
THERMODYNAMICS LLF W/ CONNECT ACCESS
9th Edition
ISBN: 9781264446889
Author: CENGEL
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3.8, Problem 44P
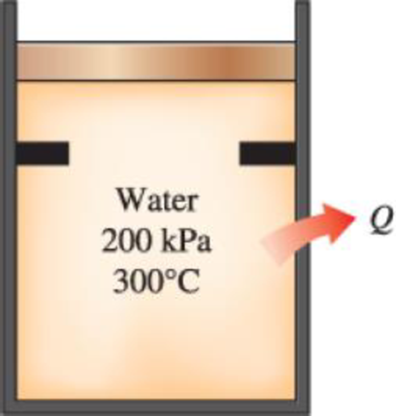
Water initially at 200 kPa and 300°C is contained in a piston–cylinder device fitted with stops. The water is allowed to cool at constant pressure until it exists as a saturated vapor and the piston rests on the stops. Then the water continues to cool until the pressure is 100 kPa. On the T-v diagram, sketch, with respect to the saturation lines, the process curves passing through the initial, intermediate, and final states of the water. Label the T, P, and v values for end states on the process curves. Find the overall change in internal energy between the initial and final states per unit mass of water.
FIGURE P3–44
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Temperature (°C)
100
4. Consider the solidification of a binary Pb-10%Sn alloy. Assume that during solidification,
there is complete mixing in the liquid and no diffusion in the solid. Use the phase diagram
below to answer the following question.
(a)
Draw (on the phase diagram) the compositions of the liquid and the solid at the
interface as a function of temperature during solidification.
(b) Illustrate on the phase diagram how one would calculate the volume fraction solidified
at a given temperature.
(c)
(d)
Indicate the temperature at which solidification is complete.
Do you expect ẞ to be present in the as-cast microstructure? Explain
300
327°C
200
a
(Pb)
20
20
a + L
18.3
183°C
α + β
40
60
Composition (wt% Sn)
Liquid
600
500
232°C
B+L
400
B
61.9
97.8
300
808
100
(Sn)
200
100
Temperature (°F)
I tried this problem a couple of times and don't know where I'm going wrong can you help me out please
y(0)=1,
Using Laplace transforms solve the following differential
equations :
11) y"-4y+4y=0,
12) y+2y+2y=0,
y(0)=2.1,
y'(0) = 3.9
y'(0)=-3.
13) y+7y+12y=21e",
y(0)=3.5,
y'(0)=-10.
14) +9y=10e.
y(0)=0,
y'(0) = 0.
15) y+3y+2.25y=91³ +64,
y(0)=1,
y'(0) = 31.5
16) -6y+5y= 29 cos(21),
y(0)=3.2,
y'(0)=6.2
17) "+2y+2y=0,
y(0)=0,
y'(0)=1.
18) +2y+17y=0,
y(0)=0,
y'(0)=12.
19) y-4y+5y=0,
y(0)=1,
y'(0) = 2.
20) 9y-6y+y=0,
y(0)=3,
y'(0)=1.
21) -2y+10y=0,
y(0)=3,
y'(0)=3.
Chapter 3 Solutions
THERMODYNAMICS LLF W/ CONNECT ACCESS
Ch. 3.8 - A propane tank is filled with a mixture of liquid...Ch. 3.8 - Is iced water a pure substance? Why?Ch. 3.8 - What is the difference between saturated vapor and...Ch. 3.8 - What is the difference between saturated liquid...Ch. 3.8 - If the pressure of a substance is increased during...Ch. 3.8 - Is it true that water boils at higher temperature...Ch. 3.8 - What is the difference between the critical point...Ch. 3.8 - A househusband is cooking beef stew for his family...Ch. 3.8 - How does a boiling process at supercritical...Ch. 3.8 - What is quality? Does it have any meaning in the...
Ch. 3.8 - Does the amount of heat absorbed as 1 kg of...Ch. 3.8 - Does the reference point selected for the...Ch. 3.8 - What is the physical significance of hfg? Can it...Ch. 3.8 - Does hfg change with pressure? How?Ch. 3.8 - Is it true that it takes more energy to vaporize 1...Ch. 3.8 - Which process requires more energy: completely...Ch. 3.8 - In what kind of pot will a given volume of water...Ch. 3.8 - It is well known that warm air in a cooler...Ch. 3.8 - In the absence of compressed liquid tables, how is...Ch. 3.8 - A perfectly fitting pot and its lid often stick...Ch. 3.8 - Complete this table for H2O:Ch. 3.8 - Complete this table for H2O:Ch. 3.8 - Complete this table for H2O:Ch. 3.8 - Complete this table for H2O:Ch. 3.8 - Complete this table for refrigerant-134a:Ch. 3.8 - Complete this table for refrigerant-134a:Ch. 3.8 - A 1.8-m3 rigid tank contains steam at 220C....Ch. 3.8 - One pound-mass of water fills a container whose...Ch. 3.8 - A pistoncylinder device contains 0.85 kg of...Ch. 3.8 - 10 kg of R-134a fill a 1.115-m3 rigid container at...Ch. 3.8 - What is the specific internal energy of water at...Ch. 3.8 - What is the specific volume of water at 5 MPa and...Ch. 3.8 - What is the specific volume of R-134a at 20C and...Ch. 3.8 - Refrigerant-134a at 200 kPa and 25C flows through...Ch. 3.8 - One kilogram of R-134a fills a 0.14-m3 weighted...Ch. 3.8 - One kilogram of water vapor at 200 kPa fills the...Ch. 3.8 - The temperature in a pressure cooker during...Ch. 3.8 - How much error would one expect in determining the...Ch. 3.8 - Water is to be boiled at sea level in a...Ch. 3.8 - Repeat Prob. 340 for a location at an elevation of...Ch. 3.8 - 10 kg of R-134a at 300 kPa fills a rigid container...Ch. 3.8 - 100 kg of R-134a at 200 kPa are contained in a...Ch. 3.8 - Water initially at 200 kPa and 300C is contained...Ch. 3.8 - Saturated steam coming off the turbine of a steam...Ch. 3.8 - A person cooks a meal in a 30-cm-diameter pot that...Ch. 3.8 - Water is boiled at 1 atm pressure in a...Ch. 3.8 - Repeat Prob. 347 for a location at 2000-m...Ch. 3.8 - Prob. 49PCh. 3.8 - A rigid tank with a volume of 1.8 m3 contains 40...Ch. 3.8 - A pistoncylinder device contains 0.005 m3 of...Ch. 3.8 - A 5-ft3 rigid tank contains a saturated mixture of...Ch. 3.8 - Superheated water vapor at 180 psia and 500F is...Ch. 3.8 - One kilogram of water fills a 150-L rigid...Ch. 3.8 - 10 kg of R-134a fill a 0.7-m3 weighted...Ch. 3.8 - A pistoncylinder device contains 0.6 kg of steam...Ch. 3.8 - A pistoncylinder device initially contains 1.4 kg...Ch. 3.8 - Water is being heated in a vertical pistoncylinder...Ch. 3.8 - A rigid tank initially contains 1.4 kg saturated...Ch. 3.8 - A pistoncylinder device initially contains 50 L of...Ch. 3.8 - The spring-loaded pistoncylinder device shown in...Ch. 3.8 - A pistoncylinder device initially contains steam...Ch. 3.8 - Under what conditions is the ideal-gas assumption...Ch. 3.8 - What is the difference between mass and molar...Ch. 3.8 - Propane and methane are commonly used for heating...Ch. 3.8 - What is the specific volume of oxygen at 25 psia...Ch. 3.8 - A 100-L container is filled with 1 kg of air at a...Ch. 3.8 - A mass of 1 lbm of argon is maintained at 200 psia...Ch. 3.8 - A 400-L rigid tank contains 5 kg of air at 25C....Ch. 3.8 - The pressure gage on a 2.5-m3 oxygen tank reads...Ch. 3.8 - A spherical balloon with a diameter of 9 m is...Ch. 3.8 - Reconsider Prob. 373. Using appropriate software,...Ch. 3.8 - A 1-m3 tank containing air at 10C and 350 kPa is...Ch. 3.8 - A mass of 10 g of oxygen fill a weighted...Ch. 3.8 - A mass of 0.1 kg of helium fills a 0.2 m3 rigid...Ch. 3.8 - A rigid tank whose volume is unknown is divided...Ch. 3.8 - A rigid tank contains 20 lbm of air at 20 psia and...Ch. 3.8 - In an informative article in a magazine it is...Ch. 3.8 - What is the physical significance of the...Ch. 3.8 - Determine the specific volume of refrigerant-134a...Ch. 3.8 - Refrigerant-134a at 400 psia has a specific volume...Ch. 3.8 - Determine the specific volume of superheated water...Ch. 3.8 - Determine the specific volume of superheated water...Ch. 3.8 - Determine the specific volume of nitrogen gas at...Ch. 3.8 - Prob. 88PCh. 3.8 - Carbon dioxide gas enters a pipe at 3 MPa and 500...Ch. 3.8 - Prob. 90PCh. 3.8 - A 0.016773-m3 tank contains 1 kg of...Ch. 3.8 - Prob. 92PCh. 3.8 - What is the percentage of error involved in...Ch. 3.8 - What is the physical significance of the two...Ch. 3.8 - Refrigerant-134a at 400 psia has a specific volume...Ch. 3.8 - A 3.27-m3 tank contains 100 kg of nitrogen at 175...Ch. 3.8 - Nitrogen at 150 K has a specific volume of...Ch. 3.8 - A 1-m3 tank contains 2.841 kg of steam at 0.6 MPa....Ch. 3.8 - Prob. 103PCh. 3.8 - Prob. 104PCh. 3.8 - On a certain day, the temperature and relative...Ch. 3.8 - Prob. 106PCh. 3.8 - Consider two rooms that are identical except that...Ch. 3.8 - A thermos bottle is half-filled with water and is...Ch. 3.8 - Complete the blank cells in the following table of...Ch. 3.8 - Complete the blank cells in the following table of...Ch. 3.8 - Prob. 111RPCh. 3.8 - Prob. 112RPCh. 3.8 - The gage pressure of an automobile tire is...Ch. 3.8 - A tank contains argon at 600C and 200 kPa gage....Ch. 3.8 - The combustion in a gasoline engine may be...Ch. 3.8 - Prob. 116RPCh. 3.8 - Prob. 117RPCh. 3.8 - A rigid tank with a volume of 0.117 m3 contains 1...Ch. 3.8 - A 9-m3 tank contains nitrogen at 17C and 600 kPa....Ch. 3.8 - A 10-kg mass of superheated refrigerant-134a at...Ch. 3.8 - A 4-L rigid tank contains 2 kg of saturated...Ch. 3.8 - Prob. 123RPCh. 3.8 - A tank whose volume is unknown is divided into two...Ch. 3.8 - Prob. 125RPCh. 3.8 - A tank contains helium at 37C and 140 kPa gage....Ch. 3.8 - Prob. 127RPCh. 3.8 - On the property diagrams indicated below, sketch...Ch. 3.8 - Ethane at 10 MPa and 100C is heated at constant...Ch. 3.8 - Steam at 400C has a specific volume of 0.02 m3/kg....Ch. 3.8 - Consider an 18-m-diameter hot-air balloon that,...Ch. 3.8 - Prob. 135FEPCh. 3.8 - A 3-m3 rigid vessel contains steam at 2 MPa and...Ch. 3.8 - Prob. 137FEPCh. 3.8 - Water is boiled at 1 atm pressure in a coffeemaker...Ch. 3.8 - Prob. 139FEPCh. 3.8 - Water is boiled in a pan on a stove at sea level....Ch. 3.8 - A rigid tank contains 2 kg of an ideal gas at 4...Ch. 3.8 - The pressure of an automobile tire is measured to...Ch. 3.8 - Consider a sealed can that is filled with...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- 4. Consider the rectangulan 2535 Let 16 a and section discussed 977b + class. in ie make a M thin" rectangle, Can you you show that Q = Go {a² = x² } . Imax = 2 Ga ты J =arrow_forward1. Consider a circular shaft in torsion that of radius r=b has a key way as shown, circle of radius a Let us try the solution x₁ (5,0) = k (6² = r²) (1- 2 awso 1.1 Does this solve the problem for the stres rer 1,2 Solve for is and 23.arrow_forward3. - a For an elliptical cross that the tangent to section resultant shear can you s stress is show ellipse with the same 24 i ratio of eccentricity, in passes through to point alb that in question, it + Parrow_forward
