
Concept explainers
Consider the following

a. Draw a mechanism using curved arrows.
b. Draw an energy diagram. Label the axes, the reactants, products,
c. Draw the structure of the transition state.
d. What is the rate equation?
e. What happens to the reaction rate in each of the following instances? [1] The leaving group is changed from
(a)
Interpretation: The mechanism of the given reaction is to be drawn by the use of curved arrows.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons are known as a nucleophile. In a nucleophilic substitution reaction, nucleophile takes the position of leaving group by attacking the electron deficient carbon atom.
Answer to Problem 7.53P
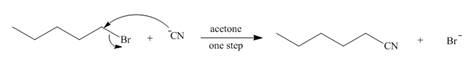
The mechanism of the given reaction is,
Explanation of Solution
The structure of the given alkyl halide shows that carbon atom, on which bromine is present, is bonded to one another carbon atom. Hence, the bromine atom is bonded to primary carbon atom and the given alkyl halide is
In

Figure 1
The mechanism of the given reaction is shown in Figure 1.
(b)
Interpretation: The energy diagram is to be drawn. The axes, reactants, products,
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The graphical representation of chemical reaction in which x-axis represents energy of the reaction and y-axis represents the reaction coordinate is called energy profile diagram.
Answer to Problem 7.53P
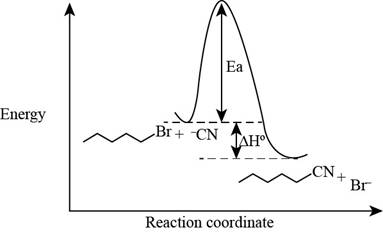
The energy diagram of the given reaction is shown below.
Explanation of Solution
In bimolecular nucleophilic substitution reaction, formation and breakage of bond takes place simultaneously in the transition state. In the given reaction, breakage of
The energy of products is lower than the energy of reactants in exothermic reaction. Therefore, the energy diagram of the given reaction is,
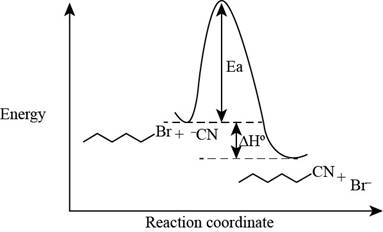
Figure 2
The x-axes of the energy diagram represent the energy of reactants and products and y-axes represent the reaction coordinate.
The energy diagram of the given reaction is shown in Figure 2.
(c)
Interpretation: The structure of the transition state is to be drawn.
Concept introduction: In bimolecular nucleophilic substitution reaction, formation and breakage of bond takes place simultaneously in the transition state. The energy of transition state is more than the energy of reactants and products.
Answer to Problem 7.53P
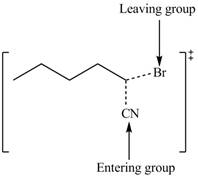
The structure of the transition state is shown below.
Explanation of Solution
In bimolecular nucleophilic substitution reaction, formation and breakage of bond takes place simultaneously in the transition state. In the given reaction, breakage of
Therefore, the transition state of the given reaction is,
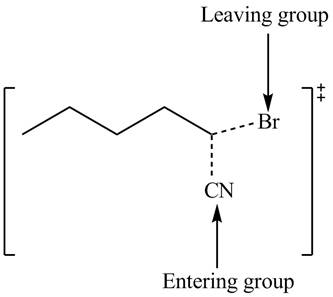
Figure 3
The structure of the transition state is shown in Figure 3.
(d)
Interpretation: The rate equation of the given reaction is to be predicted.
Concept introduction: The rate of
The rate equation of
Answer to Problem 7.53P
The rate equation of the given reaction is,
Explanation of Solution
The rate of
The rate equation of
The alkyl halide of given reaction is
Therefore, the rate equation of given reaction is,
The rate equation of the given reaction is,
(e)
Interpretation: The change that occurs to the reaction rate in given instances is to be stated.
Concept introduction: The rate of
The rate of
Answer to Problem 7.53P
The change that occurs to the reaction rate in given instances is,
[1] The rate of the reaction will increase.
[2] The rate of the reaction will decrease.
[3] The rate of the reaction will decrease.
[4] The rate of the reaction increases by five times.
[5] The rate of the reaction increases by twenty five times.
Explanation of Solution
[1]
The primary halide undergoes nucleophilic substitution by
[2]
The polar protic solvent favors
[3]
The primary halide undergoes nucleophilic substitution by
[4]
The rate of
The rate of
According the given statement, the concentration of
Therefore, the rate of the reaction increases by five times when the concentration of
[5]
The rate of
The rate of
According the given statement, the concentration of
Therefore, the rate of the reaction increases by twenty five times when the concentration
The change that occurs to the reaction rate in given instances is,
[1] The rate of the reaction will increase.
[2] The rate of the reaction will decrease.
[3] The rate of the reaction will decrease.
[4] The rate of the reaction increases by five times.
[5] The rate of the reaction increases by twenty five times.
Want to see more full solutions like this?
Chapter 7 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
Biology: Life on Earth with Physiology (11th Edition)
Microbiology Fundamentals: A Clinical Approach
General, Organic, and Biological Chemistry - 4th edition
Brock Biology of Microorganisms (15th Edition)
- What is the product of the reaction of XeF4 with H2O? Group of answer choices H2XeF2 H2XeF4 XeO3 H2XeOarrow_forwardWhile noble gas exerts the strongest London (dispersion) forces on neighboring atoms? Group of answer choices Xe Ar Kr Nearrow_forwardWhich of the following elements is corrosive to your skin due to that element breaking down C=C bonds? Group of answer choices fluorine iodine bromine chlorinearrow_forward
- What the best source of sulfide to use on a small scale in the lab? Group of answer choices thiourea H2S NaHS Na2Sarrow_forwardWhich of the following statements about sulfur is FALSE? Group of answer choices H2S is the product of an oxygen-depleted ecosystem. In the acid mine drainage reaction, FeS2 is a product. One allotrope of sulfur has the formula S20. In the environment, bacterial oxidation can convert S2− to elemental S or SO42−.arrow_forwardOf the following choices, which is the best reason that most materials DON'T spontaneously combust even though our atmosphere is about 21% oxygen? Group of answer choices The reduction of O2 in the gas phase (O2 + e− → O2−) is spontaneous. The reduction of O2 in acid solution (O2 + H+ + e− → HO2(aq)) is spontaneous. O2 is not a reactant in combustion. The O2 bond dissociation energy is 494 kJ/mol, leading to a high activation energy for combustion.arrow_forward
- please answer in the scope of the SCH4U course, I am having a hard time understanding, may you show all steps please and thank you! can you also put the final answers in the table so its understandablearrow_forwardPlan the synthesis of the following compound using the starting material provided and any other reagents needed as long as carbon based reagents have 3 carbons or less. Either the retrosynthesis or the forward synthesis (mechanisms are not required but will be graded if provided) will be accepted if all necessary reagents and intermediates are shown (solvents and temperature requirements are not needed unless specifically involved in the reaction, i.e. DMSO in the Swem oxidation or heat in the KMnO4 oxidation). There may be more than one correct answer, and chemically correct steps will be accepted. Extra points will be given if correct names are provided. The points earned here will be applied to your lowest exam score! H Harrow_forwardDraw the mechanism to make the alcohol 1-hexanol. Please use arrows.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
