
CHEMISTRY-TEXT
8th Edition
ISBN: 9780134856230
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 3, Problem 3.107SP
Interpretation Introduction
(a)
Interpretation:
An empirical formula of
Concept introduction:
The simplest positive integer ratio of atoms of the chemical compound is called as an empirical formula.
Interpretation Introduction
(b)
Interpretation:
A molecular weight and its molecular formula from mass spectrum of
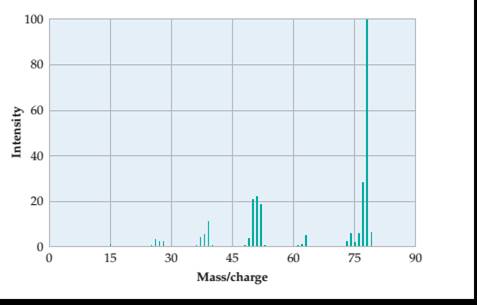
Concept introduction:
The value of empirical and molecular formula may be same or a multiple of compound empirical formula.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
presented by Morillon Leaning
Predict the organic product for the min
кусур
HSC
Adithane carved arnown to come than that to the condon
slchroruis in acid in in aquishri with
ною
6.15PM
Sun Mar 30
K
Draw the major product of this reaction. Include
any relevant stereochemistry. Ignore inorganic
byproducts.
Problem 1 of
O
H
[PhзPCH2CH3]*C|¯
NaH
Drawing
>
Q
Atoms,
Bonds and
Draw or tap a ne
8:17 PM Sun Mar 30
Draw the major product of this reaction. Ignore
inorganic byproducts.
HSCH2CH2CH2SH, BF3
Probler
Drawing
Ato
Bonds
Cl
Chapter 3 Solutions
CHEMISTRY-TEXT
Ch. 3 - Prob. 3.1PCh. 3 - If blue spheres represent nitrogen atoms and red...Ch. 3 - Prob. 3.3PCh. 3 - APPLY 3.4 The major ingredient in ordinary safety...Ch. 3 - Calculate the molecular weight of sulfuric acid...Ch. 3 - Conceptual APPLY 3.6 Use the structural formula of...Ch. 3 - PRACTICE 3.7 How many moles arc in 5.26 g of...Ch. 3 - APPLY 3.8 When a diabetic experiences low blood...Ch. 3 - PRACTICE 3.9 Aspirin is prepared by reaction...Ch. 3 - APPLY 3.10 Refer to the balanced reaction for the...
Ch. 3 - PRACTICE 3.11 Ethyl alcohol is prepared...Ch. 3 - APPLY 3.12 (a) Diethyl ether (C4H10O), the “ether”...Ch. 3 - Conceptual PRACTICE 3.13 The following diagram...Ch. 3 - Conceptual APPLY 3.14 Draw a diagram similar to...Ch. 3 - Lithium oxide is used aboard the space shuttle to...Ch. 3 - APPLY 3.16 After lithium hydroxide is produced...Ch. 3 - PRACTICE 3.17 What is the empirical formula of the...Ch. 3 - Conceptual APPLY 3.19 Use the structural formula...Ch. 3 - PRACTICE 3.20 Menthol, a flavouring agent obtained...Ch. 3 - PRACTICE 321 Combustion analysis is performed on...Ch. 3 - PRACTICE 3.23 A compound has an empirical formula...Ch. 3 - APPLY 3.24 Combustion analysis was performed on...Ch. 3 - Prob. 3.23PCh. 3 - Match the terms percent yield and percent atom...Ch. 3 - Examine two reactions important in chemical...Ch. 3 - Propene is a raw material for a wide variety of...Ch. 3 - Ibuprofen (the active ingredient in the...Ch. 3 - The original synthesis for ibuprofen, developed in...Ch. 3 - The reaction of A (red spheres) with B (blue...Ch. 3 - The diagrams represent a reaction on the molecular...Ch. 3 - Fluoxetine, marketed as an antidepressant under...Ch. 3 - The following diagram represents the reaction of...Ch. 3 - What is the percent composition of cysteine, one...Ch. 3 - Cytosine, a constituent of deoxyribonucleic acid...Ch. 3 - A hydrocarbon of unknown formula CxHy was...Ch. 3 - Which of the following equations are balanced? (a)...Ch. 3 - Which of the following equations are balanced?...Ch. 3 - Balance the following equations: (a)...Ch. 3 - Balance the following equations: (a) The explosion...Ch. 3 - Balance the following equations: (a)...Ch. 3 - Balance the following equations:...Ch. 3 - Balance the following equations. (a)...Ch. 3 - Balance the following equations. (a) CO(...Ch. 3 - What are the molecular (formula) weights of the...Ch. 3 - What are the formulas of the following substances?...Ch. 3 - What are the molecular weights of the following...Ch. 3 - Prob. 3.47SPCh. 3 - How many grams are in a mole of each of the...Ch. 3 - Prob. 3.49SPCh. 3 - How many moles of ions are in 27.5 g of MgCl2?Ch. 3 - How many moles of anions are in 35.6 g of AlF3?Ch. 3 - What is the molecular weight of chloroform if...Ch. 3 - What is the molecular weight of cholesterol if...Ch. 3 - 3.52 Iron (II) sulfate, FeSO4, is prescribed for...Ch. 3 - Prob. 3.55SPCh. 3 - An average cup of coffee contains about 125 mg of...Ch. 3 - Prob. 3.57SPCh. 3 - A sample that weighs 25.12 g contains 6.0221023...Ch. 3 - Prob. 3.59SPCh. 3 - Prob. 3.60SPCh. 3 - Prob. 3.61SPCh. 3 - In the preparation of iron from hematite, Fe2O3...Ch. 3 - An alternative method for preparing pure iron from...Ch. 3 - Magnesium metal burns in oxygen to form...Ch. 3 - Ethylene gas, C2H4 , reacts with water at high...Ch. 3 - Prob. 3.66SPCh. 3 - Titanium dioxide (TiO2) , the substance used as...Ch. 3 - Prob. 3.68SPCh. 3 - Aluminum reacts with oxygen to yield aluminum...Ch. 3 - The industrial production of hydriodic acid takes...Ch. 3 - An alternative method for producing hydriodic acid...Ch. 3 - Nickel(II) sulfate, used for nickel plating, is...Ch. 3 - Hydrazine, N2H4 , once used as a rocket...Ch. 3 - Assume that you have 1.39 mol of H2 and 3.44 mol...Ch. 3 - Hydrogen and chlorine react to yield hydrogen...Ch. 3 - How many grams of the dry-cleaning solvent...Ch. 3 - How many grams of each product result from the...Ch. 3 - Limestone (CaCO3) reacts with hydrochloric acid...Ch. 3 - Sodium azide (NaN3) yields N2 gas when heated to...Ch. 3 - Acetic acid (CH3CO2H) reacts with isopentyl...Ch. 3 - Cisplatin [Pt( NH 3)2Cl2] , a compound used in...Ch. 3 - If 1.87 g of acetic acid (CH3COOH) reacts with...Ch. 3 - If 3.42 g of K2PtCl4 and 1.61 g of NH3 give 2.08 g...Ch. 3 - The reaction of tungsten hexachloride (WCl6) with...Ch. 3 - Sodium borohydride, NaBH4 , a substance used in...Ch. 3 - Urea, a substance commonly used as a fertilizer,...Ch. 3 - Calculate the mass percent composition of each of...Ch. 3 - What are the empirical formulas of substances with...Ch. 3 - Ferrocene, a substance proposed for use as a...Ch. 3 - What is the empirical formula of stannous...Ch. 3 - What are the empirical formulas of each of the...Ch. 3 - An unknown liquid is composed of 5.57% 11, 28.01 %...Ch. 3 - An unknown liquid is composed of 34.31% C, 5.28%...Ch. 3 - Combustion analysis of 45.62 mg of toluene, a...Ch. 3 - Coniine, a toxic substance isolated from poison...Ch. 3 - Cytochrome c is an iron—containing enzyme found in...Ch. 3 - Nitrogen fixation in the root nodules of peas and...Ch. 3 - Disilane, Si2Hx, is analyzed and found to contain...Ch. 3 - A certain metal sulfide, MS2, is used extensively...Ch. 3 - Combustion analysis of a 31.472 mg sample of the...Ch. 3 - The stimulant amphetamine contains only carbon,...Ch. 3 - Prob. 3.102SPCh. 3 - Prob. 3.103SPCh. 3 - Prob. 3.104SPCh. 3 - Prob. 3.105SPCh. 3 - Prob. 3.106SPCh. 3 - Prob. 3.107SPCh. 3 - Prob. 3.108MPCh. 3 - The molar mass of HCl is 36.5 g/mol, and the...Ch. 3 - Assume that gasoline has the formula C8H18 and has...Ch. 3 - Compound X contains only carbon, hydrogen,...Ch. 3 - Prob. 3.112MPCh. 3 - A certain alcoholic beverage contains only ethanol...Ch. 3 - A mixture of FeO and Fe2O3 with a mass of 10.0 g...Ch. 3 - Prob. 3.115MPCh. 3 - When eaten, dietary carbohydrates are digested to...Ch. 3 - Prob. 3.117MPCh. 3 - Prob. 3.118MPCh. 3 - A mixture of XCl3 and XCl5 weighing 10.00 g...Ch. 3 - Ammonium nitrate, a potential ingredient of...Ch. 3 - Window glass is typically made by mixing soda ash...Ch. 3 - Prob. 3.122MPCh. 3 - Ethylene glycol, commonly used as automobile...Ch. 3 - Prob. 3.124MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Name the major organic product of the following action of 4-chloro-4-methyl-1-pentanol in neutral pollution 10+ Now the product. The product has a molecular formula f b. In a singly hain, the starting, material again converts into a secule with the molecular kormula CIO. but with comply Draw the major organic structure inhalationarrow_forwardMacmillan Learning Alcohols can be oxidized by chromic acid derivatives. One such reagent is pyridinium chlorochromate, (C,H,NH*)(CICTO3), commonly known as PCC. Draw the proposed (neutral) intermediate and the organic product in the oxidation of 1-butanol by PCC when carried out in an anhydrous solvent such as CH₂C₁₂. PCC Intermediate OH CH2Cl2 Draw the intermediate. Select Draw Templates More с H Cr о Product Draw the product. Erase Select Draw Templates More H о Erasearrow_forwardIf I have 1-bromopropene, to obtain compound A, I have to add NaOH and another compound. Indicate which compound that would be. A C6H5 CH3arrow_forward
- Provide the reagents for the following reactions.arrow_forwardIf I have 1-bromopropene, to obtain compound Z, I have to add two compounds A1 and A2. Indicate which compounds are needed. P(C6H5)3arrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts. Assume that the water side product is continuously removed to drive the reaction toward products. O CH3CH2NH2, TSOH Select to Draw >arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Step by Step Stoichiometry Practice Problems | How to Pass ChemistryMole Conversions Made Easy: How to Convert Between Grams and Moles; Author: Ketzbook;https://www.youtube.com/watch?v=b2raanVWU6c;License: Standard YouTube License, CC-BY