
Write the structure of the major organic product isolated from the reaction of
Hydrogen (
Hydrogen (
Sodium amide in liquid ammonia
Product in part (c) treated with 1-bromobutane
Product in part (c) treated with tert-butyl bromide
Hydrogen chloride (
Hydrogen chloride (
Chlorine (
Chlorine (
Aqueous sulfuric acid, mercury
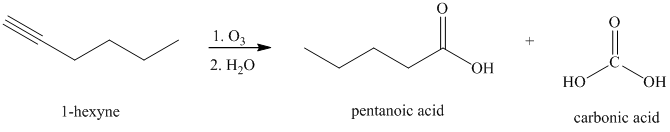
Ozone followed by hydrolysis
Interpretation:
The structure of the major product isolated from the reaction of
Concept introduction:
In the presence a metal catalyst such as platinum, palladium, nickel, or rhodium, two molar equivalents of hydrogen get added across the triple bond of an alkyne to yield an alkane. In hydrogenation reaction, two hydrogen atoms get added to each carbon atom across the triple bond.
Hydrogenation reaction of alkynes to alkenes using the Lindlar catalyst is stereoselective. In this reaction only the cis alkene is produced.
Terminal alkynes react with
Addition of hydrogen halides to alkynes gives alkenyl halides. The regioselectivity of this reaction follows Markovnikov’s rule. The hydrogen atom from hydrogen halide adds to the carbon in the alkyne that has the more number of hydrogens, and the halide adds to the carbon with the lower number of hydrogens.
In presence of excess of hydrogen halide, the sequential addition of two molecules of hydrogen halide to the carbon-carbon triple bond yields geminal dihalides.
The reaction of chlorine and bromine with alkynes yields tetrahaloderivatives. In this reaction, two molecules of the halogen get added to the triple bond. During the reaction, a dihaloalkene is get formed. When alkyne and the halogen are present in equimolar quantity, the dihaloalkene intermediate get formed and isolated. The stereochemistry of addition reaction is anti.
Alkynes, when subjected to ozonolysis followed by hydrolysis, produce carboxylic acids.
Answer to Problem 24P
Solution:
a)
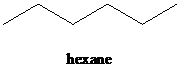
b)
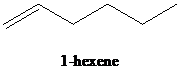
c)
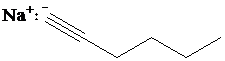
d)
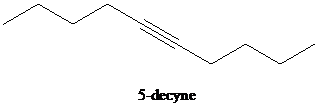
e)
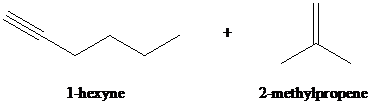
f)
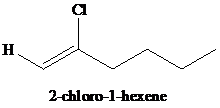
g)
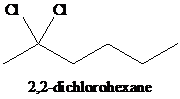
h)

i)
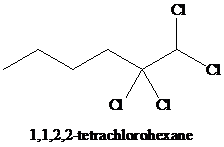
j)
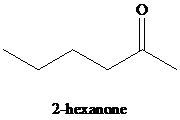
k)
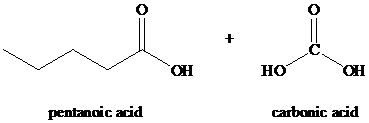
Explanation of Solution
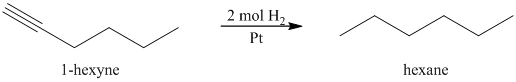
a) The reaction of
In the presence a metal catalyst such as platinum, palladium, nickel, or rhodium, two molar equivalents of hydrogen get added across the triple bond of an alkyne to yield an alkane. In case of
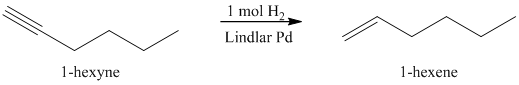
b) The reaction of
Reduction over Lindlar palladium catalyst with one mole of hydrogen partially reduces an alkyne to the corresponding cis alkene.
c) The reaction of
Terminal alkynes react with
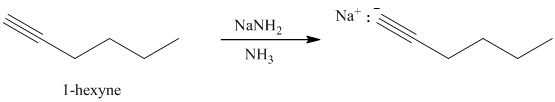
d) The reaction of
In the reaction of alkyl halide and sodium acetylide, the acetylide ion acts as a nucleophile. It displaces halide from carbon and forming a new carbon-carbon bond. This reaction follows
The product formed in part (c) is the anion formed by
The product formed is
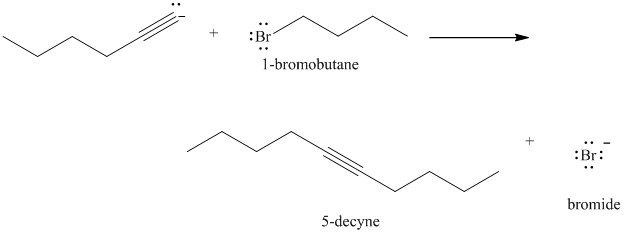
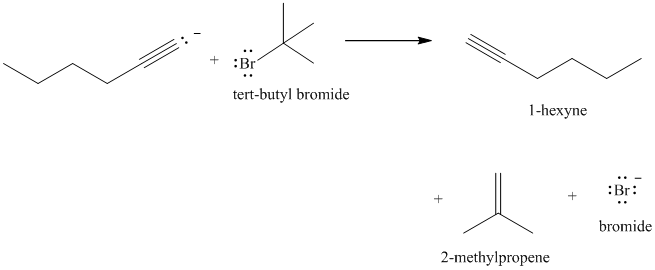
e) The reaction of
Acetylide anions are much more basic than hydroxide. Acetylide anions react with secondary and tertiary alkyl halides by elimination. Tert-butyl bromide is a tertiary alkyl halide and does not react by the
The product formed in part (c) is the anion formed by
f) The reaction of
Hydrogen halides add to alkynes to form alkenyl halides. When
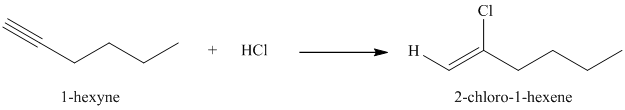
g) The reaction of
In presence of excess of hydrogen halide, the sequential addition of two molecules of hydrogen halide to the carbon-carbon triple bond yields geminal dihalides. The second mole addition of hydrogen halide to the initially formed alkenyl halide is done in accordance with Markovnikov’s rule. Overall, both protons get attached to the same carbon and both halogens to the adjacent carbon atom. In
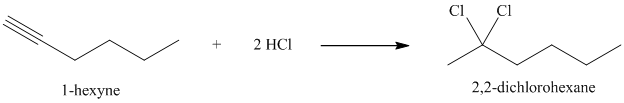
h) The reaction of
When one mole of halogen molecule is added to an alkyne, the product formed is a dihaloalkene. One halogen atom gets attached to each of the triple bonded carbon atoms, and it is reduced to a double bond.
