
Chemistry: Atoms First
2nd Edition
ISBN: 9780073511184
Author: Julia Burdge, Jason Overby Professor
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 8.46QP
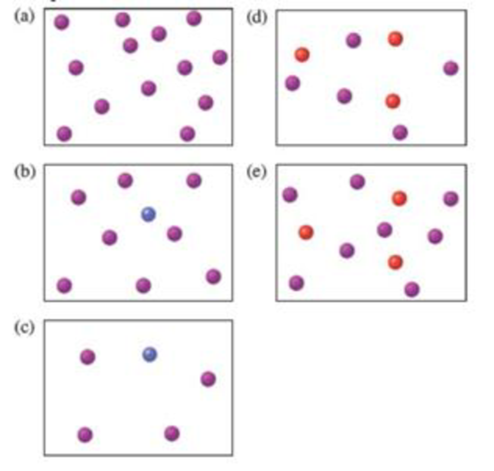
Reactants A (red) and B (blue) combine in the reaction vessel shown to form a single product C (purple) according to the equation 2A + B → C.
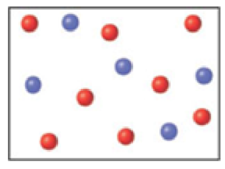
Which of the following scenes represents the contents of the reaction vessel after the reaction is complete?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw mechanism
Does Avogadro's number have units?
Explain why the total E in an Einstein depends on the frequency or wavelength of the light.
Chapter 8 Solutions
Chemistry: Atoms First
Ch. 8.1 - Write and balance the chemical equation for the...Ch. 8.1 - Write and balance the chemical equation that...Ch. 8.1 - Prob. 1PPBCh. 8.1 - Prob. 1PPCCh. 8.1 - Butyric acid (also known as butanoic acid,...Ch. 8.1 - Another compound found in milk fat that appears to...Ch. 8.1 - Prob. 2PPBCh. 8.1 - Prob. 2PPCCh. 8.1 - Prob. 8.3WECh. 8.1 - Prob. 3PPA
Ch. 8.1 - Using the chemical species A2, B, and AB, write a...Ch. 8.1 - Prob. 3PPCCh. 8.1 - Prob. 8.1.1SRCh. 8.1 - Prob. 8.1.2SRCh. 8.1 - Prob. 8.1.3SRCh. 8.1 - Prob. 8.1.4SRCh. 8.1 - Prob. 8.1.5SRCh. 8.2 - Combustion of a 5.50-g sample of benzene produces...Ch. 8.2 - The combustion of a 28.1-g sample of ascorbic acid...Ch. 8.2 - Prob. 4PPBCh. 8.2 - Prob. 4PPCCh. 8.2 - Prob. 8.2.1SRCh. 8.2 - Prob. 8.2.2SRCh. 8.2 - Prob. 8.2.3SRCh. 8.3 - Prob. 8.5WECh. 8.3 - Nitrogen and hydrogen react to form ammonia...Ch. 8.3 - Prob. 5PPBCh. 8.3 - Prob. 5PPCCh. 8.3 - Prob. 8.6WECh. 8.3 - Calculate the mass of water produced by the...Ch. 8.3 - Prob. 6PPBCh. 8.3 - The models here represent the reaction of nitrogen...Ch. 8.3 - Prob. 8.3.1SRCh. 8.3 - Prob. 8.3.2SRCh. 8.3 - Prob. 8.3.3SRCh. 8.3 - Prob. 8.3.4SRCh. 8.4 - Alka-Seltzer tablets contain aspirin, sodium...Ch. 8.4 - Ammonia is produced by the reaction of nitrogen...Ch. 8.4 - Prob. 7PPBCh. 8.4 - The diagrams show a reaction mixture before and...Ch. 8.4 - Aspirin, acetylsalicylic acid (C9H8O4), is the...Ch. 8.4 - Diethyl ether is produced from ethanol according...Ch. 8.4 - What mass of ether will be produced if 207 g of...Ch. 8.4 - The diagrams show a mixture of reactants and the...Ch. 8.4 - Prob. 8.4.1SRCh. 8.4 - Prob. 8.4.2SRCh. 8.4 - Prob. 8.4.3SRCh. 8.4 - Prob. 8.4.4SRCh. 8.4 - Prob. 8.4.5SRCh. 8 - Prob. 8.1QPCh. 8 - Prob. 8.2QPCh. 8 - Why must a chemical equation he balanced? What law...Ch. 8 - Write an unbalanced equation to represent each of...Ch. 8 - Prob. 8.5QPCh. 8 - Prob. 8.6QPCh. 8 - For each of the following unbalanced chemical...Ch. 8 - Prob. 8.8QPCh. 8 - Balance the following equations using the method...Ch. 8 - Which of the following equations best represents...Ch. 8 - Prob. 8.11QPCh. 8 - Determine whether each of the following equations...Ch. 8 - Prob. 8.13QPCh. 8 - Prob. 8.14QPCh. 8 - Prob. 8.15QPCh. 8 - Prob. 8.16QPCh. 8 - Prob. 8.17QPCh. 8 - Prob. 8.18QPCh. 8 - Prob. 8.19QPCh. 8 - Prob. 8.20QPCh. 8 - Prob. 8.21QPCh. 8 - Prob. 8.22QPCh. 8 - Prob. 8.23QPCh. 8 - On what law is stoichiometry based? Why is it...Ch. 8 - Prob. 8.25QPCh. 8 - Prob. 8.26QPCh. 8 - Prob. 8.27QPCh. 8 - Prob. 8.28QPCh. 8 - Prob. 8.29QPCh. 8 - Prob. 8.30QPCh. 8 - Prob. 8.31QPCh. 8 - Prob. 8.32QPCh. 8 - Prob. 8.33QPCh. 8 - When copper(II) sulfate pentahydrate (CuSO4 5H2O)...Ch. 8 - For many years, the extraction of gold from other...Ch. 8 - Prob. 8.36QPCh. 8 - Nitrous oxide (N2O) is