
(a)
Interpretation:The denser substance out of graphite and diamond needs to be determined on the basis of given P-T curve.
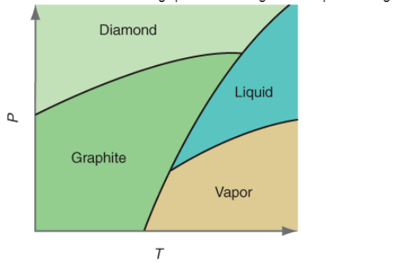
Concept Introduction:All substances can mainly exist in three phases; solid, liquid and gas. These three phases can convert into each other by the application of temperature and pressure such as heating of solid leads to the conversion to liquid and then gaseous
Similarly, the gases can be compressed at high pressure to liquefy. The interconversion of three phases of substance can be shown on the phase diagram.
(b)
Interpretation:Themore denser substance out of graphite and liquid carbon needs to be determined on the basis of given P-T curve.
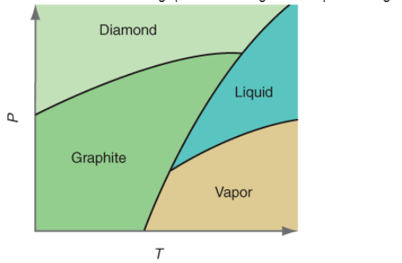
Concept Introduction:All substances can mainly exist in three phases; solid, liquid and gas. These three phases can convert into each other by the application of temperature and pressure such as heating of solid leads to the conversion to liquid and then gaseous state of matter.
Similarly, the gases can be compressed at high pressure to liquefy. The interconversion of three phases of substance can be shown on the phase diagram.
(c)
Interpretation:The reason of presence of two triple points needs to be explained in the P-T curve for carbon.
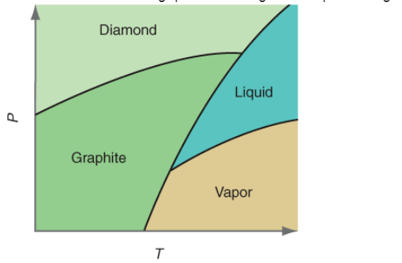
Concept Introduction:All substances can mainly exist in three phases; solid, liquid and gas. These three phases can convert into each other by the application of temperature and pressure such as heating of solid leads to the conversion to liquid and then gaseous state of matter.
Similarly, the gases can be compressed at high pressure to liquefy. The interconversion of three phases of substance can be shown on the phase diagram.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
EBK PHYSICAL CHEMISTRY
- help 20arrow_forwardProvide the drawing of the unknown structure that corresponds with this data.arrow_forward20.44 The Diels-Alder reaction is not limited to making six-membered rings with only car- bon atoms. Predict the products of the following reactions that produce rings with atoms other than carbon in them. OCCH OCCH H (b) CH C(CH₂)s COOCH མ་ནས་བ (c) N=C H -0.X- (e) H C=N COOCHS + CH2=CHCH₂ →→arrow_forward
- 3) Draw a detailed mechanism and predict the product of the reaction shown? 1) EtMgBr 2) H3O+arrow_forwardHow to draw the mechanism for this reaction?arrow_forward> H₂C=C-CH2-CH3 B. H₂O Pt C. + H2 + H₂O H D. 16. Give the IUPAC name for each of the following: B. Cl Cl c. Cl Cl 17. Draw the line-angle formula for each of the following compounds: 1. phenol 2. 1,3-dichlorobenzene 3. 4-ethyltoluene < Previous Submit Assignment Next ▸arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





