
Bundle: Chemistry for Engineering Students, 3rd, Loose-Leaf + OWLv2 with QuickPrep 24-Months Printed Access Card
3rd Edition
ISBN: 9781305367388
Author: Lawrence S. Brown, Tom Holme
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 7, Problem 7.95PAE
Interpretation Introduction
Interpretation:
Explain how the geometry of carbon dioxide is responsible for the observation that carbon dioxide cannot be separated from air by applying voltage, even though there is a significant difference in electronegativities of carbon and oxygen atom.
Concept Introduction:
Carbon dioxide molecule is a non-polar molecule and the carbon dioxide molecule has linear geometry displayed as follows:
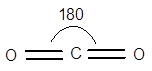
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a Haworth projection of a common cyclic form of this monosaccharide:
H
HO
H
HO
H
HO
H
H
-OH
CH2OH
Click and drag to start drawing a
structure.
Х
:
D
:
Draw the structure of valylasparagine, a dipeptide made from valine and asparagine, as it would appear at physiological pH.
Click and drag to start drawing a
structure.
P
D
Draw the Haworth projection of α-L-mannose. You will find helpful information in the ALEKS Data resource.
Click and drag to start drawing a
structure.
:
ཊི
Х
D
Chapter 7 Solutions
Bundle: Chemistry for Engineering Students, 3rd, Loose-Leaf + OWLv2 with QuickPrep 24-Months Printed Access Card
Ch. 7 - List some factors influencing the biocompatibility...Ch. 7 - • use electron configurations to explain why...Ch. 7 - • describe die energy changes in the formation of...Ch. 7 - • define electronegativity and state how...Ch. 7 - • identify or predict polar, nonpolar, and ionic...Ch. 7 - • write Lewis electron structures for molecules or...Ch. 7 - • describe chemical bonding using a model based on...Ch. 7 - • explain how hybridization reconciles observed...Ch. 7 - • predict the geometry of a molecule from its,...Ch. 7 - • use models (real or software) to help visualize...
Ch. 7 - • explain the formation of multiple bonds in terms...Ch. 7 - • identify sigma and pi bonds in a molecule and...Ch. 7 - Define the term biocompatibility.Ch. 7 - List some properties associated with biomaterials...Ch. 7 - Prob. 7.3PAECh. 7 - Prob. 7.4PAECh. 7 - Prob. 7.5PAECh. 7 - Prob. 7.6PAECh. 7 - Why is the ion not found in nature?Ch. 7 - Why do nonmetals tend to form anions rather than...Ch. 7 - Prob. 7.9PAECh. 7 - 7.10 Arrange the members of each of the following...Ch. 7 - 7.11 Arrange the following sets of anions in order...Ch. 7 - 7.12 Which pair will form a compound with the...Ch. 7 - In a lattice, a positive ion is often surrounded...Ch. 7 - Use the concept of lattice energy to rationalize...Ch. 7 - 7.13 Figure 7-2 depicts the interactions of an ion...Ch. 7 - Mat type of bond is likely to form between one...Ch. 7 - 7.14 Describe the difference between a covalent...Ch. 7 - Prob. 7.18PAECh. 7 - Sketch a graph of the potential energy of two...Ch. 7 - Prob. 7.20PAECh. 