
Concept explainers
(a)
Interpretation:
The products of the given reaction have to be given.
Concept Introduction:
Addition of hydrogen halides to
Addition of hydrogen halides to alkenes is one of the electrophilic additions to yield the corresponding halo
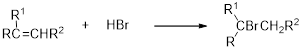
(b)
Interpretation:
The products of the given reaction have to be given.
Concept Introduction:
Addition of hydrogen halides to alkenes:
Addition of hydrogen halides to alkenes is one of the electrophilic addition to yield the corresponding halo alkanes. In this reaction the halide ion adds to the double bond carbon which is having lesser number of hydrogen or more substituted carbon (Markovnikov's rule).
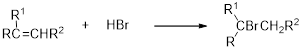
(c)
Interpretation:
The products of the given reaction have to be given.
Concept Introduction:
Addition of water across double bond:
Alcohols can be prepared from alkenes by heating with water in the presence of an acid catalyst and the process is called hydration of alkenes.
Across the double bond, water addition occurs.
(d)
Interpretation:
The products of the given reaction have to be given.
Concept Introduction:
Addition of hydrogen halides to alkenes:
Addition of hydrogen halides to alkenes is one of the electrophilic additions to yield the corresponding halo alkanes. In this reaction the halide ion adds to the double bond carbon which is having lesser number of hydrogen or more substituted carbon (Markovnikov's rule).
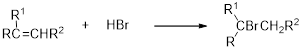
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
EBK ESSENTIAL ORGANIC CHEMISTRY
- A mixture of CaCO3 and MgC2O4 of unknown mass was heated in a 0.5 L closed rigid vessel to 900 degrees C.at 400C the following reaction occurs:MgC2O4 -> MgO (s) + CO (g) + CO2 (g)At 700C a second reaction occurs: CaCO3 -> CaO (s) + CO2 (g)The solid mass in the vessel was measured to be 3.06 g at 400C and 2.03g at 900CQuestion: What is the partial pressure of CO in both temperatures? (400 and 900C), provide detailed explanation.arrow_forwardFor the following alkyne, complete the reaction sequentially (that is draw the intermediate that we can’t stop at) and then name (complete name) all 3 molecules.arrow_forwardGiven the reaction sequence below, answer the following. A. Provide the structure for A. B. Provide the structure for B (pay attention to stereochemistry). C. Provide the structure for C. D. What are the stereochemical designations for I and II (R/S)?arrow_forward
- There are 2 reactions (that you know of) to achieve the following transformation: One reaction is favored over the other because it avoids a competing reaction. A. Draw the favored reaction scheme (not the mechanism), be sure to include all necessary reagents. B. Draw the reaction scheme that is not favored and include all the possible products.arrow_forwardBoth carbocations and carbon-radicals have trigonal planar geometry. True or Falsearrow_forwardTeflon (polytetrafluoroethene) is prepared via the radial polymerization of tetrafluoroethene. What other reaction conditions (reagent, etc.) are needed to accomplish this? A. NBS, Light B. Heat, Cl2 C. Peroxide, Heat D. H2SO4, H2O, Heatarrow_forward
- Which of the following compounds can be reacted with ethene to prepare 1,1- dichlorocyclopropane? A. CCl4 B. CCl2 C. CHCl3 D. CH2Cl2arrow_forwardusing any reactions, starting with propene, propose a synthesis for the following moleculearrow_forwardFor a free radical bromination reaction, the stereochemistry of the product is easily controlled True OR Falsearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





