
Concept explainers
Recall from section 1.10B that there is restricted rotation around carbon-carbon double
bonds. Maleic acid and fumaric acid are two isomers with vasity different physical properties and
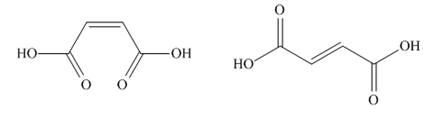
maleic acid fumaric acid
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
EBK ORGANIC CHEMISTRY
- Aspirin is prepared by the reaction of salicylic- acid with acetic anhydride as shown in the following equation. The stoichiometry of the reaction is given in the equation. Acetic acid is a by-product of the reaction and must be separated and removed so that aspirin can then be sold as a pure product. How many grams of aspirin can be prepared from 120 grams of salicylic acid? Assume that there is an excess of acetic anhydride. (Chapter 4) Acetylsalieylic acid (Aspirin)arrow_forwardFunctional Group Classes Alcohol Amide Ester • CH₂CH₂OH * CHIO-CH, 7. H₂C1O Amine Ketone Circle each functional group(s) and identify the class to which each of the groups belongs. 9. H₂C Aldehyde Thiol Aromatics Carboxylic Acid 2. CH,SH 6. Alkenes 4. H₂NCH₂CH₂ 10. Ether NM₂ 11. Which of the functional groups in the top list must be on a terminalC in the carbon chain?arrow_forward1. Consider the solubility and boiling point of the following pair of compounds: n-butyl alcohol and diethyl ether. The boiling points for the compounds are 118 °C and 35 °C respectively. The solubility for both compounds is the same (8g/100g water). Explain this observation for (i) boiling point disparity; (ii) solubility similarity a. H-bonds form in diethyl ether; n-butyl alcohol forms H-bonds in water b. H-bonds form in n-butyl alcohol; diethyl ether forms H-bonds in water c. H-bonds in n-butyl alcohol; Both compounds form H-bonds in water d. Both compounds form H-bonds; Both compounds form H-bonds in water 2. Account for the bond angle differences between (i) H-C-H (109.5°) in methane and H-S-H (90°); H-C-H (109.5°) and H-O-H (107.5°) in water. a. The H-S-H has two lone pairs; The H-O-H has two lone pairs b. The H-S-H has no hybridization at p-orbitals; The H-O-H has two lone pairs c. The H-S-H has two lone pairs; The H-O-H has no hybridization…arrow_forward
- Explain why stearic acid has higher melting point than decanoic acid. Explain why salicylic acid has higher melting point than benzoic acid. Explain why benzoic acid has higher melting point than stearic acid. Explain why octane has a higher melting point than isooctane. Explain why 2,2,3,3-tetramethylbutane has the highest melting point among the three isomers of C8H18arrow_forwardexplain why methanethiol, CH3SH, has a lower boiling point (6°C) than methanol, CH3OH (65°C), even though methanethiol has a higher molecular weightarrow_forwardwhich functional groups are in C6H12O2 with it having 1 degree of saturation, and is the molecule hexanoic acid?arrow_forward
- 3. Molecules with more than one alcohol group can react with thionyl chloride (SOCI.) in a way that is different from that of single alcohols. Consider the example below. Provide a reasonable structure for product A CI + 2 HCI OH (product A) онarrow_forwardNeutral/acidic/ basic Polar/ non-polar Hydrophobic/hydrophilicarrow_forwardThe most polar bond of a carboxyl group is the C─O single bond. true or falsearrow_forward
- Please answer #7 and 8arrow_forwardThe normal pH range for blood plasma is 7.35–7.45. Under these conditions, would you expect the carboxyl group of lactic acid (pKa 3.08) to exist primarily as a carboxyl group or as a carboxylic anion? Explain.arrow_forwardTHE ZnCl2 / HCl TEST DETERMINES THE REACTIVITY OF ALCOHOLS. ORDER IN ORDER FROM GREATEST TO LOWEST REACTIVITY METHANOL-TERBUTANOL-2PROPANOL TERBUTANOL-METHANOL-METHANOL METHANOL-2 PROPANOL-TERBUTANOLarrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