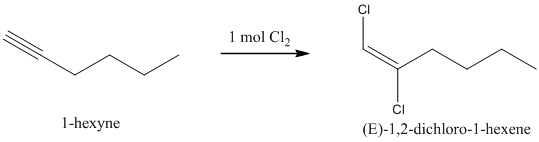
i) The reaction of
Alkynes react with two moles of chlorine to yield tetrachloroalkanes. Two molecules of the chlorine add to the triple bond. The stereochemistry of addition is anti.
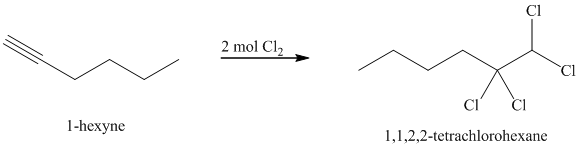
j) The reaction of
Hydration of alkynes employs aqueous sulfuric acid as the reaction medium and mercury(II) sulfate as a catalyst. The product formed is an enol which tautomerizes to the corresponding ketone. Markovnikov’s rule is followed in hydration of alkynes. Terminal alkynes yield methyl substituted ketones.
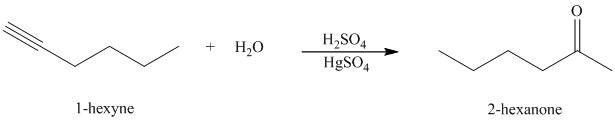
k) The reaction of
Alkynes, when subjected to ozonolysis followed by hydrolysis, produce carboxylic acids. If carbonic acid is one of the products, it dissociates into carbon dioxide and water.
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry - Standalone book
- please help with synthesisarrow_forward10. Stereochemistry. Assign R/S stereochemistry for the chiral center indicated on the following compound. In order to recieve full credit, you MUST SHOW YOUR WORK! H₂N CI OH CI カー 11. () Stereochemistry. Draw all possible stereoisomers of the following compound. Assign R/S configurations for all stereoisomers and indicate the relationship between each as enantiomer, diastereomer, or meso. NH2 H HNH, -18arrow_forwardb) 8. Indicate whether the following carbocation rearrangements are likely to occur Please explain your rational using 10 words or less not likely to occur • The double bond is still in the Same position + Likely to oc occur WHY? -3 H3C Brave Chair Conformers. Draw the chair conformer of the following substituted cyclohexane. Peform a RING FLIP and indicate the most stable conformation and briefly explain why using 20 words or less. CI 2 -cobs ?? MUST INDICATE H -2 -2 Br EQ Cl OR AT Br H& most stable WHY? - 4arrow_forward
- CH 12 Conformational Analysis. Draw all 6 conformers (one above each letter) of the compound below looking down the indicated bond. Write the letter of the conformer with the HIGHEST and LOWEST in energies on the lines provided. NOTE: Conformer A MUST be the specific conformer of the structure as drawn below -4 NOT HOH OH 3 Conformer A: Br OH A Samo Br H 04 Br H H3 CH₂ H anti stagere Br CH clipsed H Brott H IV H MISSING 2 -2 B C D E F X 6 Conformer with HIGHEST ENERGY: 13. (1 structure LOWEST ENERGY: Nomenclature. a) Give the systematic (IUPAC) name structure. b) Draw the corresponding to this name. HINT: Do not forget to indicate stereochemistry when applicable. a) ८८ 2 "Br {t༐B,gt)-bemn€-nehpརི་ཚ༐lnoa Parent name (noname) 4 Bromo Sub = 2-methylethyl-4 Bromo nonane b) (3R,4S)-3-chloro-4-ethyl-2,7-dimethyloctane # -2 -2arrow_forwardin the scope of the SCH4U course! please show all steps as im still learning how to format my answers in the format given, thank you!arrow_forwardhelp me solve this HWarrow_forward
- Molecules of the form AH2 can exist in two potential geometries: linear or bent. Construct molecular orbital diagrams for linear and bent CH2. Identify the relevant point group, include all of the appropriate symmetry labels and pictures, and fill in the electrons. Which geometry would you predict to be more stable, and why? (Please draw out the diagram and explain)arrow_forwardIndicate the variation in conductivity with concentration in solutions of strong electrolytes and weak electrolytes.arrow_forwardThe molar conductivity of a very dilute solution of NaCl has been determined. If it is diluted to one-fourth of the initial concentration, qualitatively explain how the molar conductivity of the new solution will compare with the first.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

