
Concept explainers
(a)
Interpretation:
The Lewis structure of oleum has to be drawn.
Concept Introduction:
Lewis Structure: A Lewis structure shows a covalent bond as pair of electrons shared between two atoms.
Procedure to write Lewis formulas:
- 1) The symbols of the atoms that are bonded together in the molecule next to one another are arranged.
- 2) The total number of valence electrons in the molecule is calculated by adding the number of valence electrons for all the atoms in the molecules. If the species is an ion, then the charge of ion into account by adding electrons, if it is a negative ion or subtracting electrons if it is a positive ion.
- 3) A two-electron covalent bond is represented by placing a line between the atoms, which are assumed to be bonded to each other.
- 4) The remaining valence electrons as lone pairs about each atom are arranged so that the octet rule is satisfied for each other.
(a)
Answer to Problem 8H.8E
The Lewis structure of oleum is,
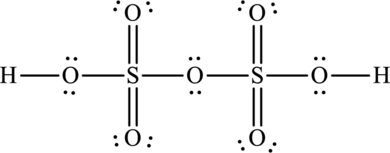
Explanation of Solution
Oleum is otherwise known as pyrosulphuric acid and its formula is
The number of valence electrons in hydrogen=
The number of valence electrons in oxygen=
The number of valence electrons in sulphur=
The total number of valence electrons in oleum is fifty-six.
The Lewis structure of oleum is,
(b)
Interpretation:
The formal charge on sulphur and oxygen in oleum has to be determined.
Concept Introduction:
Formal charge (F.C): The charges that assigned to each atom in a molecule or ion by a set of arbitrary rules and don not actually represent the actual charges on the atoms are called as formal charges.
The formal charge is calculated using the formula,
The Lewis structure with zero formal charge or least separated formal charges is the preferred structure of the molecule.
(b)
Answer to Problem 8H.8E
The formal charge in sulphur is zero.
The formal charge in oxygen is zero.
Explanation of Solution
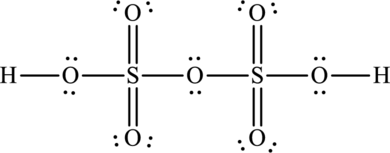
Oleum is otherwise known as pyrosulphuric acid and its formula is
The number of valence electrons in hydrogen=
The number of valence electrons in oxygen=
The number of valence electrons in sulphur=
The total number of valence electrons in oleum is fifty-six.
The Lewis structure of oleum is,
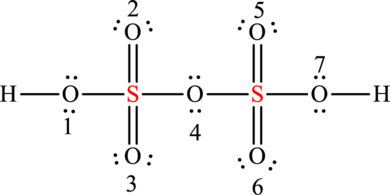
The formal charges on sulphur is,
The formal charge in sulphur is zero.
The formal charges on oxygen
The formal charge on oxygen
The formal charge on oxygen
The formal charge on oxygen
(c)
Interpretation:
The oxidation number of sulphur in oleum has to be drawn.
(c)
Answer to Problem 8H.8E
The oxidation number of sulphur in oleum is
Explanation of Solution
Oleum is otherwise known as pyrosulphuric acid and its formula is
The oxidation number of sulphur in
Want to see more full solutions like this?
Chapter 8 Solutions
ACHIEVE/CHEMICAL PRINCIPLES ACCESS 2TERM
- 1. Part 1: Naming Organic Compounds он H₁C-C-CH3 CH3 Br CI CI 2. Br-CH-CH-CH₂ H₂C-CH-C= -CH-CH2-CH3 3. HC-CH-CH-C-OH 5. H₂C-CH-CH₂-OH 7. OH 4. CH CH₂-CH₂ 6. сно CH-CH-CH-CH₂-CH₂ H₁₂C-CH-CH-CH-CH₁₂-CH₁₂ 8. OHarrow_forward11 Organic Chemistry Organic Nomenclature Practice Name/Functional Group n-butane Formula Structural Formula (1) C4tt10 H3C C- (2) CH3CH2CH2 CH 3 H₂ -CH3 Н2 name & functional group (1) and (2) OH H₁₂C Н2 name only (1) and (2) name only (1) and (2) H₁C - = - CH₂ Н2 HC=C-C CH3arrow_forwardUnder aqueous basic conditions, nitriles will react to form a neutral organic intermediate 1 that has an N atom in it first, and then they will continue to react to form the final product 2: NC H₂O он- H₂O 1 2 OH Draw the missing intermediate 1 and the final product 2 in the box below. You can draw the two structures in any arrangement you like. Click and drag to start drawing a structure.arrow_forward
- Assign these COSY Spectrumarrow_forwardAssign these C-NMR and H-NMR Spectrumarrow_forwardPredict the product of this organic reaction: IZ + HO i P+H₂O Specifically, in the drawing area below draw the skeletal ("line") structure of P. If there is no reasonable possibility for P, check the No answer box under the drawing area. No Answer Click and drag to start drawing a structure. ☐ :arrow_forward
- Predict the products of this organic reaction: 0 O ----- A + KOH ? CH3-CH2-C-O-CH2-C-CH3 Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. (If there's more than one product, draw them in any arrangement you like, so long as they aren't touching.) If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No reaction Click anywhere to draw the first atom of your structure. X ⑤ èarrow_forwardPredict the products of this organic reaction: O CH3 + H2O + HCI A A? CH3-CH2-C-N-CH3 Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. If there's more than one product, draw them in any arrangement you like, so long as they aren't touching. If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No Reaction Click anywhere to draw the first atom of your structure.arrow_forwardWhat is the missing reactant in this organic reaction? R+ HO-C-CH2-CH3 0= CH3 CH3 —CH, C−NH—CH CH3 + H₂O Specifically, in the drawing area below draw the condensed structure of R. If there is more than one reasonable answer, you can draw any one of them. If there is no reasonable answer, check the No answer box under the drawing area. Note for advanced students: you may assume no products other than those shown above are formed. No Answer Click anywhere to draw the first atom of your structure. €arrow_forward
- 个 CHEM&131 9267 - $25 - Intro to Mail - Hutchison, Allison (Student x Aktiv Learnin https://app.aktiv.com Draw the product of the reaction shown below. Ignore inorganic byproducts. + Na2Cr2O7 Acetone, H2SO4 Type here to search Dryng OH W Prarrow_forwardPredict the products of this organic reaction: OH + NaOH A? Specifically, in the drawing area below draw the skeletal ("line") structure of the product, or products, of this reaction. (If there's more than one product, draw them in any arrangement you like, so long as they aren't touching.) If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No reaction Click and drag to start drawing a structure. ✓ Sarrow_forwardPredict the products of this organic reaction: CH3-C-O-CH2-CH2-C-CH3 + H₂O ? A Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. (If there's more than one product, draw them in any arrangement you like, so long as they aren't touching.) If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No reaction Click anywhere to draw the first atom of your structure. :☐ darrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
