
Concept explainers
a)
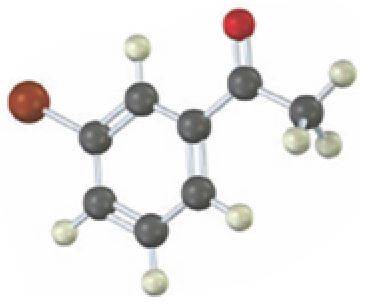
Interpretation:
Starting from which alkyne the compound shown can be prepared is to be stated.
Concept introduction:
Carbonyl compounds can be obtained by the tautomerization of enols produced when
To state:
Starting from which alkyne the compound shown can be prepared.
b)
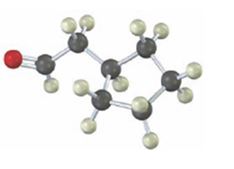
Interpretation:
Starting from which alkyne the compound shown can be prepared is to be stated.
Concept introduction:
Carbonyl compounds can be obtained by the tautomerization of enols produced when alkynes are hydrated. The hydration of terminal alkynes can yield an enol which can tautomerize to an aldehyde or ketone. If oxymercuration- demercuration hydration process is used, the OH adds to the more highly substituted carbon in the triple bond following Markovnokov regiochemistry. If hydroboration –oxidation process is used, the OH adds to the less highly substituted carbon in the triple bond following anti-Markovnokov regiochemistry.
To state:
Starting from which alkyne the compound shown can be prepared.
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
EP ORGANIC CHEMISTRY,24 MONTH-OWLV2
- 3) Draw a detailed mechanism and predict the product of the reaction shown? 1) EtMgBr 2) H3O+arrow_forwardHow to draw the mechanism for this reaction?arrow_forward> H₂C=C-CH2-CH3 B. H₂O Pt C. + H2 + H₂O H D. 16. Give the IUPAC name for each of the following: B. Cl Cl c. Cl Cl 17. Draw the line-angle formula for each of the following compounds: 1. phenol 2. 1,3-dichlorobenzene 3. 4-ethyltoluene < Previous Submit Assignment Next ▸arrow_forward
