
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
4th Edition
ISBN: 9781260269284
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8.8, Problem 8.17PP
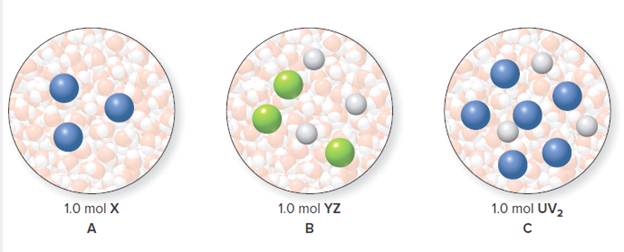
Representations A, B, and C each show an aqueous solution formed from 1.0 mol ofsolute dissolved in I kg of water. (a) Which solution has the highest melting point? (b) Which solution has the lowest melting point?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Don't used Ai solution
Let's see if you caught the essentials of the animation.
What is the valence value of carbon?
a) 4
b) 2
c) 8
d) 6
A laser emits a line at 632.8 nm. If the cavity is 12 cm long, how many modes oscillate in the cavity? How long does it take for the radiation to travel the entire cavity? What is the frequency difference between 2 consecutive modes?(refractive index of the medium n = 1).
Chapter 8 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
Ch. 8.1 - Classify each substance as a heterogeneous...Ch. 8.1 - Use the appearance of each product to classify it...Ch. 8.2 - Consider the following diagrams for an aqueous...Ch. 8.2 - Classify each solution as an electrolyte or...Ch. 8.2 - Using the given number of moles, determine how...Ch. 8.2 - Prob. 8.2PPCh. 8.2 - A solution contains the following ions:...Ch. 8.2 - If a solution contains 125 mEq of Na+ per liter,...Ch. 8.3 - Which compounds are water soluble? a. NaNO3 b. CH4...Ch. 8.3 - Prob. 8.7P
Ch. 8.3 - Use the solubility rules to predict whether the...Ch. 8.3 - Use the solubility rules for ionic compounds to...Ch. 8.4 - Why does a soft drink become "flat" faster when it...Ch. 8.4 - Predict the effect each change has on the...Ch. 8.5 - A commercial mouthwash contains 4.3 g of ethanol...Ch. 8.5 - What is the weight/volume percent concentration of...Ch. 8.5 - Prob. 8.6PPCh. 8.5 - Prob. 8.7PPCh. 8.5 - A drink sold in a health food store contains 0.50%...Ch. 8.5 - Prob. 8.12PCh. 8.5 - What is the concentration in parts per million of...Ch. 8.6 - Prob. 8.10PPCh. 8.6 - Prob. 8.13PCh. 8.6 - Prob. 8.11PPCh. 8.6 - Prob. 8.12PPCh. 8.6 - How many grams of NaCl are contained in each of...Ch. 8.6 - How many milliliters of a 0.25 M sucrose solution...Ch. 8.7 - What is the concentration of a solution formed by...Ch. 8.7 - If the solution of A+B- in X is diluted, which...Ch. 8.7 - Prob. 8.15PPCh. 8.7 - Prob. 8.16PCh. 8.8 - What is the boiling point of a solution prepared...Ch. 8.8 - Representations A, B, and C each show an aqueous...Ch. 8.8 - Prob. 8.18PPCh. 8.8 - What is the melting point of a solution that is...Ch. 8.9 - Which solution in each pair exerts the greater...Ch. 8.9 - Prob. 8.19PCh. 8.9 - Consider the two aqueous solutions separated by a...Ch. 8.9 - What happens to a red blood cell when it is placed...Ch. 8 - Prob. 21PCh. 8 - Prob. 22PCh. 8 - Prob. 23PCh. 8 - Which representation of molecular art better shows...Ch. 8 - Classify each of the following as a solution,...Ch. 8 - Classify each of the following as a solution,...Ch. 8 - Prob. 27PCh. 8 - Label each diagram as a strong electrolyte, weak...Ch. 8 - Prob. 29PCh. 8 - Prob. 30PCh. 8 - Prob. 31PCh. 8 - Prob. 32PCh. 8 - Consider a mixture of two substances shown in blue...Ch. 8 - Which diagram (C or D) best represents what occurs...Ch. 8 - If the solubilityofKClin 100 mL of H2O is 34 g at...Ch. 8 - If the solubilityofsucrosein 100 mL of H2O is 204...Ch. 8 - Prob. 37PCh. 8 - Prob. 38PCh. 8 - Using the ball-and-stick model for methanol...Ch. 8 - Prob. 40PCh. 8 - Prob. 41PCh. 8 - Prob. 42PCh. 8 - Prob. 43PCh. 8 - Prob. 44PCh. 8 - Prob. 45PCh. 8 - Prob. 46PCh. 8 - Prob. 47PCh. 8 - How is the solubility of helium gas in water...Ch. 8 - Use the solubility rules listed in Section 8.3B to...Ch. 8 - Use the solubility rules listed in Section 8.3B to...Ch. 8 - Prob. 51PCh. 8 - Prob. 52PCh. 8 - Prob. 53PCh. 8 - Prob. 54PCh. 8 - Prob. 55PCh. 8 - Prob. 56PCh. 8 - Prob. 57PCh. 8 - Prob. 58PCh. 8 - How would you use a 250-mL volumetric flask to...Ch. 8 - How would you use a 250-mLvolumetric flask to...Ch. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - Prob. 64PCh. 8 - Prob. 65PCh. 8 - What is the molarity of a 20.0% (v/v) aqueous...Ch. 8 - Prob. 67PCh. 8 - Prob. 68PCh. 8 - Prob. 69PCh. 8 - Prob. 70PCh. 8 - Prob. 71PCh. 8 - Prob. 72PCh. 8 - Prob. 73PCh. 8 - Prob. 74PCh. 8 - Prob. 75PCh. 8 - Prob. 76PCh. 8 - Prob. 77PCh. 8 - Representations A (containing 1.0 mol ofNaCl) and...Ch. 8 - What is the boiling point of a solution that...Ch. 8 - Prob. 80PCh. 8 - If 150 g of ethylene glycol (C2H6O2) is added to...Ch. 8 - Prob. 82PCh. 8 - Prob. 83PCh. 8 - Prob. 84PCh. 8 - Which solution in each pair has the higher melting...Ch. 8 - Prob. 86PCh. 8 - A flask contains two compartments (A and B) with...Ch. 8 - A flask contains two compartments (A and B) with...Ch. 8 - The molecular art illustrates a red blood cell in...Ch. 8 - Prob. 90PCh. 8 - Prob. 91PCh. 8 - Explain why more sugar dissolves in a cup of hot...Ch. 8 - If the concentration of glucose in the blood is...Ch. 8 - Prob. 94PCh. 8 - Mannitol, a carbohydrate, is supplied as a 25%...Ch. 8 - A patient receives 750 ml, of a 10.% (w/v) aqueous...Ch. 8 - Explain why a cucumber placed in a concentrated...Ch. 8 - Explain why a cucumber placed in a concentrated...Ch. 8 - Prob. 99PCh. 8 - Prob. 100PCh. 8 - Prob. 101PCh. 8 - Prob. 102PCh. 8 - The therapeutic concentration—the concentration...Ch. 8 - Prob. 104CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A laser emits a line at 632.8 nm. If the cavity is 12 cm long, how many modes oscillate in the cavity? How long does it take for the radiation to travel the entire cavity? What is the frequency difference between 2 consecutive modes?(refractive index of the medium n = 1).arrow_forwardThe number of microstates corresponding to each macrostate is given by N. The dominant macrostate or configuration of a system is the macrostate with the greatest weight W. Are both statements correct?arrow_forwardFor the single step reaction: A + B → 2C + 25 kJ If the activation energy for this reaction is 35.8 kJ, sketch an energy vs. reaction coordinate diagram for this reaction. Be sure to label the following on your diagram: each of the axes, reactant compounds and product compounds, enthalpy of reaction, activation energy of the forward reaction with the correct value, activation energy of the backwards reaction with the correct value and the transition state. In the same sketch you drew, after the addition of a homogeneous catalyst, show how it would change the graph. Label any new line "catalyst" and label any new activation energy.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Solutions: Crash Course Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=9h2f1Bjr0p4;License: Standard YouTube License, CC-BY