
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.9, Problem 4P
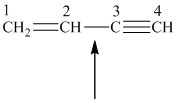
The hydrocarbon shown, called vinylacetylene, is used in the synthesis of neoprene, a
synthetic rubber. Identify the orbital overlaps involved in the indicated bond. How many
bonds are there in vinylacetylene? How many
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
find the pH of a buffer made from 0.20 M HNO2 and 0.10 M NaNO2. Ka= 4.0 x 10-4a) 4.00b) 3.40c) 3.70d) 3.10
the Ka for sodium dihydrogen phosphate is 6.32 x 10-8. Find the pH of a buffer made from 0.15 M H2PO4- and 0.15 M HPO42-.a) 6.98b) 7.42c) 7.00d) 7.20
Find the equilibrium concentration of H3O+ starting with 0.072 M solution of acetic acid. Ka = 1.8 x 10-5. Acetic acid is HC2H3O2 (aq).HC2H3O2 (aq) + H2O (l) ⇌ H3O (aq) + C2H3O2- (aq)
a) 1.3 x 10-6
b) 1.1 x 10-3
c) 1.5 x 10-2
d) 3.6 x 10-5
Chapter 2 Solutions
Organic Chemistry - Standalone book
Ch. 2.4 - Prob. 1PCh. 2.7 - Prob. 2PCh. 2.8 - Identify the orbital overlaps of all of the bonds...Ch. 2.9 - The hydrocarbon shown, called vinylacetylene, is...Ch. 2.12 - Prob. 5PCh. 2.12 - Prob. 6PCh. 2.13 - Prob. 7PCh. 2.14 - Refer to Table 2.2 as needed to answer the...Ch. 2.15 - Prob. 9PCh. 2.15 - Prob. 10P
Ch. 2.16 - Prob. 11PCh. 2.17 - Prob. 12PCh. 2.18 - Prob. 13PCh. 2.20 - Prob. 14PCh. 2.21 - Match the boiling points with the appropriate...Ch. 2.22 - Write a balanced chemical equation for the...Ch. 2.22 - Using the data in Table 2.3, estimate the heat of...Ch. 2.22 - Prob. 18PCh. 2.22 - Prob. 19PCh. 2.23 - Prob. 20PCh. 2.23 - Which of the following reactions requires an...Ch. 2 - The general molecular formula for alkanes is...Ch. 2 - Prob. 23PCh. 2 - Prob. 24PCh. 2 - Prob. 25PCh. 2 - What is the hybridization of each carbon in...Ch. 2 - Prob. 27PCh. 2 - Does the overlap of two p orbitals in the fashion...Ch. 2 - Prob. 29PCh. 2 - Aphids secrete an alarm pheromone having the...Ch. 2 - All the parts of this problem refer to the alkane...Ch. 2 - Prob. 32PCh. 2 - Prob. 33PCh. 2 - Prob. 34PCh. 2 - From among the 18 constitutional isomers of C8H18,...Ch. 2 - Give the IUPAC name for each of the following...Ch. 2 - Using the method outlined in Section 2.16, give an...Ch. 2 - Prob. 38PCh. 2 - Write a balanced chemical equation for the...Ch. 2 - The heats of combustion of methane and butane are...Ch. 2 - In each of the following groups of compounds,...Ch. 2 - Given H for the reaction H2(g)+12O2(g)H2O(l)...Ch. 2 - Prob. 43PCh. 2 - Prob. 44PCh. 2 - Prob. 45PCh. 2 - Prob. 46PCh. 2 - Prob. 47PCh. 2 - Compound A undergoes the following reactions:...Ch. 2 - Prob. 49PCh. 2 - Some Biochemical Reactions of Alkanes Alkanes...Ch. 2 - Prob. 51DSPCh. 2 - Some Biochemical Reactions of Alkanes Alkanes...Ch. 2 - Prob. 53DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- in VSEPR Theory, AX2 isarrow_forwardcalculate the pH of 0.066 M Ca(OH)2. Remember stoichiometry.arrow_forwardFind the equilibrium concentration of H3O+ starting with 0.072 M solution of acetic acid. Ka = 1.8 x 10-5. Acetic acid is HC2H3O2 (aq).HC2H3O2 (aq) + H2O (l) ⇌ H3O (aq) + C2H3O2- (aq)arrow_forward
- in VSEPR Theory AX2 isa) tetrahedralb) octahedralc) lineard) trigonal bipyramidarrow_forwardCheck How many signals would you expect to find in the H NMR spectrum of each given compound? Part 1 of 2 Part 2 of 2 Br Br 2. Cl X 2 © 2025 McGraw Hill LLC. All Rights Resarrow_forwardcalculate the pH of 0.015 M H2SO4. Remember stoichiometry.arrow_forward
- Check Consider the 13 C NMR spectrum below. 140 120 100 80 60 60 PPM 40 20 0 The spectrum belongs to which one of the following constitutional isomers of the compound C 10H14? Select the single best answer. ✓ Save © 2025 McGraw Hill LLC. All Rights Reserved.arrow_forwardThe structure of compound 1,1,2-trichloropropane is given below. Cl Cl Cl 1 How many signals would you expect to find in the 'H NMR spectrum of 1,1,2-trichloropropane? ×arrow_forward1, How many signals do you expect in the H NMR spectrum for this molecule? Write the answer below. Also, in each of the drawing areas below is a copy of the molecule, with Hs shown. In each copy, one of the H atoms is colored red. Highlight in red all other H atoms that would contribute to the same signal as the H already highlighted red. Note for advanced students: In this question, any multiplet is counted as one signal. Number of signals in the 'H NMR spectrum. For the molecule in the top drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. No additional Hs to color in top molecule For the molecule in the bottom drawing area, highlight in red any other H atoms that will contribute. to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. No additional Hs to color in bottom molecule Check…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY