
(a)
Interpretation: The compound that is likely to be more soluble in  among the given pair is to be predicted.
among the given pair is to be predicted.
Concept introduction: The polarity of solute and solvent determines the solubility solution. Solute and solvent of same polarity are soluble with each other and insoluble if polarity is different.
To determine: The compound that is likely to be more soluble in  among the given pair.
among the given pair.
(a)
Answer to Problem 10.48QP
Solution
The solubility of 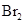 in
in  is more than that of
is more than that of 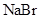 .
.
Explanation of Solution
Explanation
The dipole moment of the given compound is important for its solubility in  . The dipole moment of
. The dipole moment of  is zero and it solubilizes only non-polar compounds.
is zero and it solubilizes only non-polar compounds.
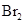 is a non-polar compound whereas
is a non-polar compound whereas 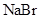 is a polar compound. Therefore,
is a polar compound. Therefore, 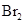 is more soluble in
is more soluble in  than
than 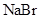 .
.
(b)
To determine: The compound that is likely to be more soluble in  among the given pair.
among the given pair.
(b)
Answer to Problem 10.48QP
Solution
The solubility of 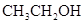 in
in  is more than that of
is more than that of 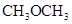 .
.
Explanation of Solution
Explanation
The polarity of ethanol in comparison to acetone is more because in ethanol, oxygen is attached to one carbon atom, whereas in acetone it lies between two carbon atoms and they cancel out each other.
Therefore, acetone being less polar solubilizes more in  than ethanol.
than ethanol.
(c)
To determine: The compound that is likely to be more soluble in  among the given pair.
among the given pair.
(c)
Answer to Problem 10.48QP
Solution
The solubility of 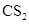 in
in  is more than that of
is more than that of 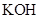 .
.
Explanation of Solution
Explanation
The given compound 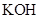 is ionic compound and
is ionic compound and 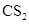 is a non-ionic compound.
is a non-ionic compound.
Ionic compounds are polar in nature and solubilize in polar solvent.
Therefore, 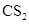 being non-polar compound solubilizes more in
being non-polar compound solubilizes more in  than
than 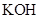 .
.
(d)
To determine: The compound that is likely to be more soluble in  among the given pair.
among the given pair.
(d)
Answer to Problem 10.48QP
Solution
The solubility of 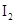 in
in  is more than that of
is more than that of 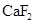 .
.
Explanation of Solution
Explanation
The given compounds are 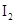 and
and 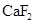 . Iodine is non-polar compound as its dipole moment is zero whereas
. Iodine is non-polar compound as its dipole moment is zero whereas 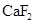 is ionic molecule and polar in nature. Therefore,
is ionic molecule and polar in nature. Therefore, 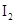 being non-polar compound solubilizes more in
being non-polar compound solubilizes more in  than
than 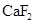 .
.
Conclusion
- a. The solubility of
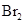 in
in  is more than that of
is more than that of 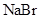 .
. - b. The solubility of
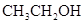 in
in  is more than that of
is more than that of 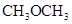 .
. - c. The solubility of
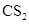 in
in  is more than that of
is more than that of 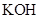 .
. - d. The solubility of
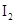 in
in  is more than that of
is more than that of 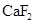 .
.
Want to see more full solutions like this?
Chapter 10 Solutions
Chemistry Smartwork Access Code Fourth Edition
- PLEASE READ!!! I DONT WANT EXAMPLES, I DONT WANT WORDS OR PARAGRAPHS!!! PLEASE I UNDERSTAND THE BASICS BUT THIS IS AN EXCEPTION THAT EVEN THE INTERNET CANT HELP!!!! THIS IS THE THIRD TIME I'VE SENT THOSE QUESTIONS SO PLEASE DONT RESEND THE SAME STUFF, ITS NOT HELPING ME!!! I ALSO ALREADY TRIED TO DRAW THE MECHANISM MYSELF, SO IF ITS RIGHT PLEASE TELL ME OR TELL ME WHAT I HAVE TO CHANGE!!! First image: I have to SHOW (DRAWING) the mechanism (with arows and structures of molecules) NOT WORDS PLEASE! of the reaction at the bottom. Also I have to show by mecanism why the reaction wouldn't work if the alcohol was primary Second image: I have to show the mechanism (IMAGE) (with arrows and structures of the molecules) NOT WORDS PLEASE !! for the reaction on the left, where the alcohol A is added fast in one portion HOMEWORK, NOT EXAM!! ALL DETAILS ARE IN THE IMAGES PLEASE LOOK AT THE IMAGES, DONT LOOK AT THE AI GENERATED TEXT!!!arrow_forwardWrite the molecular formula for a compound with the possible elements C, H, N and O that exhibits a molecular ion at M+ = 85.0899. Exact Masses of the Most Abundant Isotope of Selected Elements Isotope Natural abundance (%) Exact mass 1H 99.985 1.008 12C 98.90 12.000 14N 99.63 14.003 160 99.76 15.995 Molecular formula (In the order CHNO, with no subscripts)arrow_forwardUse the data below from an electron impact mass spectrum of a pure compound to deduce its structure. Draw your structure in the drawing window. Data selected from the NIST WebBook, https://webbook.nist.gov/chemistry/ m/z Relative intensity 59 3.0 58 64 43 100 15 23 • You do not have to consider stereochemistry. •You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. + n[] 85 // ? CH4 Previous Nextarrow_forward
- Write the molecular formula for a compound with the possible elements C, H, N and O that exhibits a molecular ion at M* = 128.0632. Exact Masses of the Most Abundant Isotope of Selected Elements Isotope Natural abundance (%) Exact mass 1H 99.985 12C 98.90 14N 99.63 160 99.76 Molecular formula 1.008 12.000 14.003 15.995 (In the order CHNO, with no subscripts)arrow_forwardCan I please get help with this? And can I please the lowest possible significant number?arrow_forwardWhat is the molar mass of a gas that takes three times longer to effuse than helium?arrow_forward
- First image: I have to show the mecanism (with arows and structures) of the reaction at the bottom. Also I have to show by mecanism why the reaction wouldn't work if the alcohol was primary Second image: I have to show the mecanism (with arrows and structures) for the reaction on the left, where the alcohol A is added fast in one portion its not an examarrow_forwardwhat is the skeletal structure of a tertiary alkyl fluoride with six carbon atoms and no rings.arrow_forwardOne step of glycolysis is a retro-aldol reaction (aldolase) to produce ATP.Below is the aldol reaction of the equilibrium. Show the mechanism for the base catalyzed reaction. *see imagearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





