
EP INTRO.TO GENERAL,ORGANIC...-OWL ACCE
12th Edition
ISBN: 9781337915984
Author: Bettelheim
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8.5, Problem 8.4QC
Problem 8-4
Which is the stronger acid?
(a) Carbonic acid, 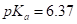 or ascorbic acid (vitamin C),
or ascorbic acid (vitamin C), 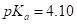
(b) Aspirin, 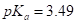 or acetic acid,
or acetic acid, 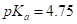
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
None
None
None
Chapter 8 Solutions
EP INTRO.TO GENERAL,ORGANIC...-OWL ACCE
Ch. 8.3 - Problem 8-1 Draw the acid and base reactions for...Ch. 8.4 - Prob. 8.2QCCh. 8.5 - Prob. 8.3QCCh. 8.5 - Problem 8-4 Which is the stronger acid? (a)...Ch. 8.6 - Problem 8-5 Write the balanced net ionic equation...Ch. 8.7 - Problem 8-6 The [OH-] of an aqueous solution is M....Ch. 8.8 - Problem 8-7 (a) The [H3O+] of an acidic solution...Ch. 8.8 - Problem 8-8 The [OH-] of a solution is M. What are...Ch. 8.9 - Problem 8-9 Calculate the concentration of an...Ch. 8.10 - Problem 8-10 What is the pH of a buffer solution...
Ch. 8.11 - Problem 8-11 What is the pH of a boric acid buffer...Ch. 8.12 - Prob. 8.12QCCh. 8 - 8-13 Define (a) an Arrhenius acid and (b) an...Ch. 8 - 8-14 Write an equation for the reaction that takes...Ch. 8 - 8-15 Write an equation for the reaction that takes...Ch. 8 - 8-16 For each of the following, tell whether the...Ch. 8 - 8-17 For each of the following, tell whether the...Ch. 8 - 8-18 Which of these acids are monoprotic, which...Ch. 8 - 8-19 Define (a) a Brønsted—Lowry acid and (b) a...Ch. 8 - 8-20 Write the formula for the conjugate base of...Ch. 8 - 8-21 Write the formula for the conjugate base of...Ch. 8 - Prob. 10PCh. 8 - Prob. 11PCh. 8 - Prob. 12PCh. 8 - 8-25 Draw the acid and base reactions for the...Ch. 8 - Prob. 14PCh. 8 - Prob. 15PCh. 8 - 8-28 Will carbon dioxide be evolved as a gas when...Ch. 8 - Prob. 17PCh. 8 - Prob. 18PCh. 8 - Prob. 19PCh. 8 - Prob. 20PCh. 8 - 8-33 Write an equation for the reaction of HCI...Ch. 8 - 8-34 When a solution of sodium hydroxide is added...Ch. 8 - 8-35 Given the following values of [H3O+),...Ch. 8 - 8-36 Given the following values of [OH-],...Ch. 8 - 8-37 What is the pH of each solution given the...Ch. 8 - 8-38 What is the pH and pOH of each solution given...Ch. 8 - 8-39 What is the pH of each solution given the...Ch. 8 - Prob. 28PCh. 8 - 8-41 What is the [OH-] and pOH of each solution?...Ch. 8 - Prob. 30PCh. 8 - 8-43 What is the molarity of a solution made by...Ch. 8 - 8-44 What is the molarity of a solution made by...Ch. 8 - 8-45 Describe how you would prepare each of the...Ch. 8 - 8-46 If 25.0 mL of an aqueous solution of H2SO4...Ch. 8 - 8-47 A sample of 27.0 mL of 0.310 M NaOH is...Ch. 8 - 8-48 A 0.300 M solution of H2SO4 was used to...Ch. 8 - 8-49 A solution of NaOH base was titrated with...Ch. 8 - 8-50 The usual concentration of HCO3- ions in...Ch. 8 - 8-51 What is the end point of a titration?Ch. 8 - Prob. 40PCh. 8 - 8-53 Write equations to show what happens when, to...Ch. 8 - 8-54 Write equations to show what happens when, to...Ch. 8 - 8-55 We commonly refer to a buffer as consisting...Ch. 8 - Prob. 44PCh. 8 - Prob. 45PCh. 8 - 8-58 What is the connection between buffer action...Ch. 8 - Prob. 47PCh. 8 - 8-60 How is the buffer capacity affected by the...Ch. 8 - 8-61 Can 100 of 0.1 M phosphate buffer at pH 7.2...Ch. 8 - 8-62 What is the pH of a buffer solution made by...Ch. 8 - 8-63 The pH of a solution made by dissolving 1.0...Ch. 8 - Prob. 52PCh. 8 - Prob. 53PCh. 8 - Prob. 54PCh. 8 - Prob. 55PCh. 8 - 8-66 Calculate the pH of an aqueous solution...Ch. 8 - Prob. 57PCh. 8 - 8-68 If you have 100 mL of a 0.1 M buffer made of...Ch. 8 - Prob. 59PCh. 8 - Prob. 60PCh. 8 - 8-71 Explain why you do not need to know the...Ch. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - Prob. 64PCh. 8 - Prob. 65PCh. 8 - 8-76 (Chemical Connections 8B) Name the most...Ch. 8 - Prob. 67PCh. 8 - Prob. 68PCh. 8 - 8-79 (Chemical Connections 8D) Another form of the...Ch. 8 - Prob. 70PCh. 8 - Prob. 71PCh. 8 - 8-82 Assume that you have a dilute solution of HCI...Ch. 8 - Prob. 73PCh. 8 - Prob. 74PCh. 8 - Prob. 75PCh. 8 - 8-86 Following are three organic acids and the...Ch. 8 - 8-87 The pKavalue of barbituric acid is 5.0. If...Ch. 8 - Prob. 78PCh. 8 - Prob. 79PCh. 8 - Prob. 80PCh. 8 - Prob. 81PCh. 8 - Prob. 82PCh. 8 - 8-93 Do a 1.0 M CH3COOH solution and a 1.0 M HCI...Ch. 8 - 8-94 Suppose you wish to make a buffer whose pH is...Ch. 8 - Prob. 85PCh. 8 - 8-96 Suppose you want to make a CH3COOH/CH3COO-...Ch. 8 - Prob. 87PCh. 8 - 8-98 When a solution prepared by dissolving 4.00 g...Ch. 8 - Prob. 89PCh. 8 - Prob. 90PCh. 8 - 8-101 Suppose you have an aqueous solution...Ch. 8 - Prob. 92PCh. 8 - 8-103 Suppose you have a phosphate buffer...Ch. 8 - Prob. 94PCh. 8 - Prob. 95PCh. 8 - Prob. 96PCh. 8 - 8-107 Following are pH ranges for several human...Ch. 8 - Prob. 98PCh. 8 - Prob. 99PCh. 8 - 8-108 What is the ratio of HPO42-/H2PO4- in a...Ch. 8 - Prob. 101PCh. 8 - 8-110 A concentrated hydrochloric acid solution...Ch. 8 - 8-111 The volume of an adult's stomach ranges from...Ch. 8 - 8-112 Consider an initial 0.040 M hypobromous acid...Ch. 8 - Prob. 105PCh. 8 - Prob. 106PCh. 8 - 8-115 When a solution prepared by dissolving 0.125...Ch. 8 - 8-116 A railroad tank car derails and spills 26...Ch. 8 - Prob. 109PCh. 8 - Prob. 110PCh. 8 - Prob. 111P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Consider the structure of 1-bromo-2-fluoroethane. Part 1 of 2 Draw the Newman projection for the anti conformation of 1-bromo-2-fluoroethane, viewed down the C1-C2 bond. ✡ ぬ Part 2 of 2 H H F Br H H ☑ Draw the Newman projection for the gauche conformation of 1-bromo-2-fluoroethane, viewed down the C1-C2 bond. H F Br H Harrow_forwardPlease help me answer this question. I don't understand how or where the different reagents will attach and it's mostly due to the wedge bond because I haven't seen a problem like this before. Please provide a detailed explanation and a drawing showing how it can happen and what the final product will look like.arrow_forwardWhich of the following compounds is the most acidic in the gas phase? Group of answer choices H2O SiH4 HBr H2Sarrow_forward
- Which of the following is the most acidic transition metal cation? Group of answer choices Fe3+ Sc3+ Mn4+ Zn2+arrow_forwardBased on the thermodynamics of acetic acid dissociation discussed in Lecture 2-5, what can you conclude about the standard enthalpy change (ΔHo) of acid dissociation for HCl? Group of answer choices You cannot arrive at any of the other three conclusions It is a positive value It is more negative than −0.4 kJ/mol It equals −0.4 kJ/molarrow_forwardPLEASE HELP URGENT!arrow_forward
- Draw the skeletal structure corresponding to the following IUPAC name: 7-isopropyl-3-methyldecanearrow_forwardWhich of the following oxyacids is the weakest? Group of answer choices H2SeO3 Si(OH)4 H2SO4 H3PO4arrow_forwardAdd conditions above and below the arrow that turn the reactant below into the product below in a single transformation. + More... If you need to write reagents above and below the arrow that have complex hydrocarbon groups in them, there is a set of standard abbreviations you can use. More... T H,N NC Datarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY