
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
12th Edition
ISBN: 9781119304241
Author: Solomons
Publisher: WILEY C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 15PP
Practice Problem 7.15
Rank the following alcohols in order of increasing ease of acid-catalyzed dehydration.
(a)
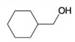
(b)
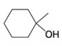
(c)
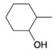
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Reagan is doing an atomic absorption experiment that requires a set of zinc standards in the 0.4-1.6 ppm range. A 1000 ppm Zn solution was prepared by dissolving the necessary amount of solid Zn(NO3)2 in water. The standards can be prepared by diluting the 1000 ppm Zn solution. Table 1 shows one possible set of serial dilutions (stepwise dilution of a solution) that Reagan could perform to make the necessary standards. Solution A was prepared by diluting 5.00 ml of the 1000 ppm Zn standard to 50.00 ml. Solutions C-E are called "calibration standards" because they will be used to calibrate the atomic absorption spectrometer.
a. Compare the solution concentrations expressed as ppm Zn and ppm Zn(NO3)2. Compare the concentrations expressed as M Zn and M Zn(NO3)2
- Which units allow easy conversion between chemical species (e.g. Zn and Zn(NO3)2)?
- Which units express concentrations in numbers with easily expressed magnitudes?
- Suppose you have an analyte for which you don't know the molar…
None
How will you prepare the following buffers? 2.5 L of 1.5M buffer, pH = 10.5 from NH4Cl and NH3
Chapter 7 Solutions
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
Ch. 7 - Prob. 1PPCh. 7 - Prob. 2PPCh. 7 - Prob. 3PPCh. 7 - Prob. 4PPCh. 7 - Practice Problem 7.5
How many stereoisomers are...Ch. 7 - Prob. 6PPCh. 7 - Prob. 7PPCh. 7 - PRACTICE PROBLEM 7.8
Examine Solved Problem 7.3....Ch. 7 - Prob. 9PPCh. 7 - Practice Problem 7.10 When...
Ch. 7 - Practice Problem 7.11
(a) When...Ch. 7 - Prob. 12PPCh. 7 - Prob. 13PPCh. 7 - Practice Problem 7.14
Dehydration of 2-propanol...Ch. 7 - Practice Problem 7.15
Rank the following alcohols...Ch. 7 - Practice Problem 7.16
Acid-catalyzed dehydration...Ch. 7 - Practice Problem 7.17 Acid-catalyzed dehydration...Ch. 7 - Prob. 18PPCh. 7 - Prob. 19PPCh. 7 - Practice Problem 7.20
Show how you might...Ch. 7 - Prob. 21PPCh. 7 - Prob. 22PPCh. 7 - Practice Problem 7.23
Write the structure of...Ch. 7 - Prob. 24PPCh. 7 - Prob. 25PPCh. 7 - Practice Problem 7.26 (a) Devise retrosynthetic...Ch. 7 - Each of the following names is incorrect, Give the...Ch. 7 - Prob. 28PCh. 7 - Prob. 29PCh. 7 - Give the IUPAC names for each of the following:...Ch. 7 - Prob. 31PCh. 7 - Prob. 32PCh. 7 - Prob. 33PCh. 7 - Prob. 34PCh. 7 - 7.35. Write structural formulas for all the...Ch. 7 - 7.36. Explain the following observations: When...Ch. 7 - Prob. 37PCh. 7 - Arrange the following alcohols in order of their...Ch. 7 - Prob. 39PPCh. 7 - Prob. 40PPCh. 7 - Prob. 41PPCh. 7 - Prob. 42PPCh. 7 - Your task is to prepare isopropyl methyl ether by...Ch. 7 - Prob. 44PCh. 7 - Prob. 45PCh. 7 - Prob. 46PCh. 7 - 7.47. Starting with an appropriate alkyl halide...Ch. 7 - Prob. 48PCh. 7 - 7.49. What is the index of hydrogen deficiency...Ch. 7 - Prob. 50PCh. 7 - Prob. 51PCh. 7 - Compounds I and J both have the molecular formula...Ch. 7 - Prob. 53PCh. 7 - 7.54. Outline a synthesis of phenylethyne from...Ch. 7 - Prob. 55PPCh. 7 - Prob. 56PPCh. 7 - Prob. 57PPCh. 7 - cis-4-Bromocyclohexanol tBuOHtBuO racemic C6H10O...Ch. 7 - Prob. 59PPCh. 7 - Consider the interconversion of cis-2-butene and...Ch. 7 - Prob. 61PCh. 7 - (a) Using reactions studied in this chapter, show...Ch. 7 - Prob. 63PCh. 7 - Prob. 64PCh. 7 - 1. Write the structure(s) of the major product(s)...Ch. 7 - Prob. 2LGPCh. 7 - (a) Write the structure of the product(s) formed...Ch. 7 - Prob. 4LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
All of the following processes are involved in the carbon cycle except: a. photosynthesis b. cell respiration c...
Human Biology: Concepts and Current Issues (8th Edition)
17.16 Use information from Appendix D to calculate the pH of
a solution that is 0.250 M in sodium formate (HCO...
Chemistry: The Central Science (14th Edition)
49. Write full electron configuration for each element.
a. Sr
b. Ge
c. Li
d. Kr
Introductory Chemistry (6th Edition)
1.2 Ask two of your friends (not in class) to define the terms in problem1.1.
Do their answers agee with the d...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
An aluminum calorimeter with a mass of 100 g contains 250 g of water. The calorimeter and water are in thermal ...
Physics for Scientists and Engineers
To four significant figures, find the following when the kinetic energy is 10.00 MeV: a and b for an electron...
Fundamentals of Physics Extended
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- How will you prepare the following buffers? 2.5 L of 1.5M buffer, pH = 10.5 from NH4Cl and NH3arrow_forwardCH₂O and 22 NMR Solvent: CDCl3 IR Solvent: neat 4000 3000 2000 1500 1000 15 [ اند 6,5 9.8 3.0 7.0 6.0 5.0 4.8 3.0 2.0 1.0 9.8 200 100arrow_forwardprotons. Calculate the mass (in grams) of H3AsO4 (MW=141.9416) needed to produce 3.125 x 1026arrow_forward
- Using what we have learned in CHEM 2310 and up through class on 1/31, propose a series of reaction steps to achieve the transformation below. Be sure to show all reagents and intermediates for full credit. You do not need to draw mechanism arrows, but you do need to include charges where appropriate. If you do not put your group name, you will get half credit at most. ? Brarrow_forwardDraw a mechanism for the formation of 2-bromovanillin using bromonium ion as the reactive electrophile.arrow_forwardNonearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY