
Organic Chemistry-Package(Custom)
4th Edition
ISBN: 9781259141089
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Question
Chapter 11, Problem 11.41P
Interpretation Introduction
Interpretation:
To determine two different
Concept introduction:
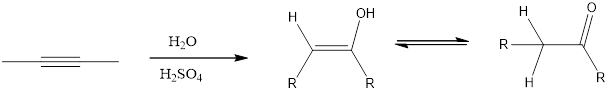
Addition of H2O to an alkyne resembles similar to the acid-catalyzed addition of H2O to an
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
3. Assign absolute configuration (Rors) to each chirality center.
a.
H
Nitz
C.
он
b.
0
H-C. C
H
7
C.
་-4
917-417
refs
H
1つ
८
ડુ
d.
Но
f.
-2-
01
Ho
-OH
2HN
How many signals do you expect in the H NMR spectrum for this molecule?
Br
Br
Write the answer below.
Also, in each of the drawing areas below is a copy of the molecule, with Hs shown. In each copy, one of the H atoms is colored red. Highlight in red all other H
atoms that would contribute to the same signal as the H already highlighted red.
Note for advanced students: In this question, any multiplet is counted as one signal.
Number of signals in the 'H NMR spectrum.
For the molecule in the top drawing area, highlight in red any other H atoms that will contribute to
the same signal as the H atom already highlighted red.
If no other H atoms will contribute, check the box at right.
No additional Hs to color in top
molecule
For the molecule in the bottom drawing area, highlight in red any other H atoms that will
contribute to the same signal as the H atom already highlighted red.
If no other H atoms will contribute, check the box at right.
No additional Hs to color in bottom
molecule
In the drawing area below, draw the major products of this organic reaction:
1. NaOH
?
2. CH3Br
If there are no major products, because nothing much will happen to the reactant under these reaction conditions, check the box under the drawing area
instead.
No reaction.
Click and drag to start drawing a
structure.
☐ : A
ค
Chapter 11 Solutions
Organic Chemistry-Package(Custom)
Ch. 11 - Draw structures for the three alkynes having...Ch. 11 - Prob. 11.2PCh. 11 - Give the IUPAC name for each compound.Ch. 11 - Give the structures corresponding to each of the...Ch. 11 - Prob. 11.5PCh. 11 - Prob. 11.6PCh. 11 - Which bases can deprotonate acetylene? The pKa...Ch. 11 - Draw the organic products formed when each alkyne...Ch. 11 - Prob. 11.9PCh. 11 - Problem 11.9 Draw the products formed when is...
Ch. 11 - Explain the following result. Although alkenes...Ch. 11 - Problem 11.11 Draw the keto tautomer of each...Ch. 11 - Prob. 11.13PCh. 11 - a Draw two different enol tautomers of...Ch. 11 - Prob. 11.15PCh. 11 - Prob. 11.16PCh. 11 - Prob. 11.17PCh. 11 - Problem. 11.17 Show how , and can be used to...Ch. 11 - Prob. 11.19PCh. 11 - Draw the products of each reaction. a. b.Ch. 11 - Prob. 11.21PCh. 11 - Problem 11.21 Use retrosynthetic analysis to show...Ch. 11 - Prob. 11.23PCh. 11 - Give the IUPAC name for each compound. a. b.Ch. 11 - Prob. 11.25PCh. 11 - 11.25 Answer the following questions about...Ch. 11 - Prob. 11.27PCh. 11 - Give the IUPAC name for each alkyne.Ch. 11 - Prob. 11.29PCh. 11 - Which of the following pairs of compounds...Ch. 11 - Prob. 11.31PCh. 11 - 11.30 How is each compound related to A? Choose...Ch. 11 - Prob. 11.33PCh. 11 - Prob. 11.34PCh. 11 - Prob. 11.35PCh. 11 - 11.33 Draw the products formed when is treated...Ch. 11 - Draw the products formed when 3-hexyne is treated...Ch. 11 - Prob. 11.38PCh. 11 - Prob. 11.39PCh. 11 - What alkynes give each of the following ketones as...Ch. 11 - Prob. 11.41PCh. 11 - 11.37 What alkyne gives each compound as the only...Ch. 11 - Prob. 11.43PCh. 11 - Draw the structure of compounds A-E in the...Ch. 11 - Prob. 11.45PCh. 11 - Prob. 11.46PCh. 11 - 11.42 What reactions are needed to convert alcohol...Ch. 11 - Prob. 11.48PCh. 11 - Prob. 11.49PCh. 11 - 11.45 Explain the following statement. Although ...Ch. 11 - Draw a stepwise mechanism for the following...Ch. 11 - Prob. 11.52PCh. 11 - Prob. 11.53PCh. 11 - Prob. 11.54PCh. 11 - Prob. 11.55PCh. 11 - Synthesize each compound from acetylene. You may...Ch. 11 - Prob. 11.57PCh. 11 - Prob. 11.58PCh. 11 - 11.55 Devise a synthesis of the ketone, , from ...Ch. 11 - 11.56 Devise a synthesis of each compound using ...Ch. 11 - Prob. 11.61PCh. 11 - Prob. 11.62PCh. 11 - 11.60 Draw a stepwise mechanism for the following...Ch. 11 - Draw a stepwise mechanism for the following...Ch. 11 - Prob. 11.65PCh. 11 - Write a stepwise mechanism for each of the...Ch. 11 - Prob. 11.67PCh. 11 - 11.65 Explain why an optically active solution of ...
Knowledge Booster
Similar questions
- Predict the major products of the following organic reaction: NC Δ ? Some important Notes: • Draw the major product, or products, of the reaction in the drawing area below. • If there aren't any products, because no reaction will take place, check the box below the drawing area instead. • Be sure to draw bonds carefully to show important geometric relationships between substituents. Note: if your answer contains a complicated ring structure, you must use one of the molecular fragment stamps (available in the menu at right) to enter the ring structure. You can add any substituents using the pencil tool in the usual way. Click and drag to start drawing a structure. Х аarrow_forwardPredict the major products of this organic reaction. Be sure you use dash and wedge bonds to show stereochemistry where it's important. + ☑ OH 1. TsCl, py .... 文 P 2. t-BuO K Click and drag to start drawing a structure.arrow_forwardConsider this organic reaction: ( Draw the major products of the reaction in the drawing area below. If there won't be any major products, because this reaction won't happen at a significant rate, check the box under the drawing area instead. Click and drag to start drawing a structure. Х : а ค 1arrow_forward
- In the drawing area below, draw the major products of this organic reaction: If there are no major products, because nothing much will happen to the reactant under these reaction conditions, check the box under the drawing area instead. 1. NaH 2. CH3Br ? Click and drag to start drawing a structure. No reaction. : ☐ Narrow_forward+ Predict the major product of the following reaction. : ☐ + ☑ ค OH H₂SO4 Click and drag to start drawing a structure.arrow_forwardConsider this organic reaction: ... OH CI Draw the major products of the reaction in the drawing area below. If there won't be any major products, because this reaction won't happen at a significant rate, check the box under the drawing area instead. ☐ No Reaction. Click and drag to start drawing a structure. : аarrow_forward
- Consider the following reactants: Br Would elimination take place at a significant rate between these reactants? Note for advanced students: by significant, we mean that the rate of elimination would be greater than the rate of competing substitution reactions. yes O no If you said elimination would take place, draw the major products in the upper drawing area. If you said elimination would take place, also draw the complete mechanism for one of the major products in the lower drawing area. If there is more than one major product, you may draw the mechanism that leads to any of them. Major Products:arrow_forwardDraw one product of an elimination reaction between the molecules below. Note: There may be several correct answers. You only need to draw one of them. You do not need to draw any of the side products of the reaction. OH + ! : ☐ + Х Click and drag to start drawing a structure.arrow_forwardFind one pertinent analytical procedure for each of following questions relating to food safety analysis. Question 1: The presence of lead, mercury and cadmium in canned tuna Question 2: Correct use of food labellingarrow_forward
- Formulate TWO key questions that are are specifically in relation to food safety. In addition to this, convert these questions into a requirement for chemical analysis.arrow_forwardWhat are the retrosynthesis and forward synthesis of these reactions?arrow_forwardWhich of the given reactions would form meso product? H₂O, H2SO4 III m CH3 CH₂ONa CH3OH || H₂O, H2SO4 CH3 1. LiAlH4, THF 2. H₂O CH3 IVarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
