
Bundle: Organic Chemistry, 9th, Loose-Leaf + OWLv2, 4 terms (24 months) Printed Access Card
9th Edition
ISBN: 9781305701021
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7.SE, Problem 29MP
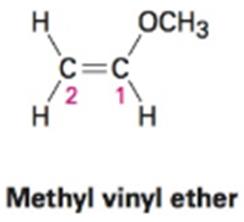
When methyl vinyl ether reacts with a strong acid, the proton adds to C2 exclusively, instead of C1 or the oxygen atom. Draw the three protonated forms of methyl vinyl ether and explain this observation.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
I need help solving my HW
i need help solving my homework
Can you please help me solve these homework questions
Chapter 7 Solutions
Bundle: Organic Chemistry, 9th, Loose-Leaf + OWLv2, 4 terms (24 months) Printed Access Card
Ch. 7.2 - Calculate the degree of unsaturation in each of...Ch. 7.2 - Prob. 2PCh. 7.2 - Diazepam, marketed as an antianxiety medication...Ch. 7.3 - Give IUPAC names for the following compounds:Ch. 7.3 - Prob. 5PCh. 7.3 - Prob. 6PCh. 7.3 - Prob. 7PCh. 7.4 - Prob. 8PCh. 7.4 - Prob. 9PCh. 7.4 - Prob. 10P
Ch. 7.5 - Which member in each of the following sets ranks...Ch. 7.5 - Prob. 12PCh. 7.5 - Prob. 13PCh. 7.5 - Prob. 14PCh. 7.6 - Prob. 15PCh. 7.8 - Prob. 16PCh. 7.8 - Prob. 17PCh. 7.9 - Show the structures of the carbocation...Ch. 7.9 - Draw a skeletal structure of the following...Ch. 7.10 - Prob. 20PCh. 7.11 - On treatment with HBr, vinylcyclohexane undergoes...Ch. 7.SE - Prob. 22VCCh. 7.SE - Prob. 23VCCh. 7.SE - The following carbocation is an intermediate in...Ch. 7.SE - Prob. 25VCCh. 7.SE - Predict the major product and show the complete...Ch. 7.SE - Prob. 27MPCh. 7.SE - When 1, 3-butadiene reacts with one mole of HBr,...Ch. 7.SE - When methyl vinyl ether reacts with a strong acid,...Ch. 7.SE - Addition of HCl to 1-isopropylcyclohexene yields a...Ch. 7.SE - Addition of HCl to...Ch. 7.SE - Limonene, a fragrant hydrocarbon found in lemons...Ch. 7.SE - Prob. 33MPCh. 7.SE - Calculate the degree of unsaturation in the...Ch. 7.SE - Prob. 35APCh. 7.SE - Prob. 36APCh. 7.SE - Name the following alkenes:Ch. 7.SE - Draw structures corresponding to the following...Ch. 7.SE - Prob. 39APCh. 7.SE - Prob. 40APCh. 7.SE - Prob. 41APCh. 7.SE - Prob. 42APCh. 7.SE - Prob. 43APCh. 7.SE - Draw and name the 17 alkene isomers, C6H12,...Ch. 7.SE - Prob. 45APCh. 7.SE - Prob. 46APCh. 7.SE - Which of the following E, Z designations are...Ch. 7.SE - Prob. 48APCh. 7.SE - trans-2-Butene is more stable than cis-2-butene by...Ch. 7.SE - Prob. 50APCh. 7.SE - Normally, a trans alkene is more stable than its...Ch. 7.SE - trans-Cyclooctene is less stable than...Ch. 7.SE - Prob. 53APCh. 7.SE - Prob. 54APCh. 7.SE - Use Hammond’s Postulate to determine which...Ch. 7.SE - Prob. 56APCh. 7.SE - Predict the major product in each of the following...Ch. 7.SE - Prob. 58APCh. 7.SE - Prob. 59APCh. 7.SE - Prob. 60APCh. 7.SE - Allene (1,2-propadiene), H2C=C=CH2, has two...Ch. 7.SE - The heat of hydrogenation for allene (Problem...Ch. 7.SE - Retin A, or retinoic acid, is a medication...Ch. 7.SE - Prob. 64APCh. 7.SE - tert-Butyl esters [RC02C(CH3)3] are converted into...Ch. 7.SE - Vinylcyclopropane reacts with HBr to yield a...Ch. 7.SE - Prob. 67APCh. 7.SE - Prob. 68APCh. 7.SE - Prob. 69APCh. 7.SE - Prob. 70APCh. 7.SE - Prob. 71APCh. 7.SE - Reaction of 2, 3-dimethyl-l-butene with HBr leads...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- An einstein is the amount of energy needed to dissociate 1 mole of a substance. If we have 0.58 moles, do we need 0.58 einsteins to dissociate that substance?arrow_forwardIf the energy absorbed per mole of gas is 480 kJ mol-1, indicate the number of Einsteins per mole.Data: Energy of each photon: 0.7835x10-18 J.arrow_forwardIf the energy absorbed per mole of gas is 480 kJ mol-1, indicate the number of Einsteins per mole.arrow_forward
- The quantum yield of the photochemical decay of HI is 2. Calculating the moles of HI per kJ of radiant energy can be decayed knowing that the energy absorbed per mole of photons is 490 kJ.arrow_forwardThe quantum yield of the photochemical decay of HI is 2. Calculate the number of Einsteins absorbed per mole knowing that the energy absorbed per mole of photons is 490 kJ.arrow_forwardThe quantum yield of the photochemical decay of HI is 2. How many moles of HI per kJ of radiant energy can be decayed knowing that the energy absorbed per mole of photons is 490 kJ.arrow_forward
- If the energy absorbed per mole of photons is 450 kJ, the number of Einsteins absorbed per 1 mole.arrow_forwardWhen propionic aldehyde in vapor form at 200 mmHg and 30°C is irradiated with radiation of wavelength 302 nm, the quantum yield with respect to the formation of CO is 0.54. If the intensity of the incident radiation is 1.5x10-3 W, find the rate of formation of CO.arrow_forwardDraw mechanismarrow_forward
- Does Avogadro's number have units?arrow_forwardExplain why the total E in an Einstein depends on the frequency or wavelength of the light.arrow_forwardIf the dissociation energy of one mole of O2 is 5.17 eV, determine the wavelength that must be used to dissociate it with electromagnetic radiation. Indicate how many Einstein's of this radiation are needed to dissociate 1 liter of O2 at 25°C and 1 atm of pressure.Data: 1 eV = 96485 kJ mol-1; R = 0.082 atm L K-1; c = 2.998x108 m s-1; h = 6.626x10-34 J s; NA = 6.022x 1023 mol-1arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License