and hydrogen bond.
Types of liquid crystal are as follows:
- Nematic Liquid crystal.
- Smectic Liquid crystal.
1. Nematic Liquid crystal: The molecules in the nematic phase are in the same direction and can move around freely very much like that of liquid. In this, the axis is parallel but the ends are not aligned.
2. Smectic Liquid crystal: The molecules in the smectic phase are perpendicular to the plane and are aligned in layers. In these, the long axis is parallel and also their ends are aligned.
(b)
Interpretation:
Whether the following molecule has a liquid crystalline phase or not should be determined.
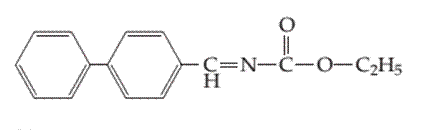
Concept introduction:
Liquid crystals are defined as a phase in which substance exhibits properties of both liquids and solids. Liquid crystal flow like a liquid but their arrangement of the molecule as well as intermolecular forces is like solid.
Liquid crystal molecules are made up of six-membered rings with on terminal polar group, a linkage group and a side chain of carbon atoms. Each carbon atom in liquid crystal molecules has trigonal planar geometry.
The molecules are rigid. The rigidity is increased due to the presence of double-bonded linkage groups such as
The terminal polar groups exhibit strong intermolecular forces such as strong dipole-dipole interaction or dipole−induced dipole interaction and hydrogen bond.
Types of liquid crystal are as follows:
- Nematic Liquid crystal.
- Smectic Liquid crystal.
1. Nematic Liquid crystal: The molecules in the nematic phase are in the same direction and can move around freely very much like that of liquid. In this, the axis is parallel but the ends are not aligned.
2. Smectic Liquid crystal: The molecules in the smectic phase are perpendicular to the plane and are aligned in layers. In these, the long axis is parallel and also their ends are aligned.
(c)
Interpretation:
Whether the following molecule has a liquid crystalline phase or not should be determined.
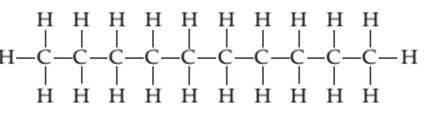
Concept introduction:
Liquid crystals are defined as a phase in which substance exhibits properties of both liquids and solids. Liquid crystal flow like a liquid but their arrangement of the molecule as well as intermolecular forces is like solid.
Liquid crystal molecules are made up of six-membered rings with on terminal polar group, a linkage group and a side chain of carbon atoms. Each carbon atom in liquid crystal molecules has trigonal planar geometry.
The molecules are rigid. The rigidity is increased due to the presence of double-bonded linkage groups such as
The terminal polar groups exhibit strong intermolecular forces such as strong dipole-dipole interaction or dipole−induced dipole interaction and hydrogen bond.
Types of liquid crystal are as follows:
- Nematic Liquid crystal.
- Smectic Liquid crystal.
1. Nematic Liquid crystal: The molecules in the nematic phase are in the same direction and can move around freely very much like that of liquid. In this, the axis is parallel but the ends are not aligned.
2. Smectic Liquid crystal: The molecules in the smectic phase are perpendicular to the plane and are aligned in layers. In these, the long axis is parallel and also their ends are aligned.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
CHEMISTRY-MASTERINGCHEMISTRY W/ETEXT
- Draw the missing intermediate 1 and final product 2 of this synthesis: 1. MeO- H3O+ 1 2 2. PrBr Δ You can draw the two structures in any arrangement you like. Click and drag to start drawing a structure.arrow_forwardWhat is the differences between: Glyceride and phosphoglyceride Wax and Fat Soap and Fatty acid HDL and LDL cholesterol Phospho lipids and sphingosine What are the types of lipids? What are the main lipid components of membrane structures? How could lipids play important rules as signaling molecules and building units? The structure variety of lipids makes them to play significant rules in our body, conclude breifly on this statement.arrow_forwardWhat is the differences between DNA and RNA for the following: - structure - function - type What is the meaning of: - replication - transcription - translation show the base pair connection(hydrogen bond) in DNA and RNAarrow_forward
- What is the IP for a amino acid- give an example what are the types of amino acids What are the structures of proteins The N-Terminal analysis by the Edman method shows saralasin contains sarcosine at the N-terminus. Partial hydrolysis of saralasin with dilute hydrochloric acid yields the following fragments: Try-Val-His Sar-Arg-Val His-Pro-Ala Val- Tyr- Val Arg-Val-Tyr What is the structure of saralasin?arrow_forwardWhat is the IP for a amino acid- give an example what are the types of amino acids What are the structures of proteins The N-Terminal analysis by the Edman method shows saralasin contains sarcosine at the N-terminus. Partial hydrolysis of saralasin with dilute hydrochloric acid yields the following fragments: Try-Val-His Sar-Arg-Val His-Pro-Ala Val- Tyr- Val Arg-Val-Tyr What is the structure of saralasin?arrow_forward> aw the missing intermediates 1 and 2, plus the final product 3, of this synthesis: 1. Eto 1. EtO¯ H3O+ 1 2 2. PrBr 2. PrBr Δ You can draw the three structures in any arrangement you like. 3 Click and drag to start drawing a structure. Explanation Check 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacarrow_forward
- There are various factors that affect an equilibrium. Give 3 of these factors and explain using examples andequations how an equilibrium is affected by these factors. Please remember that this is a communication question so that you are communicating your understanding of the factors that affect and equilibrium.arrow_forwardEEZE LETCHUP ID Draw the most likely conjugate base resulting from this acid-base reaction. Include all lone pairs. Ignore inorganic byproducts. Drawing く NaOCH2CH3 :0: :0: 狗arrow_forwardAnswerarrow_forward
- 2. Provide a clear arrow-pushing mechanism for the following reactions. Do not skip proton transfers, do not combine steps, and make sure your arrows are clear enough to be interpreted without ambiguity. a. CH3 Ph OEt هد Ph CH3 Hint: the species on the left is an ynolate, which behaves a lot like an enolate.arrow_forwardb. CH3 H3C CH3 CH3 H3C an unexpected product, containing a single 9- membered ring the expected product, containing two fused rings H3C-I (H3C)2CuLi an enolatearrow_forwardb. H3C CH3 1. 2. H3O+ H3C MgBr H3Carrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





