
Campbell Biology, Books a la Carte Plus Mastering Biology with eText -- Access Card Package (10th Edition)
10th Edition
ISBN: 9780133922851
Author: Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3.1, Problem 2CC
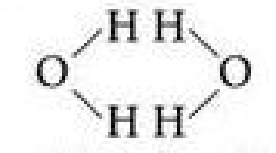
Why is it unlikely that two neighboring water molecules would be arranged like this?
Expert Solution & Answer
Trending nowThis is a popular solution!
Learn your wayIncludes step-by-step video

schedule01:15
Students have asked these similar questions
After you feel comfortable with your counting method and identifying cells in the various stages of mitosis, use the four images below of whitefish blastula to count the cells in each stage until you reach 100 total cells, recording your data below in Data Table 1. (You may not need to use all four images. Stop counting when you reach 100 total cells.)
After totaling the cells in each stage, calculate the percent of cells in each stage. (Divide total of stage by overall total of 100 and then multiply by 100 to obtain percentage.)
Data Table 1Stage Totals PercentInterphase Mitosis: Prophase Metaphase Anaphase Telophase Cytokinesis Totals 100 100%
To find the length of time whitefish blastula cells spend in each stage, multiply the percent (recorded as a decimal, in other words take the percent number and divide by 100) by 24 hours. (Example: If percent is 20%, then Time in Hours = .2 * 24 = 4.8) Record your data in Data…
What are Clathrin coated vesicles and what is their function?
How is a protein destined for the Endoplasmic Reticulum (ER), imported into the ER? Be concise.
Chapter 3 Solutions
Campbell Biology, Books a la Carte Plus Mastering Biology with eText -- Access Card Package (10th Edition)
Ch. 3.1 - MAKE CONNECTIONS What is electronegativity, and...Ch. 3.1 - Why is it unlikely that two neighboring water...Ch. 3.1 - WHAT IF? What would be the effect on the...Ch. 3.2 - Prob. 1CCCh. 3.2 - Prob. 2CCCh. 3.2 - WHAT IF? A water strider (an insect that can walk...Ch. 3.2 - WHAT IF? A water strider (an insect that can walk...Ch. 3.2 - Prob. 5CCCh. 3.3 - Compared with a basic Solution at pH 9, the same...Ch. 3.3 - HCl is a strong acid that dissociates in water:...
Ch. 3.3 - Prob. 3CCCh. 3.3 - Prob. 4CCCh. 3 - DRAW IT Label a hydrogen bond and a polar...Ch. 3 - Describe how different types of solutes dissolve...Ch. 3 - Prob. 3.3CRCh. 3 - Which of the following is a hydrophobic material?...Ch. 3 - Prob. 2TYUCh. 3 - Measurements show that the pH of a particuiar lake...Ch. 3 - What is the hydroxide ion concentration of the...Ch. 3 - A slice of pizza has 500 kcal. If we could burn...Ch. 3 - DRAW IT Draw the hydration shells that form around...Ch. 3 - In agricultural areas, farmers pay close attention...Ch. 3 - EVOLUTION CONNECTION This chapter explains how the...Ch. 3 - Prob. 9TYUCh. 3 - WRITE ABOUT A THEME: ORGANIZATION Several emergent...Ch. 3 - SYNTHESIZE YOUR KNOWLEDGE How do cats drink?...
Additional Science Textbook Solutions
Find more solutions based on key concepts
35. Consider the reaction.
The graph shows the concentration of Br2 as a function of time.
a. Use the g...
Chemistry: Structure and Properties (2nd Edition)
What name is given to the zone of greatest seismic activity?
Applications and Investigations in Earth Science (9th Edition)
Thiols such as ethanethiol and propanethiol can be used to reduce vitamin K epoxide to vitamin KH2, but they re...
Organic Chemistry (8th Edition)
Where is transitional epithelium found and what is its importance at those sites?
Anatomy & Physiology (6th Edition)
In rabbits, chocolate-colored fur (w+) is dominant to white fur (w), straight fur (c+) is dominant to curly fur...
Genetic Analysis: An Integrated Approach (3rd Edition)
An obese 55-year-old woman consults her physician about minor chest pains during exercise. Explain the physicia...
Biology: Life on Earth with Physiology (11th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Find out about the organisations and the movements aimed at the conservation of our natural resources. Eg Chipko movement and Greenpeace. Make a project report on such an organisation.arrow_forwardWhat are biofertilizers and mention the significancearrow_forwardPCBs and River Otters: Otters in Washington State’s Green-Duwamish River have high levels of polychlorinated biphenyls (PCBs) in their livers. PCBs can bind to the estrogen receptors in animals and disrupt the endocrine system of these otters. The PCBs seem to increase the estrogen to androgen ratio, skewing the ratio toward too much estrogen. How would increased estrogen affect the river otter population? Based on your reading of the materials in this unit, what factors can affect fertility in humans? Explain how each of the factors affecting human fertility that you described can disrupt the human endocrine system to affect reproduction.arrow_forward
- Other than oil and alcohol, are there other liquids you could compare to water (that are liquid at room temperature)? How is water unique compared to these other liquids? What follow-up experiment would you like to do, and how would you relate it to your life?arrow_forwardSelection of Traits What adaptations do scavengers have for locating and feeding on prey? What adaptations do predators have for capturing and consuming prey?arrow_forwardCompetition Between Species What natural processes limit populations from growing too large? What are some resources organisms can compete over in their natural habitat?arrow_forward
- Species Interactions Explain how predators, prey and scavengers interact. Explain whether predators and scavengers are necessary or beneficial for an ecosystem.arrow_forwardmagine that you are conducting research on fruit type and seed dispersal. You submitted a paper to a peer-reviewed journal that addresses the factors that impact fruit type and seed dispersal mechanisms in plants of Central America. The editor of the journal communicates that your paper may be published if you make ‘minor revisions’ to the document. Describe two characteristics that you would expect in seeds that are dispersed by the wind. Contrast this with what you would expect for seeds that are gathered, buried or eaten by animals, and explain why they are different. (Editor’s note: Providing this information in your discussion will help readers to consider the significance of the research).arrow_forwardWhat is the difference between Uniporters, Symporters and Antiporters? Which of these are examples of active transport?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning
Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College

Biology (MindTap Course List)
Biology
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Cengage Learning

Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Human Biology (MindTap Course List)
Biology
ISBN:9781305112100
Author:Cecie Starr, Beverly McMillan
Publisher:Cengage Learning

Concepts of Biology
Biology
ISBN:9781938168116
Author:Samantha Fowler, Rebecca Roush, James Wise
Publisher:OpenStax College

Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license