
EBK CHEMISTRY
9th Edition
ISBN: 9780100453807
Author: ZUMDAHL
Publisher: YUZU
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 87CP
Consider the following computer-generated model of caffeine:
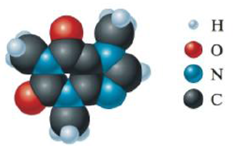
Complete a Lewis structure for caffeine in which all atoms have a formal charge of zero (as is typical with most organic compounds). How many C and N atoms are sp2 hybridized? How many C and N atoms are sp3 hybridized? sp hybridized? How many σ and π bonds are there?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Starting from bromoethane, how could you prepare the following compounds:
a. Ethanol.
b. Acetaldehyde
f. Acetone.
e. 2-Propanol
i. Acetoacetic ester.
d. 2-Bromoacetic acid.
c. Acetic acid
g. Acetamide.
j. Ethylmalonate
k. Gama ketoacid.
h. Ethyl magnesium bromide.
-
The pressure above a pure sample of solid Substance X at 60. °C is raised. At what pressure will the sample melt? Use the phase diagram of X below to find
your answer.
pressure (atm)
02
0.4
solid
Hliquid
gas
0
0
200
400
600
temperature (K)
Note: your answer must be within 0.025 atm of the exact answer to be graded correct.
☐ atm
15. What is the order of decreasing reactivity towards nucleophilic acyl substitution for the
carboxylic acid derivatives? (most reactive first)
0
O
H3C COC CH3 H₂C C N(CH3)2 H3C C OCH3
A.
a.
I, 11, 111,
b.
I, III, IV, II
C.
II, IV, III, I
°
(CH3)2CH C OCH3
IV
d. II, I, III, IV
B. R COCR
0
0
0
13=
RC NH2
RC OR
RC CI
===
IV
a.
I, III, II, IV
b. II, III, I, IV
C.
III, II, I, IV
d. IV, I, III, II
Chapter 9 Solutions
EBK CHEMISTRY
Ch. 9 - Why do we hybtidize atomic orbitals to explain the...Ch. 9 - What hybridization is required for central atoms...Ch. 9 - Describe the bonding in H2S, CH4, H2CO and HCN...Ch. 9 - What hybridization is required for central atoms...Ch. 9 - Electrons in bonding molecular orbitals are most...Ch. 9 - What are molecular orbitals? How do they compare...Ch. 9 - Explain the difference between the and MOs for...Ch. 9 - Compare Figs. 4-47 and 4-49. Why are they...Ch. 9 - Which of the following would you expect to be more...Ch. 9 - Draw the Lewis structure for HCN. Indicate the...
Ch. 9 - Which is the more correct statement: The methane...Ch. 9 - Compare and contrast the MO model with the local...Ch. 9 - What are the relationships among bond order, bond...Ch. 9 - In the hybrid orbital model, compare and contrast ...Ch. 9 - In the molecular orbital mode l, compare and...Ch. 9 - Why are d orbitals sometimes used to form hybrid...Ch. 9 - The atoms in a single bond can rotate about the...Ch. 9 - Compare and contrast bonding molecular orbitals...Ch. 9 - What modification to the molecular orbital model...Ch. 9 - Why does the molecular orbital model do a better...Ch. 9 - The three NO bonds in NO3 are all equivalent in...Ch. 9 - Use the localized electron model to describe the...Ch. 9 - Use the localized electron model to describe the...Ch. 9 - Use the localized electron model to describe the...Ch. 9 - Use the localized electron model to describe the...Ch. 9 - The space-filling models of ethane and ethanol are...Ch. 9 - The space-filling models of hydrogen cyanide and...Ch. 9 - Give the expected hybridization of the central...Ch. 9 - Give the expected hybridization of the central...Ch. 9 - Give the expected hybridization of the central...Ch. 9 - Give the expected hybridization of the central...Ch. 9 - For each of the following molecules, write the...Ch. 9 - For each of the following molecules or ions that...Ch. 9 - Prob. 31ECh. 9 - The allene molecule has the following Lewis...Ch. 9 - Indigo is the dye used in coloring blue jeans. The...Ch. 9 - Urea, a compound formed in the liver, is one of...Ch. 9 - Biacetyl and acetoin are added to margarine to...Ch. 9 - Many important compounds in the chemical industry...Ch. 9 - Two molecules used in the polymer industry are...Ch. 9 - Hot and spicy foods contain molecules that...Ch. 9 - One of the first drugs to be approved for use in...Ch. 9 - The antibiotic thiarubin-A was discovered by...Ch. 9 - Consider the following molecular orbitals formed...Ch. 9 - Sketch the molecular orbital and label its type (...Ch. 9 - Which of the following are predicted by the...Ch. 9 - Which of the following are predicted by the...Ch. 9 - Using the molecular orbital model, write electron...Ch. 9 - Consider the following electron configuration:...Ch. 9 - Using molecular orbital theory, explain why the...Ch. 9 - Using the molecular orbital model to describe the...Ch. 9 - The transport of O2 in the blood is carried out by...Ch. 9 - A Lewis structure obeying the octet rule can be...Ch. 9 - Using the molecular orbital model, write electron...Ch. 9 - Using the molecular orbital model, write electron...Ch. 9 - In which of the following diatomic molecules would...Ch. 9 - In terms of the molecular orbital model, which...Ch. 9 - Show how two 2p atomic orbitals can combine to...Ch. 9 - Show how a hydrogen 1s atomic orbital and a...Ch. 9 - Use Figs. 4-54 and 4-55 to answer the following...Ch. 9 - Acetylene (C2H2) can be produced from the reaction...Ch. 9 - Describe the bonding in NO+, NO, and NO, using...Ch. 9 - Describe the bonding in the O3 molecule and the...Ch. 9 - Describe the bonding in the CO32 ion using the...Ch. 9 - Draw the Lewis structures, predict the molecular...Ch. 9 - FClO2 and F3ClO can both gain a fluoride ion to...Ch. 9 - Two structures can be drawn for cyanuric acid: a....Ch. 9 - Give the expected hybridization for the molecular...Ch. 9 - Vitamin B6 is an organic compound whose deficiency...Ch. 9 - Aspartame is an artificial sweetener marketed...Ch. 9 - Prob. 69AECh. 9 - The three most stable oxides of carbon are carbon...Ch. 9 - Complete the following resonance structures for...Ch. 9 - Prob. 73AECh. 9 - Describe the bonding in the first excited state of...Ch. 9 - Using an MO energy-level diagram, would you expect...Ch. 9 - Show how a dxz. atomic orbital and a pz, atomic...Ch. 9 - What type of molecular orbital would result from...Ch. 9 - Consider three molecules: A, B, and C. Molecule A...Ch. 9 - Draw the Lewis structures for TeCl4, ICl5, PCl5,...Ch. 9 - A variety of chlorine oxide fluorides and related...Ch. 9 - Pelargondin is the molecule responsible for the...Ch. 9 - Complete a Lewis structure for the compound shown...Ch. 9 - Which of the following statements concerning SO2...Ch. 9 - Consider the molecular orbital electron...Ch. 9 - Place the species B2+ , B2, and B2 in order of...Ch. 9 - Consider the following computer-generated model of...Ch. 9 - Cholesterol (C27liu;O) has the following...Ch. 9 - Cyanamide (H2NCN), an important industrial...Ch. 9 - A flask containing gaseous N2 is irradiated with...Ch. 9 - Prob. 92CPCh. 9 - Values of measured bond energies may vary greatly...Ch. 9 - Use the MO model to explain the bonding in BeH2....Ch. 9 - Prob. 95CPCh. 9 - Arrange the following from lowest to highest...Ch. 9 - Use the MO model to determine which of the...Ch. 9 - Given that the ionization energy of F2 is 290...Ch. 9 - Carbon monoxide (CO) forms bonds to a variety of...Ch. 9 - Prob. 100CPCh. 9 - As the bead engineer of your starship in charge of...Ch. 9 - Determine the molecular structure and...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the formula of the product obtained by reacting D-Tallose with bromine water.arrow_forwardChoose the best reagent(s) for carrying out the following conversions from the list below. Place the letter corresponding to the best choice in the blank to the left of the conversion. a. KMnO4, H3O+ b. Tollens' Reagent [oxidizing reagent] C. NaBH4, ethanol d. 1. BH3 2. H3O+ e. 1. CH3MgBr, ether 2. H3O+ f. CrO3, H2SO4, H₂O g. 1. Mg, ether 2. CO2 3. H3O+ h. 1. NaCN 2. H2SO4, H2, heat i. O3, then Zn and HOAC j. CH₂I A. B. C. CH CH=CHCH2COOH Br CEN CH COOH + HOOCCH COOH COOH 010 CH3arrow_forwardDraw the structures for each of the intermediates in the boxes provided for the synthesis below. OCH3 Fe HO HNO (CHOO pynding H₂504 LHNO2 NACH-I Fa H₂O HCL HNO 180arrow_forward
- Provide structure(s) for the starting material(s), reagent(s) or the major organic product(s) of each of the following reactions or sequences of reactions. Show all relevant stereochemistry [three only] A. o 11 (CH3)CH — C— C ether (CH3)2CH-C-O-C-CH3 B. CH3 CHy CI Staf OH C. HC OCHS + H₂Oarrow_forwardConsider the reaction sequence below to answer the following questions: EtO Compound X 1. NaOEt, EtOH OEt Br CO₂Et NaOEt, EtOH Compound Z CO₂Et Compound Y A. Compound X, diethyl propanedioate, is more commonly known as a. ethyl acetoacetate b. acetoacetic ester C. oxalic ester d. malonic ester 3. Write the complete stepwise mechanism for the conversion of Compound X into Compound Y. Show all electron flow with arrows and draw all intermediate structures.arrow_forwardClassify each of the following nitrogen atoms in the following compounds as primary, secondary, tertiary, or quaternary [three only] CH3 HO-CHCHNHCH3 A. B. C. H&C CH3 D. HO phedrine CH2CHCH3 amphetamine NH₂ mepiquat chloride faxofenadine OH H&C CH CO₂Harrow_forward
- Draw the structure of the aldol self-condensation product for each of the following compounds. If a compound does not undergo aldol self-condensation, explain why it does not. A. B. CHICHCH₂OH CH3CHCH2CH CH3 CH3 C. CH 30 H3C-C-C-H CH3 questionsarrow_forward. A.Propose a synthesis for propylbenzene which avoids the problems of direct Friedel-Crafts alkylation. B. Consider the reaction below to answer the following questions. A B C NO2 Febr Brz D The Lewis acid catalyst in the reaction is: a. The nucleophile in the reaction is: b. C. d. This reaction proceeds Draw the structure of product D. (faster or slower) than benzene.arrow_forwardConsider the reaction below to answer the following questions. HOCH CHOH На A B C D H₂Oarrow_forward
- Consider the structures below to answer the following questions. A. Indicate the most acidic hydrogens in each of the molecules. OH CH-H CH₂C-H H&C མིངྒཱའི B. Rank the molecules above in order of increasing acidity (least acidic to most acidic). a. III, II, I b. II, III, I C. I, II, III d. II, I, IIIarrow_forwardConsider the reaction below to answer the following questions. H H+ A B CH₂OH 5% NaOCH, CH₂OH A. Which carbonyl compound functions as the electrophile in this reaction? B. Draw the structure of the enolate ion that is generated during the course of this reaction. C. This reaction is an example of: a. a mixed Claisen condensation. b. C. d. a Dieckman condensation. a Michael reaction. a mixed aldol reaction.arrow_forwardGive the major organic product(s) of each of the following reactions or sequences of reactions. Show all relevant stereochemistry. [two only] CH3O (11 HC-C-C-CH3 A. CH3 12. NaOHarrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY