
Student's Study Guide and Solutions Manual for Organic Chemistry
8th Edition
ISBN: 9780134066585
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 109P
The experiment shown next and discussed in Section 8.13 shows that the proximity of the chloride ion to C-2 in the transition state causes the 1,2·addition product to form more rapidly than the 1,4-addition product.
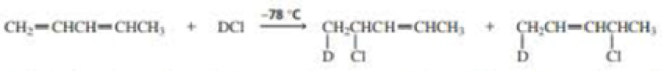
- a. Why was it important for the investigators to know that the preceding reaction was being carried out under kinetic control?
- b. How could the investigators know that the reaction was being carried out under kinetic control?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Don't used Ai solution
Don't used Ai solution
Please correct answer and don't used hand raiting
Chapter 8 Solutions
Student's Study Guide and Solutions Manual for Organic Chemistry
Ch. 8.1 - Prob. 1PCh. 8.1 - Prob. 2PCh. 8.4 - Prob. 3PCh. 8.5 - Prob. 4PCh. 8.5 - Prob. 6PCh. 8.6 - a. Predict the relative bond lengths of the three...Ch. 8.6 - Prob. 8PCh. 8.6 - Prob. 9PCh. 8.6 - Prob. 10PCh. 8.7 - Prob. 11P
Ch. 8.7 - Prob. 12PCh. 8.7 - Prob. 13PCh. 8.8 - Prob. 14PCh. 8.8 - Prob. 15PCh. 8.8 - Prob. 16PCh. 8.9 - Which member of each pair is the stronger acid?Ch. 8.9 - Which member of each pair is the stronger base? a....Ch. 8.9 - Rank the following compounds from strongest acid...Ch. 8.10 - Prob. 20PCh. 8.10 - Which acid in each of the following pairs is...Ch. 8.10 - Prob. 23PCh. 8.11 - Prob. 24PCh. 8.11 - Prob. 26PCh. 8.12 - Prob. 27PCh. 8.12 - Prob. 28PCh. 8.12 - Prob. 29PCh. 8.12 - Prob. 30PCh. 8.12 - Prob. 31PCh. 8.12 - Prob. 32PCh. 8.13 - Prob. 33PCh. 8.13 - Prob. 34PCh. 8.13 - Prob. 35PCh. 8.13 - What are the major 1,2- and 1,4-addition products...Ch. 8.13 - Prob. 38PCh. 8.14 - Prob. 39PCh. 8.14 - Prob. 40PCh. 8.14 - Prob. 41PCh. 8.14 - Prob. 42PCh. 8.14 - Prob. 43PCh. 8.14 - Prob. 44PCh. 8.14 - Prob. 46PCh. 8.15 - Prob. 47PCh. 8.17 - Prob. 48PCh. 8.17 - Prob. 49PCh. 8.18 - Prob. 50PCh. 8.18 - Prob. 52PCh. 8.18 - Prob. 53PCh. 8.18 - Prob. 54PCh. 8.19 - Prob. 55PCh. 8.20 - Prob. 56PCh. 8.20 - What orbitals contain the electrons represented as...Ch. 8.20 - Prob. 59PCh. 8.20 - Prob. 60PCh. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - Prob. 64PCh. 8 - Prob. 65PCh. 8 - Prob. 66PCh. 8 - Prob. 67PCh. 8 - Prob. 68PCh. 8 - Prob. 69PCh. 8 - Prob. 70PCh. 8 - Prob. 71PCh. 8 - Prob. 72PCh. 8 - Prob. 73PCh. 8 - Which compound is the strongest base?Ch. 8 - Prob. 75PCh. 8 - Prob. 76PCh. 8 - a. The A ring (Section 3.16) of cortisone (a...Ch. 8 - Prob. 78PCh. 8 - Prob. 79PCh. 8 - Prob. 80PCh. 8 - Prob. 81PCh. 8 - Purine is a heterocyclic compound with four...Ch. 8 - Prob. 83PCh. 8 - Why is the delocalization energy of pyrrole (21...Ch. 8 - Prob. 85PCh. 8 - Prob. 86PCh. 8 - Prob. 87PCh. 8 - A student obtained two products from the reaction...Ch. 8 - Prob. 89PCh. 8 - a. How could each of the following compounds be...Ch. 8 - Draw the products obtained from the reaction of...Ch. 8 - How would the following substituents affect the...Ch. 8 - Prob. 93PCh. 8 - The acid dissociation constant (Ka) for loss of a...Ch. 8 - Protonated cyclohexylamine has a Ka = 1 1011...Ch. 8 - Draw the product or products that would be...Ch. 8 - Prob. 97PCh. 8 - Prob. 98PCh. 8 - Prob. 99PCh. 8 - Prob. 100PCh. 8 - Prob. 101PCh. 8 - a. Propose n mechanism for the following reaction:...Ch. 8 - Prob. 103PCh. 8 - As many as 18 different Diels-Alder products can...Ch. 8 - Prob. 105PCh. 8 - Prob. 106PCh. 8 - Prob. 107PCh. 8 - Prob. 108PCh. 8 - The experiment shown next and discussed in Section...Ch. 8 - Prob. 110PCh. 8 - Prob. 111PCh. 8 - Prob. 112PCh. 8 - Prob. 1PCh. 8 - Prob. 2PCh. 8 - Prob. 3PCh. 8 - Prob. 4PCh. 8 - Prob. 5PCh. 8 - Prob. 6PCh. 8 - Prob. 7PCh. 8 - Prob. 8PCh. 8 - Prob. 9PCh. 8 - Prob. 10PCh. 8 - Prob. 11PCh. 8 - Prob. 12P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (12) Which one of the following statements about fluo- rometry is FALSE? a) Fluorescence is better detected at 90 from the exci- tation direction. b) Fluorescence is typically shifted to longer wave- length from the excitation wavelength. c) For most fluorescent compounds, radiation is pro- duced by a transitionarrow_forwardDon't used Ai solutionarrow_forwardDon't used Ai solutionarrow_forward
- Don't used Ai solutionarrow_forwardIndicate the correct option.a) Graphite conducts electricity, being an isotropic materialb) Graphite is not a conductor of electricityc) Both are falsearrow_forward(f) SO: Best Lewis Structure 3 e group geometry:_ shape/molecular geometry:, (g) CF2CF2 Best Lewis Structure polarity: e group arrangement:_ shape/molecular geometry: (h) (NH4)2SO4 Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forward
- 1. Problem Set 3b Chem 141 For each of the following compounds draw the BEST Lewis Structure then sketch the molecule (showing bond angles). Identify (i) electron group geometry (ii) shape around EACH central atom (iii) whether the molecule is polar or non-polar (iv) (a) SeF4 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: (b) AsOBr3 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles):arrow_forward(c) SOCI Best Lewis Structure 2 e group arrangement: shape/molecular geometry:_ (d) PCls Best Lewis Structure polarity: e group geometry:_ shape/molecular geometry:_ (e) Ba(BrO2): Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forwardDon't used Ai solutionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License