
Concept explainers
Show how you would modify the synthesis given in the previous problem to synthesize the following drugs:
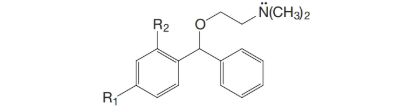
(a) Bromodiphenhydramine (an antihistamine), where
(b) Orphenadrine (an antispasmodic, used in controlling Parkinson’s disease), where
Want to see the full answer?
Check out a sample textbook solution
Chapter SRP Solutions
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
Additional Science Textbook Solutions
Biology: Life on Earth with Physiology (11th Edition)
Principles of Anatomy and Physiology
Anatomy & Physiology (6th Edition)
Human Physiology: An Integrated Approach (8th Edition)
Applications and Investigations in Earth Science (9th Edition)
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
- Q2: Draw all applicable resonance forms for the acetate ion CH3COO. Clearly show all lone pairs, charges, and arrow formalism.arrow_forwardIn the following molecule, indicate the hybridization and shape of the indicated atoms. -z: CH3 CH 3 HO: H3C :Ö: CIarrow_forwardShow mechanism with explanation. don't give Ai generated solutionarrow_forward
- Please Help!!!arrow_forwardQ2: Resonance Forms a) Draw all resonance forms of the molecules. Include curved arrow notation. Label major resonance contributor. SO2 NO3 Page 3 of 4 Chem 0310 Organic Chemistry 1 HW Problem Sets CH3NSO (Thionitromethane, skeleton on the right) H N H3C Sarrow_forwardA 10.00-mL pipet was filled to the mark with distilled water at the lab temperature of 22 oC. The water, delivered to a tared weighing bottle was found to weigh 9.973 g. The density of water at 22 oC is 0.99780 g/mL. Calculate the volume of the pipet in mL. (disregard air displacement for this calculation and record your answer to the proper number of significant digits.)arrow_forward
- Resonance Formsa) Draw all resonance forms of the molecules. Include curved arrow notation. Label majorresonance contributor.arrow_forwardShow work with explanation needed. Don't give Ai generated solutionarrow_forwardf) The unusual molecule [2.2.2] propellane is pictured. 1) Given the bond length and bond angles in the image, what hybridization scheme best describes the carbons marked by the askerisks? 2) What types of orbitals are used in the bond between the two carbons marked by the askerisks? 3) How does this bond compare to an ordinary carbon-carbon bond (which is usually 1.54 Å long)? CH2 1.60Å H2C た C CH2 H2C H₂C * 120° C H2arrow_forward
- Denote the dipole for the indicated bonds in the following molecules. H3C CH3 B F-CCl3 Br-Cl | H3C Si(CH3)3 OH НО. HO H O HO OH vitamin C CH3arrow_forwardQ2: Draw all applicable resonance forms for the acetate ion CH3COO. Clearly show all lone pairs, charges, and arrow formalism.arrow_forwardHow do I calculate the amount of quarks in magnesium?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT


