
Concept explainers
Draw all products, including stereoisomers, in each reaction.
a.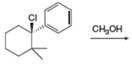 c.
c.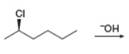
b.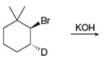 d.
d.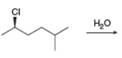
(a)
Interpretation: All products formed in the given reaction are to be drawn.
Concept introduction: The one-step bimolecular elimination reaction that favors the removal of a proton by a base from carbon adjacent to the leaving group that results in the formation of a carbocation is termed as
The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
Stereoisomers have the same molecular formula but they differ in the three-dimensional arrangement of their bonds.
Answer to Problem 8.56P
The products that are formed in the given reaction are
Explanation of Solution
In the given reaction, the secondary alkyl halide is treated with a strong base due to which both substitution and elimination reactions takes place with the given secondary alkyl halide. This reaction results in the formation of three alkenes via
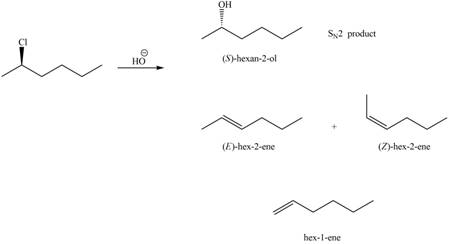
Figure 1
Thus, the products formed in the given reaction are
The products that are formed in the given reaction are
(b)
Interpretation: All products formed in the given reaction are to be drawn.
Concept introduction: The one-step bimolecular elimination reaction that favors the removal of a proton by a base from carbon adjacent to the leaving group that results in the formation of a carbocation is termed as
The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
Stereoisomers have the same molecular formula but they differ in the three-dimensional arrangement of their bonds.
Answer to Problem 8.56P
The products that are formed in the given reaction are s
Explanation of Solution
In the given reaction, the secondary alkyl halide is treated with a strong base due to which both substitution and elimination reactions takes place with the given secondary alkyl halide. This reaction results in the formation of three alkenes via
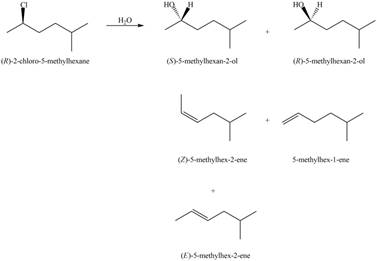
Figure 2
Thus, the products formed in the given reaction are s
The products that are formed in the given reaction are s
(c)
Interpretation: All products formed in the given reaction are to be drawn.
Concept introduction: The two-step unimolecular elimination reaction that favors the removal of a HX substituent and the formation of a carbocation intermediate takes place in its first step. In the second step of the reaction, the carbocation forms a double bond. This type of reaction is termed as
The two-step unimolecular reaction which favors the removal of a HX substituent and the formation of a carbocation intermediate takes place in its first step. Then, in the second step, the carbocation undergoes substitution. This type of reaction is termed as
Stereoisomers have the same molecular formula but they differ in the three-dimensional arrangement of their bonds.
Answer to Problem 8.56P
The products that are formed in the given reaction are
Explanation of Solution
The given reaction takes place between the tertiary alkyl halide and methanol that acts as a weak base as well as a weak nucleophile. The given tertiary alkyl halide undergoes elimination reaction via
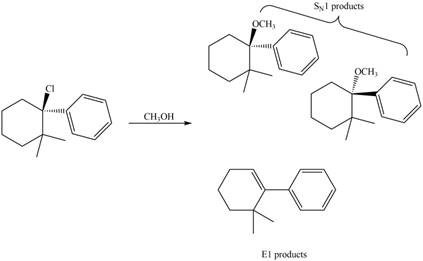
Figure 3
Thus, the products formed in the given reaction are
The products that are formed in the given reaction are
(d)
Interpretation: All products formed in the given reaction are to be drawn.
Concept introduction: The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
Stereoisomers have the same molecular formula but they differ in the three-dimensional arrangement of their bonds.
Answer to Problem 8.56P
The product that is formed in the given reaction is shown in Figure 4.
Explanation of Solution
In the given reaction, the secondary alkyl halide is treated with a strong base due to which both substitution and elimination reactions takes place with the given secondary alkyl halide. This reaction results in the formation of three alkenes via
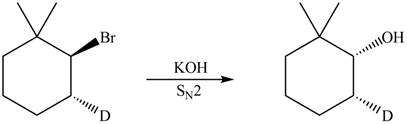
Figure 4
The product that is formed in the given reaction is shown in Figure 4.
Want to see more full solutions like this?
Chapter 8 Solutions
ORGANIC CHEMISTRY
Additional Science Textbook Solutions
Cosmic Perspective Fundamentals
Organic Chemistry
Human Physiology: An Integrated Approach (8th Edition)
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
- Calculate the variation in the potential of the Pt/MnO4-, Mn2+ pair with pH, indicating the value of the standard potential. Data: E0 = 1.12.arrow_forwardGiven the cell: Pt l H2(g) l dis X:KCl (sat) l Hg2Cl2(s) l Hg l Pt. Calculate the emf of the cell as a function of pH.arrow_forwardThe decimolar calomel electrode has a potential of 0.3335 V at 25°C compared to the standard hydrogen electrode. If the standard reduction potential of Hg22+ is 0.7973 V and the solubility product of Hg2Cl2 is 1.2x 10-18, find the activity of the chlorine ion at this electrode.Data: R = 8.314 J K-1 mol-1, F = 96485 C mol-1, T = 298.15 K.arrow_forward
- 2. Add the following group of numbers using the correct number of significant figures for the answer. Show work to earn full credit such as rounding off the answer to the correct number of significant figures. Replace the question marks with the calculated answers or write the calculated answers near the question marks. 10916.345 37.40832 5.4043 3.94 + 0.0426 ? (7 significant figures)arrow_forwardThe emf at 25°C of the cell: Pt l H2(g) l dis X:KCl (sat) l Hg2Cl2(s) l Hg l Pt was 612 mV. When solution X was replaced by normal phosphate buffer solution with a pH of 6.86, the emf was 741 mV. Calculate the pH of solution X.arrow_forwardIndicate how to calculate the potential E of the reaction Hg2Cl2(s) + 2e ⇄ 2Hg + 2Cl- as a function of the concentration of Cl- ions. Data: the solubility product of Hg2Cl2.arrow_forward
- How can Beer’s Law be used to determine the concentration in a selected food sample. Provide an in-depth discussion and examples of this.arrow_forwardb) H3C- H3C Me CH 3 I HN Me H+arrow_forwardUsing Luther's rule, determine the reference potentials of the electrodes corresponding to the low stability systems Co³+/Co and Cr²+/Cr from the data in the table. Electrodo ΕΝ Co²+/Co Co3+/Co²+ -0,28 +1,808 Cr³+ / Cr -0,508 Cr3+ / Cr²+ -0,41arrow_forward
- The molecule PYRIDINE, 6tt electrons and is there pore aromuntre and is Assigned the Following structure contenus Since aromatk moleculey undergo electrophilic allomatic substitution, Pyridine should undergo The Following reaction + HNO3 12504 a. write all of the possible Mononitration Products that could Result From this roaction Based upon the reaction the reaction mechanism determine which of these producty would be the major Product of the hegetionarrow_forwardUsing Benzene as starting materia Show how each of the Following molecules could Ve synthesked 9. CHI d. 10450 b 0 -50311 ८ City -5034 1-0-650 e NO2arrow_forwardBA HBr of the fol 1)=MgCI 2) H₂O major NaOEt Ts Cl Py (pyridine) 1) 03 2) Me2S 1arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





