
Concept explainers
(a)
Interpretation:
Explain why the following equilibrium lies far to the right.
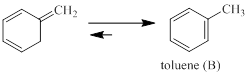
Concept introduction:
In chemistry, resonance is a systematic way to describe bonding nature of certain molecules of ions (cation or anion) by combining several structures which are contributing to the core structure. The various structures derived for a molecule is referred as resonance structures or canonical structures.
(b)
Interpretation:
Chemists had always assumed that this reaction would be so fast that compound A could never be isolated. However, this compound was prepared in 1962 and shown to be stable in the gas phase at 70°C, despite the favorable equilibrium constant for its transformation to B.Show why the conversion of A into B above would not be expected to occur as a concerted reaction.
Concept introduction:
Generally, Sigmatropic reaction is referred as the migration of allylic sigma bond at one end of the π-electron system to the other end of the π-electron system as an uncatalyzed intramolecular reaction. Though, the position of π-bond is changed in Sigmatropic reaction, the total number of π-bonds remain unchanged. The sigma bond can be cleaved at the middle or at the end of the π-system. The formation of sigma bond at 3, 3-position of a 1, 5-diene is called as cope rearrangement.
(c)
Interpretation:
The concerted mechanism for the following reaction is to be explained.
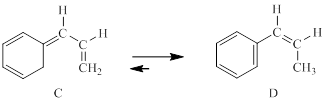
Concept introduction:
Generally, Sigmatropic reaction is referred as the migration of allylic sigma bond at one end of the π-electron system to the other end of the π-electron system as an uncatalyzed intramolecular reaction. Though, the position of π-bond is changed in Sigmatropic reaction, the total number of π-bonds remain unchanged. The sigma bond can be cleaved at the middle or at the end of the π-system. The formation of sigma bond at 3, 3-position of a 1, 5-diene is called as cope rearrangement.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- Can I please get help with this?arrow_forwardC. I, II, III Consider the reaction sequence below to answer the following questions: 0 0 1. NaOEt, EtOH ΕΙΟ OEt 2 Compound X CO₂Et NaOEt, EtOH CO₂Et Br Compound Y A Compound Z A. Compound X, diethyl propanedioate, is more commonly known as a. ethyl acetoacetate acetoacetic ester b. C. oxalic ester d. malonic ester B. Write the complete stepwise mechanism for the conversion of Compound X into Compound Y. Show all electron flow with arrows and draw all intermediate structures.arrow_forwardDiethyl malonate can be prepared by the following reaction sequence. Draw the structures of each of the missing intermediates in the boxes provided EtO 0 H3C 11 C 1. Br₂ PBr OH 2 H₂O 010 0 CH3CH₂OH C CH2 OEt Ha CH3CH2OH на NaCN H₂SO4 NC H₂O, heat CH2 OCH2CH3arrow_forward
- Show how you would accomplish each of the following transformations. More than one step may be quired. Show all reagents and all intermediate structures. [three only] A. 0 CH3 B. C. D. H 0 0 OCH 3 CH₂CO₂CH2CH3 H3C ➤ HN C NO₂ Clarrow_forwardChoose the BEST reagent for carrying out each of the following conversions. A. CO₂CH3 CO₂CH3 0 CO₂H a. LiAlH4, ether C. CrO3, pyridine B. 0 H a. C. NaBH4, ethanol NaOH, H2O CO₂H OH HD b. NaBH4, ethanol d. H₂/Pd CH₂OH b. CH₂PPh3 d. All of the abovearrow_forwardWrite the complete stepwise mechanism for the acid-catalyzed hydrolysis of the following amide to yield mandelic acid. Show all electron flow with arrows and draw the structures of all intermediate species. OH H-OH₂ CnH2 :0: OH C OH + NH4 10: The purpose of the acid catalyst in the hydrolysis of an amide is: to enhance the electrophilicity of the amide carbonyl carbon a. to enhance the nucleophilicity of the water molecule b. C. to enhance the electrophilicity of the water molecule d. to shift the equilibrium of the reactionarrow_forward
- 1.arrow_forwardCan I please get help with this?arrow_forward. Provide IUPAC names for each of the following structures OR draw structures corresponding to each of the following names: [Three only]kk a. H₂N- 0 COCH2CH3 benzocaine b. What is the correct structure for phenylbenzoate? C a. 0 C-O O b. H3C-C-O 0 0 C-O-CH3 d. CH₂O C-CHZ c. Acetyl chloride d. 3,4,5-trimethoxybenzoyl chloridearrow_forward
- . Draw structures corresponding to each of the following names or Provide IUPAC names for each of the ollowing structures [for 4 ONLY]. A. 2-propylpentanoic acid. B. m-chlorobenzoic acid. C. 0 0 HOC(CH2) COH glutaricadd D. E. F. 0 OH HO OH HO INCO salicylicadd H3C CH3 C=C tgicadd H COOH CH₂C=N 4arrow_forwardThe reaction of a carboxylic acid with an alcohol in the presence of acid is termed Fischer esterification. 0 0 C .C. OH + CH3OH OCH3 + H₂O HCI A B C A. The nucleophile in this reaction is B. Compound C functions as a. a base scavenger b. a solvent C. a catalyst in this reaction. d. a neutralizer C. Fischer esterification is an example of: ........ a. nucleophilic acyl addition b. nucleophilic acyl substitution c. nucleophilic acyl elimination d. nucleophilic acyl rearrangementarrow_forwardThe Handbook of Chemistry and Physics gives solubilities of the following compounds in grams per 100 mL of water. Because these compounds are only slightly soluble, assume that the volume does not change on dissolution and calculate the solubility product for each. (a) BaSeO4, 0.0118 g/100 mLarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





