Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and...
Related questions
Question
100%
4.
![**Exercise 4: Balancing Redox Reactions in Basic Solutions**
**Objective:** Balance the following redox reaction in a basic solution.
**Chemical Equation:**
\[ \text{H}_2\text{O}_2 + \text{MnO}_4^- \rightarrow \text{Mn}^{2+} + \text{O}_2 \]
**Instructions:** Use the half-reaction method to balance the equation, considering the transfer of electrons and ensuring mass and charge balance. Make sure to add the necessary hydroxide ions (OH⁻) to balance the hydrogen ions (H⁺) in a basic solution.](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F41774409-65e0-43ad-a43d-34f80707331e%2F58df6678-6ed2-4e7f-b650-5be5355be205%2Fmn7yszx_processed.jpeg&w=3840&q=75)
Transcribed Image Text:**Exercise 4: Balancing Redox Reactions in Basic Solutions**
**Objective:** Balance the following redox reaction in a basic solution.
**Chemical Equation:**
\[ \text{H}_2\text{O}_2 + \text{MnO}_4^- \rightarrow \text{Mn}^{2+} + \text{O}_2 \]
**Instructions:** Use the half-reaction method to balance the equation, considering the transfer of electrons and ensuring mass and charge balance. Make sure to add the necessary hydroxide ions (OH⁻) to balance the hydrogen ions (H⁺) in a basic solution.
Expert Solution
Step 1
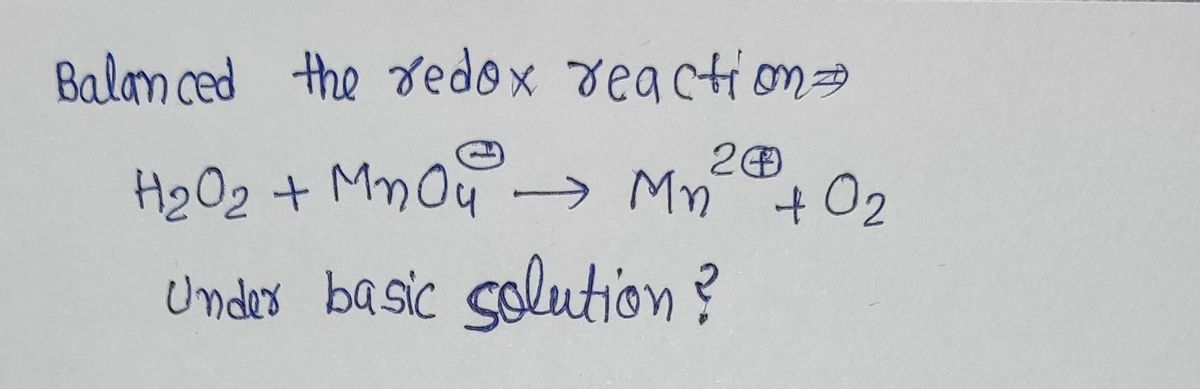
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9780078021558
Author:
Janice Gorzynski Smith Dr.
Publisher:
McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Elementary Principles of Chemical Processes, Bind…
Chemistry
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY