
Concept explainers
(a)
Interpretation:
The distance between
Concept introduction:
The bond length or bond distance is the average distance between the nuclei of two bonded atoms in a molecule. When two similar atoms are bonded together, half of the bond length is known as the covalent radius. The bond length depends on the number of bonded of two atoms. The unit of bond length is picometer.
In single, double and triple bonds, the order of bond length is as follows:
(a)
Answer to Problem 10.95P
Explanation of Solution
The total number of electrons in
So
The Lewis structure of

The two hydrogen atoms are separated by two
The total distance between
The bond length between the two atoms in a molecule depends not only on the atoms but also on other factors like orbital hybridization and the steric nature of the substituents.
(b)
Interpretation:
The distance between
Concept introduction:
The bond length or bond distance is the average distance between the nuclei of two bonded atoms in a molecule. When two similar atoms are bonded together, half of the bond length is known as the covalent radius. The bond length depends on the number of bonded of two atoms. The unit of bond length is picometer.
In single, double and triple bonds, the order of bond length is as follows:
(b)
Answer to Problem 10.95P
Explanation of Solution
The total number of electrons in
So
The Lewis structure of
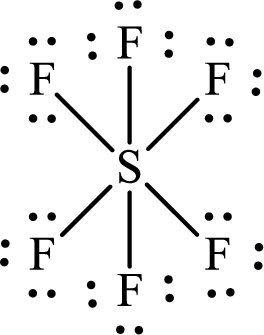
The two fluorine atoms that are opposite to the sulfur atom are separated by two
The total distance between
The adjacent fluorine atoms are present at the two corners of a right-angled triangle. The two sides of the triangle are the bond lengths of
The distance between the adjacent
Or,
Substitute
The bond length between the two atoms in a molecule depends not only on the atoms but also on other factors like orbital hybridization and the steric nature of the substituents.
(c)
Interpretation:
The distance between equatorial
Concept introduction:
The bond length or bond distance is the average distance between the nuclei of two bonded atoms in a molecule. When two similar atoms are bonded together, half of the bond length is known as the covalent radius. The bond length depends on the number of bonded of two atoms. The unit of bond length is picometer.
In single, double and triple bonds, the order of bond length is as follows:
(c)
Answer to Problem 10.95P
Explanation of Solution
The total number of electrons in
So
The Lewis structure of
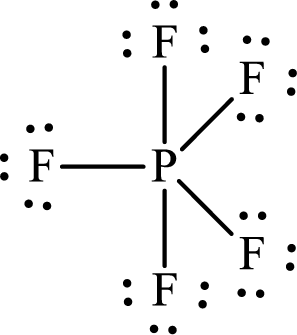
The adjacent fluorine atoms are present at the corners of a triangle with
The distance between the adjacent
Or,
Substitute
The bond length between the two atoms in a molecule depends not only on the atoms but also on other factors like orbital hybridization and the steric nature of the substituents.
Want to see more full solutions like this?
Chapter 10 Solutions
CHEMISTRY(LOOSELEAF) W/CONNECT+EBOOK
- Consider this molecule: How many H atoms are in this molecule? How many different signals could be found in its 'H NMR spectrum? Note: A multiplet is considered one signal. ☐arrow_forwardStudy this 'H NMR spectrum, and then answer the questions about it in the table below. Check 1.0- 0.5- 0.0 10.0 9.0 8.0 7.0 6.0 5.0 4.0 3.0 2.0 1.0 0.0 What unit symbol should be written on the horizontal axis? What is the chemical shift & of the doublet? If there is no doublet, just check the box instead. Give your answer to 2 significant digits. What is the chemical shift of the signal immediately upfield of the doublet? If there is no doublet, or no signal upfield of it, check the box instead. What is the chemical shift & of the least deshielded proton? If you can't tell without more information, check the box instead. 血 8 = ☐ There is no doublet. 8 = ☐ No such signal. 8 = 0 Need more information.arrow_forwardhow many moles of H2O2 are required to react with 11g of N2H4 according to the following reaction? (atomic weights: N=14.01, H=1.008, O= 16.00) 7H2O2 + N2H4 -> 2HNO3 + 8H20arrow_forward
- calculate the number of moles of H2 produced from 0.78 moles of Ga and 1.92 moles HCL? 2Ga+6HCL->2GaCl3+3H2arrow_forwardan adult human breathes 0.50L of air at 1 atm with each breath. If a 50L air tank at 200 atm is available, how man y breaths will the tank providearrow_forwardWhat are the advantages and/or disadvantages of using the MOHR titration method & AOEC method?arrow_forward
- Are there any alternative methods better than the MOHR titration to quantitatively determine salt in a sample?arrow_forwardhybridization of nitrogen of complex moleculesarrow_forwardUsing reaction free energy to predict equilibrium composition Consider the following equilibrium: 2NO2 (g) = N2O4(g) AGº = -5.4 kJ Now suppose a reaction vessel is filled with 4.53 atm of dinitrogen tetroxide (N2O4) at 279. °C. Answer the following questions about this system: Under these conditions, will the pressure of N2O4 tend to rise or fall? Is it possible to reverse this tendency by adding NO2? In other words, if you said the pressure of N2O4 will tend to rise, can that be changed to a tendency to fall by adding NO2? Similarly, if you said the pressure of N2O4 will tend to fall, can that be changed to a tendency to '2' rise by adding NO2? If you said the tendency can be reversed in the second question, calculate the minimum pressure of NO 2 needed to reverse it. Round your answer to 2 significant digits. 00 rise ☐ x10 fall yes no ☐ atm G Ar 1arrow_forward
- Why do we analyse salt?arrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. H H CH3OH, H+ H Select to Add Arrows H° 0:0 'H + Q HH ■ Select to Add Arrows CH3OH, H* H. H CH3OH, H+ HH ■ Select to Add Arrows i Please select a drawing or reagent from the question areaarrow_forwardWhat are examples of analytical methods that can be used to analyse salt in tomato sauce?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