also called laughing gas....Ch. 8 - Prob. 8.38QPCh. 8 - Prob. 8.39QPCh. 8 - Prob. 8.1VCCh. 8 - Prob. 8.2VCCh. 8 - Prob. 8.3VCCh. 8 - Prob. 8.4VCCh. 8 - Prob. 8.40QPCh. 8 - Prob. 8.41QPCh. 8 - Why is the theoretical yield of a reaction...Ch. 8 - Why is the actual yield of a reaction almost...Ch. 8 - Prob. 8.44QPCh. 8 - Prob. 8.45QPCh. 8 - Reactants A (red) and B (blue) combine in the...Ch. 8 - Prob. 8.47QPCh. 8 - Prob. 8.48QPCh. 8 - Prob. 8.49QPCh. 8 - Propane (C3H8) is a minor component of natural gas...Ch. 8 - Prob. 8.51QPCh. 8 - Prob. 8.52QPCh. 8 - Prob. 8.53QPCh. 8 - Prob. 8.54QPCh. 8 - Prob. 8.55QPCh. 8 - Prob. 8.56QPCh. 8 - Disulfur dichloride (S2Cl2) is used in the...Ch. 8 - Prob. 8.58QPCh. 8 - Prob. 8.59QPCh. 8 - Prob. 8.60QPCh. 8 - Prob. 8.61QPCh. 8 - Prob. 8.62QPCh. 8 - Prob. 8.63QPCh. 8 - Prob. 8.64QPCh. 8 - Prob. 8.65QPCh. 8 - Industrially, nitric acid is produced by the...Ch. 8 - Prob. 8.67QPCh. 8 - Prob. 8.68QPCh. 8 - Prob. 8.69QPCh. 8 - Prob. 8.70QPCh. 8 - Prob. 8.71QPCh. 8 - Prob. 8.72QPCh. 8 - Prob. 8.73QPCh. 8 - Prob. 8.74QPCh. 8 - Prob. 8.75QPCh. 8 - Prob. 8.76QPCh. 8 - Prob. 8.77QPCh. 8 - Prob. 8.78QPCh. 8 - Prob. 8.79QPCh. 8 - The combustion of a 5.50-g sample of oxalic acid...Ch. 8 - Prob. 8.81QPCh. 8 - Prob. 8.82QPCh. 8 - Prob. 8.83QPCh. 8 - Prob. 8.84QPCh. 8 - Prob. 8.85QPCh. 8 - Prob. 8.86QPCh. 8 - Potash is any potassium mineral that is used for...Ch. 8 - A 21.496-g sample of magnesium is burned in air to...Ch. 8 - Prob. 8.89QPCh. 8 - Prob. 8.90QPCh. 8 - Prob. 8.91QPCh. 8 - Prob. 8.92QPCh. 8 - Prob. 8.93QPCh. 8 - Prob. 8.94QPCh. 8 - Prob. 8.95QPCh. 8 - Prob. 8.96QPCh. 8 - Prob. 8.97QPCh. 8 - Prob. 8.98QPCh. 8 - A compound X contains 63.3 percent manganese (Mn)...Ch. 8 - Calculate the mass of water produced in the...Ch. 8 - Calcium phosphide (Ca3P2) and water react to form...Ch. 8 - Prob. 8.3KSPCh. 8 - Prob. 8.4KSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- If the dissociation energy of one mole of O2 is 5.17 eV, determine the wavelength that must be used to dissociate it with electromagnetic radiation. Indicate how many Einstein's of this radiation are needed to dissociate 1 liter of O2 at 25°C and 1 atm of pressure.Data: 1 eV = 96485 kJ mol-1; R = 0.082 atm L K-1; c = 2.998x108 m s-1; h = 6.626x10-34 J s; NA = 6.022x 1023 mol-1arrow_forwardIndicate the number of Einsteins that are equivalent to 550 kJ mol⁻¹ of absorbed energy (wavelength 475 nm).arrow_forwardIndicate the number of einsteins that are equivalent to 550 kJ mol⁻¹ of absorbed energy?arrow_forward
- A unit used in photochemistry is the einstein. If 400 kJ mol-1 of energy has been absorbed, how many einsteins is this equivalent to?arrow_forwardFor the condensation reaction between Alanine and histidine write the amididation reaction mechanism using arrows then write the three letter code for the product of the reaction and the one letter code for the product of the reaction.arrow_forwardWrite the amididation reaction mechanism of p-aminophenol and acetic acid to produce acetaminophen please use arrows.arrow_forward
- Write the amididation reaction mechanism of a-aminophenol and acetic acid to produce acetaminophenarrow_forwardFor the condensation reaction between Alamine and histamine, please help me write the amididation reaction mechanism. Then write the three letter code for the product of the reaction, then write the one letter code for the product of the reaction. arrow_forwardHow to draw the reaction mechasnism belowarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY