
Study Guide for Chemistry: The Central Science
14th Edition
ISBN: 9780134554075
Author: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus, James C. Hill
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 4E
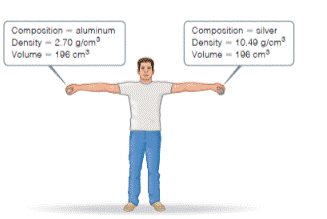
Consider the two spheres shown here, one made of silver and the other of aluminum.
- What is the mass of each sphere in kg?
- The force of gravity acting on an object is F = mg, where m is the mass of an object and g is the acceleration of gravity (9.8 m/s2). How much work do you do on each sphere it you raise it from the floor to a height of 2.2 m?
- Does the act of lifting the sphere off the ground increase the potential energy of the aluminum sphere by a larger, smaller, or same amount as the silver sphere?
- If you release the spheres simultaneously, they will have the same velocity when they hit the ground. Will they have the same kinetic energy? If not, which sphere will have more kinetic energy? [Section 1.4 Q]
Expert Solution & Answer
Learn your wayIncludes step-by-step video

schedule04:21
Students have asked these similar questions
achieve.macmillanlearning.com
Canvas
EA eac
h Hulu
YouTube
G 3 methyl cyclobutanol - Google Search
Ranking Phenol Acidity
Course -236 - Organic Chemistry - Mac...
←
Assessment
Completed 10 of 22 Questions
1 +
Netflix
paramount plus
chem hw
Galdehyde reaction with grignard reagent...
b My Questions | bartleby
M Inbox - chenteislegit@gmail.com - Gmail
Due: Fri, Jan 31
Resources
Solution
Penalized
? Hint
Submit Answer
Use retrosynthetic analysis to suggest two paths to synthesize 2-methyl-3-hexanol using the Grignard reaction. (Click and drag
the appropriate image to the correct position in the reactions.)
Route 1
Aldehyde 1
or
+98
Aldehyde 2
Route 2
Q6
+100
Solved in 1 attempt
Q7
+95
Solved in 2 attempts
Q8
+98
Unlimited attempts
possible
+
+
Grignard 1
OH
H3O+
Grignard 2
Answer Bank
Q9
+90
MgBr
Unlimited attempts
possible
CH3CH2CH2MgBr
Q10
Unlimited attempts
Q11
?
?
+100
in 1 attempt
2-methyl-3-hexanol
CH3CH2MgBr
H
H
о
H
Attempt 3
2) (4 pt) After the reaction was completed, the student collected the following data. Crude
product data is the data collected after the reaction is finished, but before the product
is purified. "Pure" product data is the data collected after attempted purification using
recrystallization.
Student B's data:
Crude product data
"Pure"
product data
after
recrystallization
Crude mass: 0.93 g grey solid
Crude mp: 96-106 °C
Crude % yield:
Pure mass: 0.39 g white solid
Pure mp: 111-113 °C
Pure % yield:
a) Calculate the crude and pure percent yields for the student's reaction.
b) Summarize what is indicated by the crude and pure melting points.
Don't used hand raiting
Chapter 1 Solutions
Study Guide for Chemistry: The Central Science
Ch. 1.2 - Practice Exercise 1 Which of the following is the...Ch. 1.2 - Aspirin is composed of 60.0% carbon, 4.5%...Ch. 1.5 - Practice Exercise 1 Which of the following weights...Ch. 1.5 - Practice Exercise 2 How many picometers are there...Ch. 1.5 - Practice Exercise 1 Using Wolfram Alpha...Ch. 1.5 - Practice Exercise 2 Ethylene glycol, the major...Ch. 1.5 - Practice Exercise 1 Platinum, Pt. is one of the...Ch. 1.5 - Practice Exercise 2 Calculate the density of a...Ch. 1.5 - Which of the following objects has the greatest...Ch. 1.5 - Prob. 1.5.2PE
Ch. 1.6 - Which of the following numbers in your personal...Ch. 1.6 - Practice Exercise 2 The back inside cover of the...Ch. 1.6 - Practice Exercise 1 An object is determined to...Ch. 1.6 - Practice Exercise 2 How many significant figures...Ch. 1.6 - Ellen recently purchased a new hybrid car and...Ch. 1.6 - Practice Exercise 2 It takes 10.5 s for a sprinter...Ch. 1.6 - Practice Exercise 1 You are asked to determine the...Ch. 1.6 - Practice Exercise 1 At a particular instant in...Ch. 1.7 - Practice Exercise 2 By using a conversion factor...Ch. 1.7 - Practice Exercise 1 Fabiola, who lives in Mexico...Ch. 1.7 - Practice Exercise 2 A car travels 28 mi per gallon...Ch. 1.7 - Practice Exercise 2 A car travels 28 mi per gallon...Ch. 1.7 - Practice Exercise 2 The surface area of Earth is...Ch. 1.7 - Practice Exercise 1 Composite decking is a...Ch. 1.7 - Practice Exercise 2 The density of the organic...Ch. 1.7 - Practice Exercise 2 If the mass of the container...Ch. 1 - Prob. 1ECh. 1 - Prob. 2ECh. 1 - Musical instruments like trumpets and trombones...Ch. 1 - Consider the two spheres shown here, one made of...Ch. 1 - Is the separation method used in brewing a cup of...Ch. 1 - Identify each of the following as measurements of...Ch. 1 - Three spheres of equal size are composed of...Ch. 1 - The three targets from a rifle range shown below...Ch. 1 - What is the length of the pencil in the following...Ch. 1 - How many significant figures should be reported...Ch. 1 - Consider the jar of jelly beans in the photo. To...Ch. 1 - The photo below shows a picture of an agate stone....Ch. 1 - Classify each of the following as a pure substance...Ch. 1 - Classify each of the following as a pure substance...Ch. 1 - 1.15 Give the chemical symbol or name for the...Ch. 1 - 1.16 Give the chemical symbol or name for each of...Ch. 1 - A solid white substance A is heated strongly in...Ch. 1 - 1.18 You are hiking in the mountains and find a...Ch. 1 - 1.19 In the process of attempting to characterize...Ch. 1 - 1.20
Read the following description of the element...Ch. 1 - Prob. 21ECh. 1 - A match is lit and held under a cold piece of...Ch. 1 - Which separation method is better suited for...Ch. 1 - Two beakers contain clear, colorless liquids. When...Ch. 1 - Prob. 25ECh. 1 - Prob. 26ECh. 1 - Prob. 27ECh. 1 - Prob. 28ECh. 1 - Prob. 29ECh. 1 - Prob. 30ECh. 1 - 121 What exponential notation do the following...Ch. 1 -
1.32 Use appropriate metric prefixes to write the...Ch. 1 - Make the following conversions. 72 °F to °C, 216.7...Ch. 1 - a. The temperature on a warm summer day is 87 °F....Ch. 1 - Prob. 35ECh. 1 - A cube of osmium metal 1.500 cm on a side has a...Ch. 1 - To identify a liquid substance, a student...Ch. 1 - a. After the label fell off a bottle containing a...Ch. 1 - Prob. 39ECh. 1 -
1.40 Silicon for computer chips is grown in large...Ch. 1 - Prob. 41ECh. 1 - 1.42 A watt is a measure of power (the rate of...Ch. 1 - Indicate which of the following are exact numbers;...Ch. 1 - Indicate which of the following are exact numbers:...Ch. 1 - 1.45 What is the number of significant figures in...Ch. 1 - Indicate the number of significant figures in each...Ch. 1 - 1.47 Round each of the following numbers to four...Ch. 1 - 1.48
The diameter of Earth at the equator is 7926...Ch. 1 - Carry out the following operations and express the...Ch. 1 - Carry out the following operations and express the...Ch. 1 - You weigh an object on a balance and read the mass...Ch. 1 - You have a graduated cylinder that contains a...Ch. 1 - 153 Using your knowledge of metric units, English...Ch. 1 - 1.54 Using your knowledge of metric units, English...Ch. 1 - A bumblebee flies with a ground speed of 15.2 m/s....Ch. 1 - 1 56
a The speed of light in a vacuum is 2.998 x...Ch. 1 - Perform the following conversions: 5.00 days to s,...Ch. 1 - Carry out the following conversions: 0.105 in. to...Ch. 1 - How many liters of wine can be held in a wine...Ch. 1 - If an electric car is capable of going 225 km on a...Ch. 1 - The density of air at ordinary atmospheric...Ch. 1 - 1.62 The concentration of carbon monoxide in an...Ch. 1 - Prob. 63ECh. 1 - 1.64 A copper refinery produces a copper ingot...Ch. 1 - 165 Classify ea. al the folbwing as a pure...Ch. 1 - 1.66
Which is more likely to eventually be shown...Ch. 1 -
1.67 A sample of ascorbic acid (vitamin C) is...Ch. 1 - Prob. 68AECh. 1 - SO Two students deterrmne the percen.ge of lead in...Ch. 1 - 1.70
Is Om use of significant figures in ea. of...Ch. 1 - What type of quantity (for example, length,...Ch. 1 - 1.72 Give the derived SI units for each of the...Ch. 1 - 1.73 The distance from Earth to the Moon is...Ch. 1 - 1.74 Which of the following would you characterize...Ch. 1 -
1.75 The U.S. quarter has a mass of 5.67 g and is...Ch. 1 -
1.76 In the United States, water used for...Ch. 1 -
1.77 By using estimation techniques, determine...Ch. 1 - Suppose you decide to define your own temperature...Ch. 1 -
1.79 The liquid substances mercury (density =...Ch. 1 -
1.80 Two spheres of equal volume are placed on...Ch. 1 - Water has a density of 0.997 g/cm3 at 25C ; ice...Ch. 1 - A 32.65-g sample of a solid is placed in a flask....Ch. 1 - A thief plans to steal a gold sphere with a radius...Ch. 1 - Automobile batteries contain sulfuric acid, which...Ch. 1 - A 40-lb container of peat moss measures 14 x 20 x...Ch. 1 - A package of aluminum foil contains 50 ft2of foil,...Ch. 1 - Prob. 87AECh. 1 -
1.88 In 2005, J. Robin Warren and Barry J....Ch. 1 -
1 89 A 25 0-cm.long cylindrical glass tube,...Ch. 1 -
1.90 Gold is alloyed (mixed) with other metals to...Ch. 1 -
1.91 Paper chromatography is a simple but...Ch. 1 -
1.92 Judge the following statements as true or...Ch. 1 -
1.93 You are assigned the task of separating a...Ch. 1 - Prob. 94AE
Additional Science Textbook Solutions
Find more solutions based on key concepts
3. Trails that are derived from a common ancestor, like the bones of human arms and bird wings, are said to be_...
Biological Science (6th Edition)
7. Jack and Jill ran up the hill at 3.0 m/s. The horizontal component of Jill’s velocity vector was 2.5 m/s.
a....
College Physics: A Strategic Approach (3rd Edition)
56. Global Positioning System. Learn more about the global positioning system and its uses. Write a short repo...
The Cosmic Perspective (8th Edition)
What is the difference in elevation between Points A and B? Difference in elevation: _____________feet
Applications and Investigations in Earth Science (9th Edition)
What type of cut would separate the brain into anterior and posterior parts?
Anatomy & Physiology (6th Edition)
55. For the reaction shown, find the limiting reactant for each of the initial quantities of reactants.
a.
b....
Introductory Chemistry (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A DEPT NMR spectrum is shown for a molecule with the molecular formula of C5H12O. Draw the structure that best fits this data. 200 180 160 140 120 100 一盆 00 40 8- 20 ppm 0 Qarrow_forwardDon't used hand raitingarrow_forwardShown below is the major resonance structure for a molecule. Draw the second best resonance structure of the molecule. Include all non-zero formal charges. H. H. +N=C H H H Cl: Click and drag to start drawing a structure. : ? g B S olo Ar B Karrow_forward
- Don't used hand raitingarrow_forwardS Shown below is the major resonance structure for a molecule. Draw the second best resonance structure of the molecule. Include all non-zero formal charges. H H = HIN: H C. :0 H /\ H H Click and drag to start drawing a structure. ×arrow_forwardPlease help me figure out these calculation and what should be plotted. These are notes for my chemistry class.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Measurement and Significant Figures; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Gn97hpEkTiM;License: Standard YouTube License, CC-BY
Trigonometry: Radians & Degrees (Section 3.2); Author: Math TV with Professor V;https://www.youtube.com/watch?v=U5a9e1J_V1Y;License: Standard YouTube License, CC-BY