
Concept explainers
(a)
Interpretation:
The name and identification of all the present groups or substituents should be determined in the following compound.
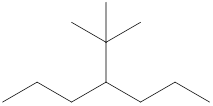
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms.
(b)
Interpretation:
The name and identification of all the present groups or substituents should be determined in the following compound.
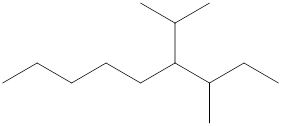
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that are double and triple bonds.
(c)
Interpretation:
The name and identification of all the present groups or substituents should be determined in the following compound.
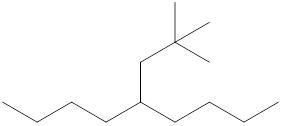
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that are double and triple bonds.
(d)
Interpretation:
The name and identification of all the present groups or substituents should be determined in the following compound.
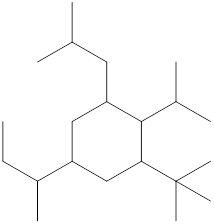
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that are double and triple bonds.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
ORGANIC CHEMISTRY-PRINT (LL)-W/WILEY
- The Ka for sodium dihydrogen phosphate is 6.32 x 10-8. Find the pH of a buffer made from 0.15 M H2PO4- and 0.25 M HPO42- .arrow_forwardThe Ka for lactic acid is 1.4 x 10-4. Find the pH of a buffer made from 0.066 M lactic acid and 0.088 M sodium lactate.arrow_forwardZaitsev's Rule 3) (a) Rank the following alkenes in order of decreasing stability. most stable A B C D > > > (b) Rank the following carbocations in order of decreasing stability least stable B C Darrow_forward
- Calculate the percent ionization for 0.35 M nitrous acid. Use the assumption to find [H3O+] first. K = 7.1 x 10-4arrow_forwardFor each of the following reactions: Fill in the missing reactant, reagent, or product (s), indicating stereochemistry where appropriate using dashed and wedged bonds. If the reaction forms a racemic mixture, draw both structures in the box and write the word “racemic”.arrow_forward5) Using the carbon-containing starting material(s), propose a synthesis based on the following retrosynthetic analysis. Provide structures for all intermediates. The carbon atoms in the product must originate from the starting material(s), but you may use as many equivalents of each starting material as you would like, and any reagent/reaction you know (note: no mechanisms are required). H H =arrow_forward
- Calculate the percent ionization for 0.0025 M phenol. Use the assumption to find [H3O+] first. K = 1.0 x 10-10arrow_forward10:04 AM Tue Mar 25 Sunday 9:30 AM 95% Edit Draw the corresponding structures in each of the boxes below: Ester Name Methyl butyrate (Example) Alcohol Structure H3C-OH Acid Structure Ester Structure Isoamyl acetate Ethyl butyrate Propyl acetate Methyl salicylate Octyl acetate Isobutyl propionate Benzyl butyrate Benzyl acetate Ethyl acetate H₂C OH HCarrow_forward2) For each of the following reactions: (i) (ii) Fill in the missing reactant, reagent, or product (s), indicating stereochemistry where appropriate using dashed and wedged bonds. If the reaction forms a racemic mixture, draw both structures in the box and write the word "racemic". (a) (b) 1) R₂BH 2) H₂O2, NaOH (aq) HBr Br racemic Br + Br Br racemicarrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





