
To find:
The enthalpy changes (
Answer to Problem 9.93QA
Solution:
a)
b)
c)
Explanation of Solution
1) Concept:
For
2) Formula:
Positive values of
3) Given:
The required average bond energy values are taken from Table A4.1 of Appendix 4.
| Bond | Bond energy (kJ/mol) |
| H-H | |
| N-H | |
| N-N | |
| N=N | |
| N=O |
4) Calculations:
a) The given balanced reaction is
The reaction involves the formation of two moles of ammonia from its constituent elements in their standard states.
First, we have to draw the Lewis structures of reactants and products into the balanced chemical equation to get the number of bonds involved in each reactant and product.
![]() +
+ ![]() ⤍
⤍ ![]()
We can construct a table using the appropriate bond energies:
| Bonds | Number of bonds(mol) | Bond energy kJ/mol | ||
| Bonds broken | ||||
| Bonds broken | ||||
| Bonds formed |
We can get the value of
b) The given balanced reaction is
The reaction involves the formation of one mole of hydrazine from its constituent elements in their standard states.
First, we have to draw the Lewis structures of reactants and products into the balanced chemical equation to get the number of bonds involved in each reactant and product.
![]() +
+ ![]() ⤍
⤍ 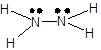
We can construct a table using the appropriate bond energies:
| Bonds | Number of bonds(mol) | Bond energy (kJ/mol) | ||
| Bonds broken | ||||
| Bonds broken | H-H | |||
| Bonds formed | N-N | |||
| Bonds formed | N-H |
We can get the value of
c)The given balanced reaction is:
The reaction involves the formation of two moles of nitrous oxide gas from its constituent elements in their standard states.
First, we have to draw the Lewis structures of reactants and products into the balanced chemical equation to get the number of bonds involved in each reactant and product.
![]() +
+ ![]() ⤍
⤍ ![]()
We can construct a table using the appropriate bond energies:
| Bonds | Number of bonds(mol) | Bond energy (kJ/mol) | ∆H | |
| Bonds broken | 2 | 945 | ||
| Bonds broken | 1 | 498 | ||
| Bonds formed | N=N | 2 | 418 | |
| Bonds formed | N=O | 2 | 607 |
We can get the value of
Conclusion:
The standard enthalpy change of a reaction can be calculated using the standard average bond energies per mole for each reactant and product.
Want to see more full solutions like this?
Chapter 9 Solutions
CHEM:ATOM FOC 2E CL (TEXT)
- b. H3C CH3 H3O+ ✓ H OHarrow_forward2. Provide reagents/conditions to accomplish the following syntheses. More than one step is required in some cases. a. CH3arrow_forwardIdentify and provide an explanation that distinguishes a qualitative and quantitative chemical analysis. Provide examples.arrow_forward
- Identify and provide an explanation of the operational principles behind a Atomic Absorption Spectrometer (AAS). List the steps involved.arrow_forwardInstructions: Complete the questions in the space provided. Show all your work 1. You are trying to determine the rate law expression for a reaction that you are completing at 25°C. You measure the initial reaction rate and the starting concentrations of the reactions for 4 trials. BrO³¯ (aq) + 5Br¯ (aq) + 6H* (aq) → 3Br₂ (l) + 3H2O (l) Initial rate Trial [BrO3] [H*] [Br] (mol/L) (mol/L) | (mol/L) (mol/L.s) 1 0.10 0.10 0.10 8.0 2 0.20 0.10 0.10 16 3 0.10 0.20 0.10 16 4 0.10 0.10 0.20 32 a. Based on the above data what is the rate law expression? b. Solve for the value of k (make sure to include proper units) 2. The proposed reaction mechanism is as follows: i. ii. BrО¸¯ (aq) + H+ (aq) → HBrO3 (aq) HBrO³ (aq) + H* (aq) → H₂BrO3* (aq) iii. H₂BrO³* (aq) + Br¯ (aq) → Br₂O₂ (aq) + H2O (l) [Fast] [Medium] [Slow] iv. Br₂O₂ (aq) + 4H*(aq) + 4Br(aq) → 3Br₂ (l) + H2O (l) [Fast] Evaluate the validity of this proposed reaction. Justify your answer.arrow_forwardе. Д CH3 D*, D20arrow_forward
- H3C. H3C CH 3 CH 3 CH3 1. LDA 2. PhSeCl 3. H2O2arrow_forwardPlease predict the products for each of the following reactions: 1.03 2. H₂O NaNH, 1. n-BuLi 2. Mel A H₂ 10 9 0 H2SO4, H₂O HgSO4 Pd or Pt (catalyst) B 9 2 n-BuLi ♡ D2 (deuterium) Lindlar's Catalyst 1. NaNH2 2. EtBr Na, ND3 (deuterium) 2. H₂O2, NaOH 1. (Sia)2BH с Darrow_forwardin the scope of ontario SCH4U grade 12 course, please show ALL workarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





