
(a)
Interpretation:
The missing C3H5 and OH substituents needs to be added to the template in order to get one possible chair conformation for each compound.
Concept Introduction:
Chair-like conformation for cyclohexane molecule is a three-dimensional way of representing the molecule. This is a non-planar conformation with all the angles approximately equal to 109.5o. This is stable conformation because it has lower strain energy than the flat hexagonal shape. All the carbon centers are equivalent in the chair conformation. In the flat hexagonal shape, the wedge and dashed bonds represent the group attached directed away and towards the plane of paper respectively. In the chair conformation, these wedge and dashed bonds are axial and equatorial respectively.
(b)
Interpretation:
The most appropriate method from MM2 or MOPAC to determine whether compound 1 or 2 is most stable product in each reaction needs to be determined.
Concept Introduction:
Computational chemistry uses computer simulation to monitor solution of chemical problems. The theoretical chemistry is used in which efficient computer programs are incorporated to determine the structures and properties of molecules.
In the computational chemistry, the MM2 and MOPAC methods are defined as follows:
MM2:
This method is developed mainly for conformational analysis of small organic compounds such as hydrocarbons. This is designed to reproduce the equilibrium covalent geometry of molecules as close as possible.
MOPAC:
This is a computer program used in the computational chemistry for semi-empirical quantum chemistry algorithms. It runs on Mac, Windows and Linux.
(c)
Interpretation:
The number of chair-like conformation for the energy minimization needs to be determined. The alcohol and
Concept Introduction:
Chair-like conformation for cyclohexane molecule is a three-dimensional way of representing the molecule. This is a non-planar conformation with all the angles approximately equal to 109.5o. This is stable conformation because it has lower strain energy than the flat hexagonal shape. All the carbon centers are equivalent in the chair conformation. In the flat hexagonal shape, the wedge and dashed bonds represent the group attached directed away and towards the plane of paper respectively. In the chair conformation, these wedge and dashed bonds are axial and equatorial respectively.
(d)
Interpretation:
The input structure for compound 1 needs to be determined.
Concept Introduction:
Computational chemistry uses computer simulation to monitor solution of chemical problems. The theoretical chemistry is used in which efficient computer programs are incorporated to determine the structures and properties of molecules.
MM2 method is developed mainly for conformational analysis of small organic compounds such as hydrocarbons. This is designed to reproduce the equilibrium covalent geometry of molecules as close as possible.
The compound 1 is given as follows:
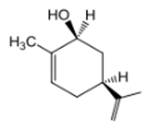
The chair conformation is represented as follows:
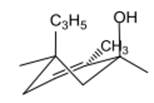
Here, both C3H5 and OH groups are at axial positions.
Thus, the input for compound 1 is represented as follows:
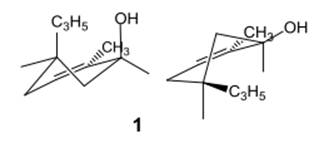
(e)
Interpretation:
The input structure for compound 2 needs to be determined.
Concept Introduction:
Computational chemistry uses computer simulation to monitor solution of chemical problems. The theoretical chemistry is used in which efficient computer programs are incorporated to determine the structures and properties of molecules.
MM2 method is developed mainly for conformational analysis of small organic compounds such as hydrocarbons. This is designed to reproduce the equilibrium covalent geometry of molecules as close as possible.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Laboratory Techniques in Organic Chemistry
- Identify the unknown compound from its IR and proton NMR spectra. C4H6O: 'H NMR: 82.43 (1H, t, J = 2 Hz); 8 3.41 (3H, s); 8 4.10 (2H, d, J = 2 Hz) IR: 2125, 3300 cm¹ The C4H6O compound liberates a gas when treated with C2H5 MgBr. Draw the unknown compound. Select Draw с H Templates Morearrow_forwardPlease help with number 6 I got a negative number could that be right?arrow_forward1,4-Dimethyl-1,3-cyclohexadiene can undergo 1,2- or 1,4-addition with hydrogen halides. (a) 1,2-Addition i. Draw the carbocation intermediate(s) formed during the 1,2-addition of hydrobromic acid to 1,4-dimethyl-1,3-cyclohexadiene. ii. What is the major 1,2-addition product formed during the reaction in (i)? (b) 1,4-Addition i. Draw the carbocation intermediate(s) formed during the 1,4-addition of hydrobromic acid to 1,4-dimethyl-1,3-cyclohexadiene. ii. What is the major 1,4-addition product formed from the reaction in (i)? (c) What is the kinetic product from the reaction of one mole of hydrobromic acid with 1,4-dimethyl-1,3-cyclohexadiene? Explain your reasoning. (d) What is the thermodynamic product from the reaction of one mole of hydrobro-mic acid with 1,4-dimethyl-1,3-cyclohexadiene? Explain your reasoning. (e) What major product will result when 1,4-dimethyl-1,3-cyclohexadiene is treated with one mole of hydrobromic acid at - 78 deg * C ? Explain your reasoning.arrow_forward
- Give the product of the bimolecular elimination from each of the isomeric halogenated compounds. Reaction A Reaction B. КОВ CH₂ HotBu +B+ ко HOIBU +Br+ Templates More QQQ Select Cv Templates More Cras QQQ One of these compounds undergoes elimination 50x faster than the other. Which one and why? Reaction A because the conformation needed for elimination places the phenyl groups and to each other Reaction A because the conformation needed for elimination places the phenyl groups gauche to each other. ◇ Reaction B because the conformation needed for elimination places the phenyl groups gach to each other. Reaction B because the conformation needed for elimination places the phenyl groups anti to each other.arrow_forwardFive isomeric alkenes. A through each undergo catalytic hydrogenation to give 2-methylpentane The IR spectra of these five alkenes have the key absorptions (in cm Compound Compound A –912. (§), 994 (5), 1643 (%), 3077 (1) Compound B 833 (3), 1667 (W), 3050 (weak shoulder on C-Habsorption) Compound C Compound D) –714 (5), 1665 (w), 3010 (m) 885 (3), 1650 (m), 3086 (m) 967 (5), no aharption 1600 to 1700, 3040 (m) Compound K Match each compound to the data presented. Compound A Compound B Compound C Compound D Compoundarrow_forward7. The three sets of replicate results below were accumulated for the analysis of the same sample. Pool these data to obtain the most efficient estimate of the mean analyte content and the standard deviation. Lead content/ppm: Set 1 Set 2 Set 3 1. 9.76 9.87 9.85 2. 9.42 9.64 9.91 3. 9.53 9.71 9.42 9.81 9.49arrow_forward
- Draw the Zaitsev product famed when 2,3-dimethylpentan-3-of undergoes an El dehydration. CH₂ E1 OH H₁PO₁ Select Draw Templates More QQQ +H₂Oarrow_forwardComplete the clean-pushing mechanism for the given ether synthesia from propanol in concentrated sulfurica140°C by adding any mining aloms, bands, charges, nonbonding electron pairs, and curved arrows. Draw hydrogen bonded to cayan, when applicable. ore 11,0 HPC Step 1: Draw curved arrows Step 2: Complete the intend carved Q2Q 56 QQQ Step 3: Complete the intermediate and add curved Step 4: Modify the structures to draw the QQQ QQQarrow_forward6. In an experiment the following replicate set of volume measurements (cm3) was recorded: (25.35, 25.80, 25.28, 25.50, 25.45, 25.43) A. Calculate the mean of the raw data. B. Using the rejection quotient (Q-test) reject any questionable results. C. Recalculate the mean and compare it with the value obtained in 2(a).arrow_forward
- A student proposes the transformation below in one step of an organic synthesis. There may be one or more reactants missing from the left-hand side, but there are no products missing from the right-hand side. There may also be catalysts, small inorganic reagents, and other important reaction conditions missing from the arrow. • Is the student's transformation possible? If not, check the box under the drawing area. • If the student's transformation is possible, then complete the reaction by adding any missing reactants to the left-hand side, and adding required catalysts, inorganic reagents, or other important reaction conditions above and below the arrow. • You do not need to balance the reaction, but be sure every important organic reactant or product is shown. + T G OH де OH This transformation can't be done in one step.arrow_forwardMacmillan Leaming Draw the major organic product of the reaction. 1. CH3CH2MgBr 2. H+ - G Select Draw Templates More H о QQarrow_forwardDraw the condensed structure of 3-hydroxy-2-butanone. Click anywhere to draw the first atom of your structure.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