- 2. Consider the rod with an elliptical that strain 4 a Cross secton considered in class, Integrate the was displacement displacements, relations to obtain thearrow_forwardPlease answer Oxygen at 300 kPa and 90°C flowing at an average velocity of 3 m/s is expanded in an adiabatic nozzle. What is the maximum velocity of the oxygen at the outlet of this nozzle when the outlet pressure is 60 kPa? Use the table containing the ideal gas specific heats of various common gases. The maximum velocity of the oxygen at the outlet of this nozzle is 532.5 Numeric ResponseEdit Unavailable. 532.5 incorrect.m/s.arrow_forwardA container filled with 70 kg of liquid water at 95°C is placed in a 90-m3 room that is initially at 12°C. Thermal equilibrium is established after a while as a result of heat transfer between the water and the air in the room. Assume the room is at the sea level, well sealed, and heavily insulated. NOTE: This is a multi-part question. Once an answer is submitted, you will be unable to return to this part. Determine the amount of heat transfer between the water and the air in the room. The amount of heat transfer between the water and the air in the room is kJ.arrow_forward
- A strain gauge rosette that is attached to the surface of a stressed component gives 3 readings (ɛa = A, b = B, &c = C). If the strain gauge rosette is of the D° type (indicating the angle between each of the gauges), construct a Mohr's Strain Circle overleaf. You should assume that gauge A is aligned along the x-axis. Using the Mohr's Strain Circle calculate the: (i) principal strains (ε1, 2)? (ii) principal angles (1, 2)? You should measure these anticlockwise from the y-axis. (iii) maximum shear strain in the plane (ymax)?arrow_forwardQ1. If the yield stress (σy) of a material is 375MPa, determine whether yield is predicted for the stresses acting on both the elements shown below using: (a) Tresca Criterion (b) Von Mises Criterion P Element A R S Element B Note: your values for P (vertical load on Element A) should be negative (i.e. corresponding to a compressive vertical load).arrow_forwardQ. After a puncture a driver is attempting to remove a wheel nut by applying a force of P KN to one end of a wheel brace as shown in Fig. 1. In cross-section the brace is a hollow steel tube (see section aa) of internal diameter r mm and external diameter q mm. wheel nut n Position S P m r q Section aa Fig, 1 (a) Calculate (i) the twisting moment, (ii) the bending moment, and (iii) the shear force in the brace at position S due to the applied load P. (b) Calculate (i) the shear stress due to twisting, and (ii) the bending stress at position S. Note that the shear force will not produce any shear stress at S. (c) Calculate the maximum shearing stress in the brace at position S using the Maximum Shear Stress Criterion. 2 Mechanics of Materials 2 Tutorials Portfolio: Exercise 5 (d) If the maximum permissible shear stress in the steel is 200 MPa, determine the maximum torque that can be applied by the brace without the risk of failure at S.arrow_forward
- Calculate the first 5 Fourier series coefficients (A0-4 and B1-5 ) for the estimated R wave.arrow_forwardRefrigerant-134a is expanded isentropically from 600 kPa and 70°C at the inlet of a steady-flow turbine to 100 kPa at the outlet. The outlet area is 1 m2, and the inlet area is 0.5 m2. Calculate the inlet and outlet velocities when the mass flow rate is 0.65 kg/s. Use the tables for R-134a. The inlet velocity is m/s. The outlet velocity is m/s.arrow_forwardA container filled with 70 kg of liquid water at 95°C is placed in a 90-m3 room that is initially at 12°C. Thermal equilibrium is established after a while as a result of heat transfer between the water and the air in the room. Assume the room is at the sea level, well sealed, and heavily insulated. NOTE: This is a multi-part question. Once an answer is submitted, you will be unable to return to this part. Determine the final equilibrium temperature. Use the table containing the ideal gas specific heats of various common gases. The final equilibrium temperature is °C.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY
First Law of Thermodynamics, Basic Introduction - Internal Energy, Heat and Work - Chemistry; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=NyOYW07-L5g;License: Standard youtube license