
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
12th Edition
ISBN: 9780321908445
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.6, Problem 6.58QAP
For each of the following bonds, indicate the positive end with 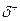 and the negative end with .
and the negative end with . 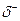 Draw an arrow to show the dipole for each.
Draw an arrow to show the dipole for each.
a.P and CI
b. Se and F
c. Br and F
d. N and H
e. B and Cl
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Drawing Arrows
1
I
I
1
heat
1
51
MO
+
Drag To
Und Settings
Done
0 0
Jan 31
3:5
Don't used hand raiting
Gramicidin A can adopt more than one structure; NMR spectroscopy has revealed an “end-to-end” dimer form, and x-ray crystallography has revealed an “anti-parallel double- helical” form. Briefly outline and describe an experimentalapproach/strategy to investigate WHICH configuration (“end-to-end dimer” vs “anti-paralleldouble helical”) gramicidin adopts in an actual lipid bilayer.
Chapter 6 Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Ch. 6.1 - State the number of electrons that be must be lost...Ch. 6.1 - State the number of electrons that must be gained...Ch. 6.1 - State the number of electrons lost or gained when...Ch. 6.1 - Prob. 6.4QAPCh. 6.1 - Write the symbols for the ions with the following...Ch. 6.1 - Write the symbols for the ions with the following...Ch. 6.1 - Write the symbol for the ion of each of the...Ch. 6.1 - Write the symbol for the ion of each of the...Ch. 6.2 - Which of the following pairs of elements are...Ch. 6.2 - Which of the following pairs of elements are...
Ch. 6.2 - Write the correct ionic formula for the compound...Ch. 6.2 - Write the correct ionic formula for the compound...Ch. 6.2 - Write the symbols for the ions, and the correct...Ch. 6.2 - Write the symbols for the ions, and the correct...Ch. 6.3 - Prob. 6.15QAPCh. 6.3 - Prob. 6.16QAPCh. 6.3 - Write the name for each of the following ions...Ch. 6.3 - Prob. 6.18QAPCh. 6.3 - Write the name for each of the following ionic...Ch. 6.3 - Prob. 6.20QAPCh. 6.3 - Prob. 6.21QAPCh. 6.3 - Prob. 6.22QAPCh. 6.3 - Prob. 6.23QAPCh. 6.3 - Write the formula for each of the following ionic...Ch. 6.3 - Prob. 6.25QAPCh. 6.3 - Prob. 6.26QAPCh. 6.4 - Write the formula including the charge for each of...Ch. 6.4 - Prob. 6.28QAPCh. 6.4 - Prob. 6.29QAPCh. 6.4 - Prob. 6.30QAPCh. 6.4 - Prob. 6.31QAPCh. 6.4 - Prob. 6.32QAPCh. 6.4 - Write the correct formula for the following ionic...Ch. 6.4 - Write the correct formula for the following ionic...Ch. 6.4 - Prob. 6.35QAPCh. 6.4 - Prob. 6.36QAPCh. 6.5 - Prob. 6.37QAPCh. 6.5 - Prob. 6.38QAPCh. 6.5 - Prob. 6.39QAPCh. 6.5 - Prob. 6.40QAPCh. 6.5 - Name each of the following molecular compounds:...Ch. 6.5 - Prob. 6.42QAPCh. 6.5 - Prob. 6.43QAPCh. 6.5 - Name each of the following molecular compounds: a....Ch. 6.5 - Write the formula for each of the following...Ch. 6.5 - Write the formula for each of the following...Ch. 6.5 - Write the formula for each of the following...Ch. 6.5 - Write the formula for each of the following...Ch. 6.5 - Prob. 6.49QAPCh. 6.5 - Prob. 6.50QAPCh. 6.6 - Describe the trend in electronegativity as...Ch. 6.6 - Prob. 6.52QAPCh. 6.6 - Using the periodic table, arrange the atoms in...Ch. 6.6 - Using the periodic table, arrange the atoms in...Ch. 6.6 - Predict whether each of the following bonds is...Ch. 6.6 - Predict whether each of the following bonds is...Ch. 6.6 - For each of the following bonds, indicate the...Ch. 6.6 - For each of the following bonds, indicate the...Ch. 6.7 - Choose the shape (1 to 6) that matches each of the...Ch. 6.7 - Prob. 6.60QAPCh. 6.7 - Prob. 6.61QAPCh. 6.7 - Complete each of the following statements for a...Ch. 6.7 - Prob. 6.63QAPCh. 6.7 - Prob. 6.64QAPCh. 6.7 - Prob. 6.65QAPCh. 6.7 - Prob. 6.66QAPCh. 6.7 - Prob. 6.67QAPCh. 6.7 - Prob. 6.68QAPCh. 6.7 - Prob. 6.69QAPCh. 6.7 - Prob. 6.70QAPCh. 6.8 - Prob. 6.71QAPCh. 6.8 - Prob. 6.72QAPCh. 6.8 - Prob. 6.73QAPCh. 6.8 - Prob. 6.74QAPCh. 6 - Prob. 6.75UTCCh. 6 - a. How does the octet rule explain the formation...Ch. 6 - Prob. 6.77UTCCh. 6 - Prob. 6.78UTCCh. 6 - Prob. 6.79UTCCh. 6 - Prob. 6.80UTCCh. 6 - Prob. 6.81UTCCh. 6 - Prob. 6.82UTCCh. 6 - Prob. 6.83UTCCh. 6 - Prob. 6.84UTCCh. 6 - Prob. 6.85UTCCh. 6 - 6.102 State the number of valence electrons,...Ch. 6 - Prob. 6.87AQAPCh. 6 - Prob. 6.88AQAPCh. 6 - Prob. 6.89AQAPCh. 6 - Prob. 6.90AQAPCh. 6 - Prob. 6.91AQAPCh. 6 - Prob. 6.92AQAPCh. 6 - Prob. 6.93AQAPCh. 6 - Prob. 6.94AQAPCh. 6 - Prob. 6.95AQAPCh. 6 - Prob. 6.96AQAPCh. 6 - Prob. 6.97AQAPCh. 6 - 6.120 Write the formula for each of the following...Ch. 6 - Prob. 6.99AQAPCh. 6 - Prob. 6.100AQAPCh. 6 - Prob. 6.101AQAPCh. 6 - Prob. 6.102AQAPCh. 6 - Prob. 6.103AQAPCh. 6 - Prob. 6.104AQAPCh. 6 - Prob. 6.105AQAPCh. 6 - Prob. 6.106AQAPCh. 6 - Prob. 6.107AQAPCh. 6 - Prob. 6.108AQAPCh. 6 - Prob. 6.109AQAPCh. 6 - Prob. 6.110AQAPCh. 6 - Prob. 6.111AQAPCh. 6 - Prob. 6.112AQAPCh. 6 - Prob. 6.113AQAPCh. 6 - Prob. 6.114AQAPCh. 6 - Prob. 6.115CQCh. 6 - Prob. 6.116CQCh. 6 - Prob. 6.117CQCh. 6 - Prob. 6.118CQCh. 6 - Prob. 6.119CQCh. 6 - Prob. 6.120CQCh. 6 - Prob. 7CICh. 6 - Prob. 8CICh. 6 - Prob. 9CICh. 6 - Prob. 10CICh. 6 - Prob. 11CICh. 6 - Of much concern to environmentalists is radon-222,...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Don't used hand raitingarrow_forwardCHEM2323 Problem 2-24 Tt O e: ל Predict the product(s) of the following acid/base reactions. Draw curved arrows to show the formation and breaking of bonds. If the bonds needed are not drawn out, you should redraw them. + BF3 (a) (b) HI + (c) OH -BF Problem 2-25 Use curved arrows and a proton (H+) to draw the protonated form of the following Lewis bases. Before starting, add all missing lone pairs. (a) (b) :0: (c) N 1 CHEM2323 PS CH02 Name:arrow_forwardCHEM2323 Problem 2-26 Tt O PS CH02 Name: Use the curved-arrow formalism to show how the electrons flow in the resonance form on the left to give the one on the right. (Draw all lone pairs first) (a) NH2 NH2 + (b) Problem 2-27 Double bonds can also act like Lewis bases, sharing their electrons with Lewis acids. Use curved arrows to show how each of the following double bonds will react with H-Cl and draw the resulting carbocation. (a) H2C=CH2 (b) (c) Problem 2-28 Identify the most electronegative element in each of the following molecules: (a) CH2FCI F Problem 2-29 (b) FCH2CH2CH2Br (c) HOCH2CH2NH2 (d) CH3OCH2Li F 0 0 Use the electronegativity table in Figure 2.3 to predict which bond in the following pairs is more polar and indicate the direction of bond polarity for each compound. (a) H3C-Cl or Cl-CI (b) H3C-H or H-CI (c) HO-CH3 or (CH3)3Si-CH3 (d) H3C-Li or Li-OHarrow_forward
- Reagan is doing an atomic absorption experiment that requires a set of zinc standards in the 0.4-1.6 ppm range. A 1000 ppm Zn solution was prepared by dissolving the necessary amount of solid Zn(NO3)2 in water. The standards can be prepared by diluting the 1000 ppm Zn solution. Table 1 shows one possible set of serial dilutions (stepwise dilution of a solution) that Reagan could perform to make the necessary standards. Solution A was prepared by diluting 5.00 ml of the 1000 ppm Zn standard to 50.00 ml. Solutions C-E are called "calibration standards" because they will be used to calibrate the atomic absorption spectrometer. a. Compare the solution concentrations expressed as ppm Zn and ppm Zn(NO3)2. Compare the concentrations expressed as M Zn and M Zn(NO3)2 - Which units allow easy conversion between chemical species (e.g. Zn and Zn(NO3)2)? - Which units express concentrations in numbers with easily expressed magnitudes? - Suppose you have an analyte for which you don't know the molar…arrow_forwardNonearrow_forwardHow will you prepare the following buffers? 2.5 L of 1.5M buffer, pH = 10.5 from NH4Cl and NH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY