
Chemistry the Central Science 13th Edition Custom for Lamar University
13th Edition
ISBN: 9781269962667
Author: Theodore L. Brown, H. Eugene LeMay Jr., Bruce E. Bursten, Batherine J. Murphy, Patrick M. Woodward, Matthew W. Stoltzfus
Publisher: Pearson Learning Center
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 5, Problem 81E
(b)
Interpretation Introduction
To determine:
The greater source of energy, as food, among 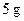 of fat and
of fat and 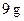 of carbohydrate.
of carbohydrate.
(c)
Interpretation Introduction
To determine:
An explanation on how the human body expels the reaction products of
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Help fix my arrows please
no Ai walkthroughs
Given the data attached, provide a drawing of the corresponding structure.
Chapter 5 Solutions
Chemistry the Central Science 13th Edition Custom for Lamar University
Ch. 5.2 - Prob. 5.1.1PECh. 5.2 - Prob. 5.1.2PECh. 5.3 - Prob. 5.2.1PECh. 5.3 - Prob. 5.2.2PECh. 5.3 - Prob. 5.3.1PECh. 5.3 - Prob. 5.3.2PECh. 5.4 - Prob. 5.4.1PECh. 5.4 - Prob. 5.4.2PECh. 5.5 - Prob. 5.5.1PECh. 5.5 - Prob. 5.5.2PE
Ch. 5.5 - Practice Exercise 1 Suppose you have equal masses...Ch. 5.5 - Prob. 5.6.2PECh. 5.5 - Prob. 5.7.1PECh. 5.5 - Prob. 5.7.2PECh. 5.6 - Prob. 5.8.1PECh. 5.6 - Prob. 5.8.2PECh. 5.6 - Prob. 5.9.1PECh. 5.6 - Prob. 5.9.2PECh. 5.7 - Prob. 5.10.1PECh. 5.7 - Prob. 5.10.2PECh. 5.7 - Prob. 5.11.1PECh. 5.7 - Prob. 5.11.2PECh. 5.7 - Prob. 5.12.1PECh. 5.7 - Practice Exercise 2 Use Table 5.3 to calculate the...Ch. 5.8 - Prob. 5.13.1PECh. 5.8 - Practice Exercise 2 Given the following standard...Ch. 5.8 - Prob. 5.14.1PECh. 5.8 - Prob. 5.14.2PECh. 5 - Prob. 1DECh. 5 - Prob. 1ECh. 5 - Prob. 2ECh. 5 - Prob. 3ECh. 5 - Practice Exercise 2
Using Table 20.1, rank...Ch. 5 - Prob. 5ECh. 5 - Prob. 6ECh. 5 - Prob. 7ECh. 5 - Prob. 8ECh. 5 - Prob. 9ECh. 5 - Prob. 10ECh. 5 - Prob. 11ECh. 5 - Prob. 12ECh. 5 - Prob. 13ECh. 5 - Prob. 14ECh. 5 - Prob. 15ECh. 5 - Prob. 16ECh. 5 - Prob. 17ECh. 5 - Prob. 18ECh. 5 - Prob. 19ECh. 5 - Prob. 20ECh. 5 - Prob. 21ECh. 5 - Prob. 22ECh. 5 - Prob. 23ECh. 5 - Prob. 24ECh. 5 - Prob. 25ECh. 5 - Prob. 26ECh. 5 - Prob. 27ECh. 5 - In chemical kinetics, the entropy of activation is...Ch. 5 - Prob. 29ECh. 5 - Prob. 30ECh. 5 - Prob. 31ECh. 5 - The following data compare the standard enthalpies...Ch. 5 - Prob. 33ECh. 5 - Prob. 34ECh. 5 - Prob. 35ECh. 5 - What is the reducing agent in the following...Ch. 5 - Prob. 37ECh. 5 - Prob. 38ECh. 5 - Prob. 39ECh. 5 - Prob. 40ECh. 5 - Prob. 41ECh. 5 - Prob. 42ECh. 5 - Prob. 43ECh. 5 - The standard cell potential is 1.46 V for a...Ch. 5 - Prob. 45ECh. 5 - Prob. 46ECh. 5 - Prob. 47ECh. 5 - Prob. 48ECh. 5 - Prob. 49ECh. 5 - Practice Exercise 1
Which of the following...Ch. 5 - Prob. 51ECh. 5 - Prob. 52ECh. 5 - Prob. 53ECh. 5 - Prob. 54ECh. 5 - Prob. 55ECh. 5 - Prob. 56ECh. 5 - Prob. 57ECh. 5 - Prob. 58ECh. 5 - Prob. 59ECh. 5 - Prob. 60ECh. 5 - What is the connection between Hess’s law and the...Ch. 5 - Prob. 62ECh. 5 - 20.2 You may have heard that “antioxidants” are...Ch. 5 - Prob. 64ECh. 5 - Prob. 65ECh. 5 - Prob. 66ECh. 5 - Prob. 67ECh. 5 - Prob. 68ECh. 5 - Prob. 69ECh. 5 - Prob. 70ECh. 5 - Prob. 71ECh. 5 - Prob. 72ECh. 5 - 20.13
What is meant by the term oxidation?
On...Ch. 5 - Prob. 74ECh. 5 - Prob. 75ECh. 5 - Prob. 76ECh. 5 - Prob. 77ECh. 5 - Prob. 78ECh. 5 - Prob. 79ECh. 5 - Prob. 80ECh. 5 - Prob. 81ECh. 5 - Prob. 82ECh. 5 - Prob. 83ECh. 5 - Prob. 84ECh. 5 - Prob. 85ECh. 5 - The heat of combustion of ethanol, C2H5OH(l) is...Ch. 5 - Prob. 87ECh. 5 - Prob. 88ECh. 5 - Prob. 89ECh. 5 - The automobile fuel called E85 consists of 85%...Ch. 5 - Prob. 91AECh. 5 - Prob. 92AECh. 5 - Prob. 93AECh. 5 - Prob. 94AECh. 5 - 5.95 Consider a system consisting of the following...Ch. 5 - A sample of gas is contained in a...Ch. 5 - Prob. 97AECh. 5 - Prob. 98AECh. 5 - A house is designed to have passive solar energy...Ch. 5 - Prob. 100AECh. 5 - Prob. 101AECh. 5 - Prob. 102AECh. 5 - Burning methane in oxygen can produce three...Ch. 5 - Prob. 104AECh. 5 - Prob. 105AECh. 5 - The hydrocarbons acetylene (C2H2) and benzene...Ch. 5 - Prob. 107AECh. 5 - Three common hydrocarbons that contain four...Ch. 5 - Prob. 109AECh. 5 - The Sun supplies about 1.0 kilowatt of energy for...Ch. 5 - It is estimated that the net amount of carbon...Ch. 5 - Prob. 112IECh. 5 - Prob. 113IECh. 5 - Prob. 114IECh. 5 - Prob. 115IECh. 5 - Prob. 116IECh. 5 - Prob. 117IECh. 5 - The methane molecule, CH4, has the geometry shown...Ch. 5 - Prob. 119IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- help 20arrow_forwardProvide the drawing of the unknown structure that corresponds with this data.arrow_forward20.44 The Diels-Alder reaction is not limited to making six-membered rings with only car- bon atoms. Predict the products of the following reactions that produce rings with atoms other than carbon in them. OCCH OCCH H (b) CH C(CH₂)s COOCH མ་ནས་བ (c) N=C H -0.X- (e) H C=N COOCHS + CH2=CHCH₂ →→arrow_forward
- 3) Draw a detailed mechanism and predict the product of the reaction shown? 1) EtMgBr 2) H3O+arrow_forwardHow to draw the mechanism for this reaction?arrow_forward> H₂C=C-CH2-CH3 B. H₂O Pt C. + H2 + H₂O H D. 16. Give the IUPAC name for each of the following: B. Cl Cl c. Cl Cl 17. Draw the line-angle formula for each of the following compounds: 1. phenol 2. 1,3-dichlorobenzene 3. 4-ethyltoluene < Previous Submit Assignment Next ▸arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY