A refrigeration cycle operating as shown in figure below has Qout = 1600 Btu and Wcycle = 900 Btu. Determine the coefficient of performance for the cycle. Hot body Qout System lin Cold body cycle=Qout-lin
Q: For the isobaric process as shown in the figure, find an expression for the first law that involves…
A: The process which is taking place under constant pressure is known as an isobaric process. In an…
Q: For a power cycle operating as shown in the figure, Wcycle = 800 Btu and Qout = 1750 Btu. Hot body…
A:
Q: Isotherm T=423 K 2. The above figure shows the PV diagram for a reversible heat engine that uses…
A: Given: Number of moles n =2 Diatomic gas At point a , temperature = 20 degrees and pressure = 105…
Q: Find the work done in the P-V process from shown in the diagram above
A: It is known that if a P-V diagram is given , then the net work done is equal to the area bounded by…
Q: in the figure. The arrows of the curve indicate the direction of the process, and the points of…
A: As we know,Area under P-V curve is…
Q: A heat pump takes in 440J of heat from a low temp reservoir in each cycle and uses 200J of work to…
A:
Q: A heat pump has a coefficient of performance of 3.1. Hint If it requires 290 J of work per…
A:
Q: A Carnot engine produces 25 kW while operating between temperature limits of 10000K and 3000K.…
A: Given,output W=25kWtemperature TH=10000Ktemperature TC=3000K
Q: A heat engine operates in a Carnot cycle between 70.0°C and 345°C. It absorbs 20,800 J of energy per…
A:
Q: The four processes for a Carnot Cycle are: isobaric expansion, isentropic expansion, isobaric…
A: Answer - The correct option is (4) isothermal expansion, isentropic expansion, isothermal…
Q: The inside and outside temperature of a refrigerator are 273K and 303K respectively. Assuming that…
A: Option
Q: An engine with 12% efficiency does 125J of work in each cycle. How much heat is absorbed in each…
A: Efficiency of the engine = 12 % ∴ e =0.12 =WQhwhere W= work done in each cycle =125 J Qh…
Q: A refrigerator has a coefficient of performance of 2.30. Each cycle it absorbs 3.23x10ª J of heat…
A:
Q: Question 13 The coefficient of performance of a refrigerator is 2 . It absorbs 50 J of heat from the…
A: Concept: Here, the question is from Thermodynamics. Given in the Question The Coefficient of…
Q: The thermal efficiency of a reversible power cycle operating between hot and cold reservoirs is 70%.…
A:
Q: Please use the appropriate format in answering word problem: Problem 03 A heat engine is operated…
A: Given: Heat source temperature: TH=1470°C The cold reservoir temperature: TC=260°C
Q: Needs Complete solution with 100 % accuracy don't use chat gpt or ai please plz plz plz plz.
A: Step 1:Step 2:
Q: A heat engine with an efficiency of 30% takes in 800 J of heat from the high-temperature reservoir…
A: Given: The efficiency of the heat engine is 30%. The heat input to the engine is 800 J.
Q: The choices below are all efficiency ratings for heat engines. Which is a Carnot engine?
A: Efficiency of any Carnot engine is always less than 1. η=1-T2T1<…
Q: An engine releases 0.450 kJ of heat for every 0.510 kJ of work it does. What is the efficiency of…
A: heat released Q2=0.450kJwork done by engine W=0.510kJ
Q: 2. A heat engine has an efficiency of 52% when it runs with a hot-reservoir temperature of 400°C and…
A:
Q: If a 35.9%-efficient Carnot heat engine (left figure) is run in reverse so as to form a refrigerator…
A: Given that the efficiency of heat engine is efficiency=35.9%= 35.9/100=0.359 Coefficient of…
Q: A Carnot heat engine operates between reservoirs at TH = 1630 K and Tc = 160 K. %3D %3D In each…
A: Solution: The efficiency of the heat engine is given by,…
Q: Describe Carnot cycle and derive an expression for its efficiency.
A: Carnot Cycle is considered as an ideal cycle.It is a reversible cycle ,it has four processes and all…
Q: A heat pump has a coefficient of performance of 2.2 in heating mode. If it requires 140 J of work…
A:
Q: The coefficient of performance of the heat pump used as a cooler (illustrated below) is x-3. What is…
A: Given: The coefficient of performance is 3.
Q: V
A: The shown process is an isochoric process. Isochoric process : it is a process in which the volume…
Q: The gas in a certain heat engine starts each cycle at STP. In the first step of the cycle, the gas…
A:
Q: In the following image, if we intend to increase the theoretical efficiency of this engine, we can…
A: As we know the efficiency of Carnot heat engine Efficiency η = work doneHeat taken η = Q1 - Q2Q1 =…
Q: Q3) (a) A Carnot heat engine is operating with the hot reservoir at 100 °C and the cold reservoir at…
A:
Q: Consider the cycle using monoatomic ideal gas described by the PV diagram below. The process 1 2 is…
A: For a monoatomic ideal gas, the value of the adiabatic ratio is γ=1.67. Let us find the value of P2…
Q: An engine has a hot-reservoir temperature of 900 K and a cold-reservoir temperature of 600 K. The…
A: We are given a heat engine. This heat engine is not Carnot engine. We are given the efficiency of…
Q: For the same maximum pressure and temperature, A. Otto cycle is more efficient than Diesel cycle B.…
A: Option b is correct
Q: 1. Two heat engines are shown in the figure below. Engine 1 follows the cycle ABCDA and engine 2…
A: Efficiency of engine n = Work /heat absorbed
Q: A reversible power cycle operating as in the figure receives energy QH by heat transfer from a hot…
A: Given Data, Rejected EnergyQC=10 Btu Temperature of the cold reservoirTC=400F Work for the cycleW=30…
Q: A certain refrigerator requires 31.0J of work to remove 150J of heat from its interior. 1. What is…
A: Work done is The removed heat energy is Temperature of surrounding is Find:(a) The coefficient of…
Q: A heat engine provides a net power output of 14 kW and has an efficiency of 23 %. The engine rejects…
A:

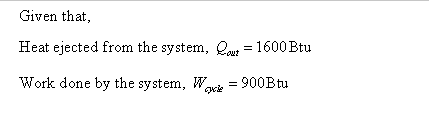
Step by step
Solved in 2 steps with 2 images
