
Concept explainers
(a)
Interpretation:
Correct IUPAC name for the structure given has to be written.
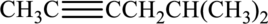
Concept Introduction:
A common nomenclature of naming organic compounds has been developed by IUPAC. By usage of this nomenclature or rules, memorizing of names of organic compounds is not necessary.
IUPAC rules for naming
There are about five rules that has to be followed for naming an alkene and an alkyne. They are,
- The longest continuous carbon chain in the compound that contains double bond or triple has to be identified. This is known as parent compound.
- Suffix “–ane” (in name of
alkane ) is replaced with “-ene” for alkene or “-yne” for alkyne. - Numbering has to be done so that the lowest number is given to the double or triple bond.
- Naming and numbering has to be given for each atom or group that is attached to the parent chain. Numbering has to be done in a way that substituents get the least numbering.
- If the alkenes have more than one double bond they are called as alkadienes (two double bonds) or alkatrienes (three double bonds). Appropriate suffix has to be used depending on the number of multiple bonds present in the compound.
(a)
Explanation of Solution
Given compound and IUPAC name is.
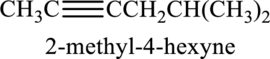
IUPAC name given is wrong as the triple bond is not given the least numbering. Correct IUPAC name for the given structure can be given as shown below.
Longest carbon chain with triple bond is found to contain six carbon atoms. Therefore, the parent alkane is hexane. As a triple bond is present, the alkyne name is hexyne.
Numbering has to be given in a way that the triple bond gets the least numbering. In this case, triple bond is present between carbon-2 and carbon-3. Therefore, the parent alkyne is 2-hexyne.
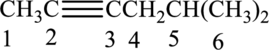
The substituent present in the given structure are a methyl group on carbon-5. Number has to be added before the substituent names which indicate the carbon atom in which it is present. This gives the name of alkene as 5-methyl-2-hexyne.
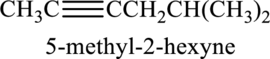
Longest carbon chain containing triple bond is hexane. Position of triple bond is 2-hexyne. Substituent present in the chain is 5-methyl. Correct IUPAC name of the structure given is 5-methyl-2-hexyne.
(b)
Interpretation:
Correct IUPAC name for the structure given has to be written.
Concept Introduction:
Refer part (a).
(b)
Explanation of Solution
Given compound and IUPAC name is.
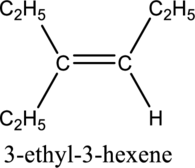
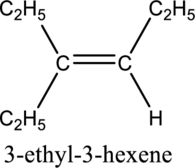
Longest carbon chain with double bond is found to contain six carbon atoms. Therefore, the parent alkane is hexane. As a double bond is present, the alkene name is hexene.
Numbering has to be given in a way that the triple bond gets the least numbering. In this case, triple bond is present between carbon-3 and carbon-4. Therefore, the parent alkyne is 3-hexene.
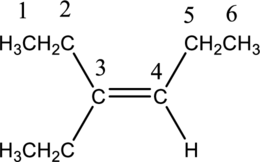
The substituent present in the given structure are an ethyl group on carbon-3. Number has to be added before the substituent names which indicate the carbon atom in which it is present. This gives the name of alkene as 3-ethyl-3-hexene.
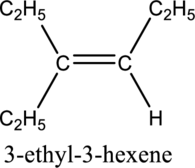
Longest carbon chain containing double bond is hexene. Position of double bond is 3-hexene. Substituent present in the chain is 3-ethyl. IUPAC name of the structure given is 3-methyl-3-hexene. Therefore, the given name is correct.
(c)
Interpretation:
Correct IUPAC name for the structure given has to be written.
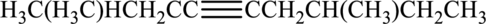
Concept Introduction:
Refer part (a).
(c)
Explanation of Solution
Given compound and IUPAC name is.
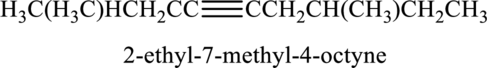
IUPAC name given is wrong as the parent chain considered and the substituents are not given correctly. Correct IUPAC name for the given structure can be given as shown below.
Longest carbon chain with triple bond is found to contain nine carbon atoms. Therefore, the parent alkane is nonane. As a triple bond is present, the alkyne name is nonyne.
Numbering has to be given in a way that the triple bond gets the least numbering. In this case, triple bond is present between carbon-4 and carbon-5. Therefore, the parent alkyne is 4-nonyne.
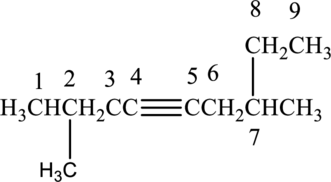
The substituent present in the given structure two methyl groups, each on carbon-2 and carbon-7. Number has to be added before the substituent names which indicate the carbon atom in which it is present. This gives the name of alkyne as 2,7-dimethyl-4-nonyne.
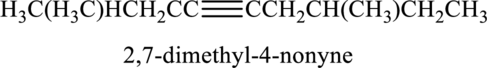
Longest carbon chain containing triple bond is nonane. Position of triple bond is 4-nonyne. Substituent present in the chain is 2,7-dimethyl. Correct IUPAC name of the structure given is 2,7-dimethyl-4-nonyne.
(d)
Interpretation:
Correct IUPAC name for the structure given has to be written.
Concept Introduction:
Refer part (a).
(d)
Explanation of Solution
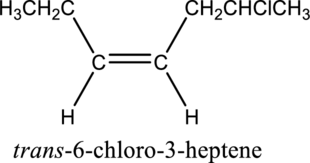
Given compound and IUPAC name is.
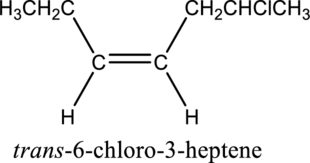
The configuration given in the IUPAC name is wrong as both hydrogen atoms are present on same side of the double bond. Correct IUPAC name can be given as shown below.
Numbering has to be given in a way that the double bond gets the least numbering. In this case, double bond is present between carbon-3 and carbon-4. Therefore, the parent alkyne is 3-heptene.
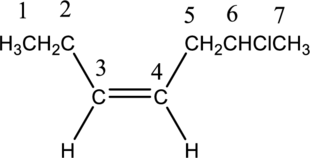
The substituent present in the given structure is a chloro group. Number has to be added before the substituent names which indicate the carbon atom in which it is present. This gives the name of alkene as 6-chloro-3-heptene. Looking into the stereo information, there are hydrogen atoms present on the same side of the double bond. Hence, the structure given is in cis conformation. Therefore, the name of the structure can be given as cis-6-chloro-3-heptene.
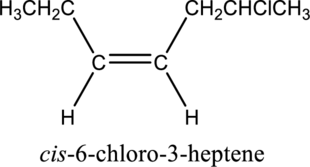
Longest carbon chain containing double bond is heptene. Position of double bond is 3-heptene. Substituent present in the chain is 6-chloro. Configuration is “cis”. Correct IUPAC name of the structure given is cis-6-chloro-3-heptene.
(e)
Interpretation:
Correct IUPAC name for the structure given has to be written.
Concept Introduction:
Refer part (a).
(e)
Explanation of Solution
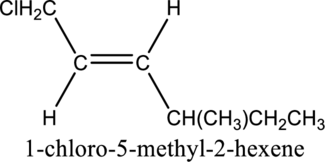
Given compound and IUPAC name is.
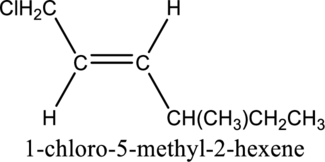
The configuration given in the IUPAC name is wrong as configuration is not given and the position of methyl substituent is wrong. Correct IUPAC name can be given as shown below.
Parent carbon chain is found to contain six carbon atoms. Therefore, the alkane is hexane.
Numbering has to be given in a way that the double bond gets the least numbering. In this case, double bond is present between carbon-2 and carbon-3. Therefore, the parent alkyne is 2-hexene.
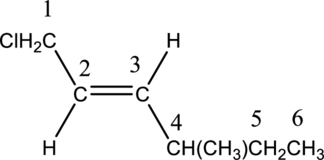
The substituent present in the given structure are a chloro group and a methyl group. Number has to be added before the substituent names which indicate the carbon atom in which it is present. This gives the name of alkene as 1-chloro-4-methyl. Looking into the stereo information, there are hydrogen atoms present on opposite side of the double bond. Hence, the structure given is in trans conformation. Therefore, the name of the structure can be given as trans-1-chloro-4-methyl2-hexene.
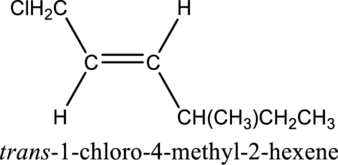
Longest carbon chain containing double bond is hexene. Position of double bond is 2-hexene. Substituent present in the chain is 1-chloro-4-methyl. Configuration is “trans”. Correct IUPAC name of the structure given is trans-1-chloro-4-methyl-2-hexene.
Want to see more full solutions like this?
Chapter 11 Solutions
GENERAL,ORGANIC+BIOCHEM (LOOSELEAF)
- Give reason(s) for six from the followings [using equations if possible] a. Addition of sodium carbonate to sulfanilic acid in the Methyl Orange preparation. b. What happened if the diazotization reaction gets warmed up by mistake. c. Addition of sodium nitrite in acidified solution in MO preparation through the diazotization d. Using sodium dithionite dihydrate in the second step for Luminol preparation. e. In nitroaniline preparation, addition of the acid mixture (nitric acid and sulfuric acid) to the product of step I. f. What is the main reason of the acylation step in nitroaniline preparation g. Heating under reflux. h. Fusion of an organic compound with sodium. HAND WRITTEN PLEASEarrow_forwardedict the major products of the following organic reaction: u A + ? CN Some important notes: • Draw the major product, or products, of the reaction in the drawing area below. • If there aren't any products, because no reaction will take place, check the box below the drawing area instead. Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. Explanation Check Click and drag to start drawing a structure. Х © 2025 McGraw Hill LLC. All Rights Reserved. Te LMUNDARYarrow_forwardSketch the intermediates for A,B,C & D.arrow_forward
- Can the molecule on the right-hand side of this organic reaction be made in good yield from no more than two reactants, in one step, by moderately heating the reactants? O ? A . If your answer is yes, then draw the reactant or reactants in the drawing area below. You can draw the reactants in any arrangement you like. . If your answer is no, check the box under the drawing area instead. Explanation Check Click and drag to start drawing a structure. ㅇ 80 F5 F6 A 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Cente FIGarrow_forwardIn methyl orange preparation, if the reaction started with 0.5 mole of sulfanilic acid to form the diazonium salt of this compound and then it converted to methyl orange [0.2 mole]. If the efficiency of the second step was 50%, Calculate: A. Equation(s) of Methyl Orange synthesis: Diazotization and coupling reactions. B. How much diazonium salt was formed in this reaction? C. The efficiency percentage of the diazotization reaction D. Efficiency percentage of the whole reaction.arrow_forwardHand written equations pleasearrow_forward
- Hand written equations pleasearrow_forward> each pair of substrates below, choose the one that will react faster in a substitution reaction, assuming that: 1. the rate of substitution doesn't depend on nucleophile concentration and 2. the products are a roughly 50/50 mixture of enantiomers. Substrate A Substrate B Faster Rate X Ś CI (Choose one) (Choose one) CI Br Explanation Check Br (Choose one) © 2025 McGraw Hill LLC. All Rights Farrow_forwardNMR spectrum of ethyl acetate has signals whose chemical shifts are indicated below. Which hydrogen or set of hydrogens corresponds to the signal at 4.1 ppm? Select the single best answer. The H O HỌC—C—0—CH, CH, 2 A ethyl acetate H NMR: 1.3 ppm, 2.0 ppm, 4.1 ppm Check OA B OC ch B C Save For Later Submit Ass © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center |arrow_forward
- How many signals do you expect in the H NMR spectrum for this molecule? Br Br Write the answer below. Also, in each of the drawing areas below is a copy of the molecule, with Hs shown. In each copy, one of the H atoms is colored red. Highlight in red all other H atoms that would contribute to the same signal as the H already highlighted red Note for advanced students: In this question, any multiplet is counted as one signal. 1 Number of signals in the 'H NMR spectrum. For the molecule in the top drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. Check For the molecule in the bottom drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. O ✓ No additional Hs to color in top molecule ง No additional Hs to color in bottom…arrow_forwardin the kinetics experiment, what were the values calculated? Select all that apply.a) equilibrium constantb) pHc) order of reactiond) rate contstantarrow_forwardtrue or false, given that a 20.00 mL sample of NaOH took 24.15 mL of 0.141 M HCI to reach the endpoint in a titration, the concentration of the NaOH is 1.17 M.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