7 - 7.17 Coulombic forces are often used to explain...Ch. 7 - 7.18 In terms of the strengths of the covalent...Ch. 7 - 7.19 If the formation of chemical bonds always...Ch. 7 - 7.20 Draw the Lewis dot symbol for each of the...Ch. 7 - 7.21 Theoretical models for the structure of...Ch. 7 - 7.22 Use Lewis dot symbols to explain why chlorine...Ch. 7 - 7.23 Define the term lone pair.Ch. 7 - 7.24 How many electrons are shared between two...Ch. 7 - 7.25 How does the bond energy of a double bond...Ch. 7 - 7.26 How is electronegativity defined?Ch. 7 - 7.27 Distinguish between electron affinity and...Ch. 7 - 7.28 Certain elements in the periodic table shown...Ch. 7 - 7.29 When two atoms with different...Ch. 7 - 7.30 The bond in HF is said to be polar, with the...Ch. 7 - 7.31 Why is a bond between two atoms with...Ch. 7 - Prob. 7.36PAECh. 7 - 7.33 In each group of three bonds, which bond is...Ch. 7 - Prob. 7.38PAECh. 7 - Prob. 7.39PAECh. 7 - 7.35 Which one of the following contains botb...Ch. 7 - Prob. 7.41PAECh. 7 - Prob. 7.42PAECh. 7 - 7.37 Draw the Lewis structure for each of the...Ch. 7 - 7.38 Draw a Lewis structure for each of the...Ch. 7 - Prob. 7.45PAECh. 7 - 7.40 Why is it impossible for hydrogen to be the...Ch. 7 - Prob. 7.47PAECh. 7 - 7.42 Draw resonance structure for (a) (b) and (c)Ch. 7 - Prob. 7.49PAECh. 7 - Prob. 7.50PAECh. 7 - Prob. 7.51PAECh. 7 - 7.46 Consider the nitrogen-oxygen bond lengths in...Ch. 7 - 7.47 Which of the species listed has a Lewis...Ch. 7 - 7.48 Identify what is incorrect in the Lewis...Ch. 7 - 7.49 Identify what is incorrect in the Lewis...Ch. 7 - 7.50 Chemical species are said to be isoelectronic...Ch. 7 - 7.51 Explain the concept of wave interference in...Ch. 7 - Distinguish between constructive and destructive...Ch. 7 - How is the concept of orbital overlap related to...Ch. 7 - 7.52 How does orbital overlap explain the buildup...Ch. 7 - 7.53 How do sigma and pi bonds differ? How are...Ch. 7 - 7.54 CO , CO2 , CH3OH , and CO32 , all contain...Ch. 7 - 7.55 Draw the Lewis dot structure of the following...Ch. 7 - 7.56 Draw the Lewis dot structures of the...Ch. 7 - 7.57 What observation about molecules compels us...Ch. 7 - Prob. 7.66PAECh. 7 - 7.59 What type of hybrid orbital is generated by...Ch. 7 - Considering only s and p atomic orbitals, list all...Ch. 7 - 7.61 What hybrid orbitals would be expected for...Ch. 7 - 7.62 What type of hybridization would you expect...Ch. 7 - 7.63 What physical concept forms the premise of...Ch. 7 - 7.64 Predict the geometry of the following...Ch. 7 - Prob. 7.73PAECh. 7 - Prob. 7.74PAECh. 7 - Prob. 7.75PAECh. 7 - 7.68 Give approximate values for the indicated...Ch. 7 - 7.69 Propene has the chemical formula Describe the...Ch. 7 - Prob. 7.78PAECh. 7 - Describe what happens to the shape about the...Ch. 7 - Prob. 7.80PAECh. 7 - Prob. 7.81PAECh. 7 - 7.72 How does an MSN differ from amorphous silica...Ch. 7 - Prob. 7.83PAECh. 7 - Prob. 7.84PAECh. 7 - Prob. 7.85PAECh. 7 - Prob. 7.86PAECh. 7 - 7.91 A Lewis structure for the oxalate ion is...Ch. 7 - Prob. 7.88PAECh. 7 - 7.93 An unknown metal M forms a chloride with the...Ch. 7 - Prob. 7.90PAECh. 7 - Prob. 7.91PAECh. 7 - 7.96 Consider the hydrocarbons whose structures...Ch. 7 - 7.97 Consider the structure shown below for as...Ch. 7 - Prob. 7.94PAECh. 7 - Prob. 7.95PAECh. 7 - Prob. 7.96PAECh. 7 - 7.101 Lead selenide nanocrystals may provide a...Ch. 7 - Prob. 7.98PAECh. 7 - Prob. 7.99PAECh. 7 - 7.104 Hydrogen azide, HN3 , is a liquid that...Ch. 7 - Prob. 7.101PAECh. 7 - Prob. 7.102PAECh. 7 - 7.107 How do the Lewis symbols for C, Si, and Ge...Ch. 7 - Prob. 7.104PAECh. 7 - Prob. 7.105PAE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the structure of serine at pH 6.8. Click and drag to start drawing a structure. : d كarrow_forwardTake a look at this molecule, and then answer the questions in the table below it. CH2OH H H H OH OH OH CH2OH H H H H OH H H OH H OH Is this a reducing sugar? yes α β ロ→ロ no ☑ yes Does this molecule contain a glycosidic bond? If you said this molecule does contain a glycosidic bond, write the symbol describing it. O no 0+0 If you said this molecule does contain a glycosidic bond, write the common names (including anomer and enantiomer labels) of the molecules that would be released if that bond were hydrolyzed. If there's more than one molecule, separate each name with a comma. ☐arrow_forwardAnswer the questions in the table below about this molecule: H₂N-CH₂ -C—NH–CH–C—NH–CH—COO- CH3 CH CH3 What kind of molecule is this? 0= CH2 C If you said the molecule is a peptide, write a description of it using 3-letter codes separated ☐ by dashes. polysaccharide peptide amino acid phospolipid none of the above Хarrow_forward
- Draw a Haworth projection of a common cyclic form of this monosaccharide: CH₂OH C=O HO H H -OH H OH CH₂OH Click and drag to start drawing a structure. : ☐ Х S '☐arrow_forwardNucleophilic Aromatic Substitution 22.30 Predict all possible products formed from the following nucleophilic substitution reactions. (a) (b) 9 1. NaOH 2. HCI, H₂O CI NH₁(!) +NaNH, -33°C 1. NaOH 2. HCl, H₂Oarrow_forwardSyntheses 22.35 Show how to convert toluene to these compounds. (a) -CH,Br (b) Br- -CH3 22.36 Show how to prepare each compound from 1-phenyl-1-propanone. 1-Phenyl-1-propanone ہتی. Br. (b) Br (racemic) 22.37 Show how to convert ethyl benzene to (a) 2,5-dichlorobenzoic acid and (b) 2,4-dichlorobenzoic acid. 22.38 Show reagents and conditions to bring about the following conversions. (a) 9 NH2 8 CO₂H NH2 CO₂Et (d) NO2 NH2 S NH₂ NO2 CHS CHarrow_forward
- ive the major organic product(s) of each of the following reactions or sequences of reactions. Show all rant stereochemistry. [10 only] A. B. NaN3 1. LiAlH4, ether Br 2. H₂O CH3 HNO3 H₂/Pt H₂SO ethanol C. 0 0 CH3CC1 NaOH NHCCH AICI H₂O . NH₂ CH3CH2 N CH2CH3 + HCI CH₂CH 3 1. LIAIH, THE 2. H₂Oarrow_forwardCalculate the stoichiometric amount of CaCl2 needed to convert all of the CuSO4 into CuCl2.arrow_forwardH CH تنی Cl 1. NaCN, DMF 2. LIAIH4, ether H₂O pyridine N NH₂ 5 CH H 1 HNO, H₂SO 2. Nal NH2 Br Br HNO₂ CuCl H₂SO HCI CH3 H3C NN HSO KCN CuCN 1. HNO₂, H₂SO O₂N NH2 2. OH ཀ་ལས། །ས་ཅན་ :i་དེ་མ་མ་སེ་ NH₂ CH3 1. HNO₂, H₂SO4 2. H3PO₂ 1 HNO2, H2SO4 2. Nalarrow_forward
- ive the major organic product(s) of each of the following reactions or sequences of reactions. Show all rant stereochemistry. [10 only] A. B. NaN3 1. LiAlH4, ether Br 2. H₂O CH3 HNO3 H₂/Pt H₂SO ethanol C. 0 0 CH3CC1 NaOH NHCCH AICI H₂O . NH₂ CH3CH2 N CH2CH3 + HCI CH₂CH 3 1. LIAIH, THE 2. H₂Oarrow_forwardIf a pharmacy chain sold 65 million 500-mg tablets of aspirin, how many US tons of aspirin does this represent? Report your answer to 2 significant figures.arrow_forwardHere are the options: reducing a monosaccharide a non reducing disaccharide amylopectin cellulose 1,4' beta- glycosidearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY