
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 27.4, Problem 27.6P
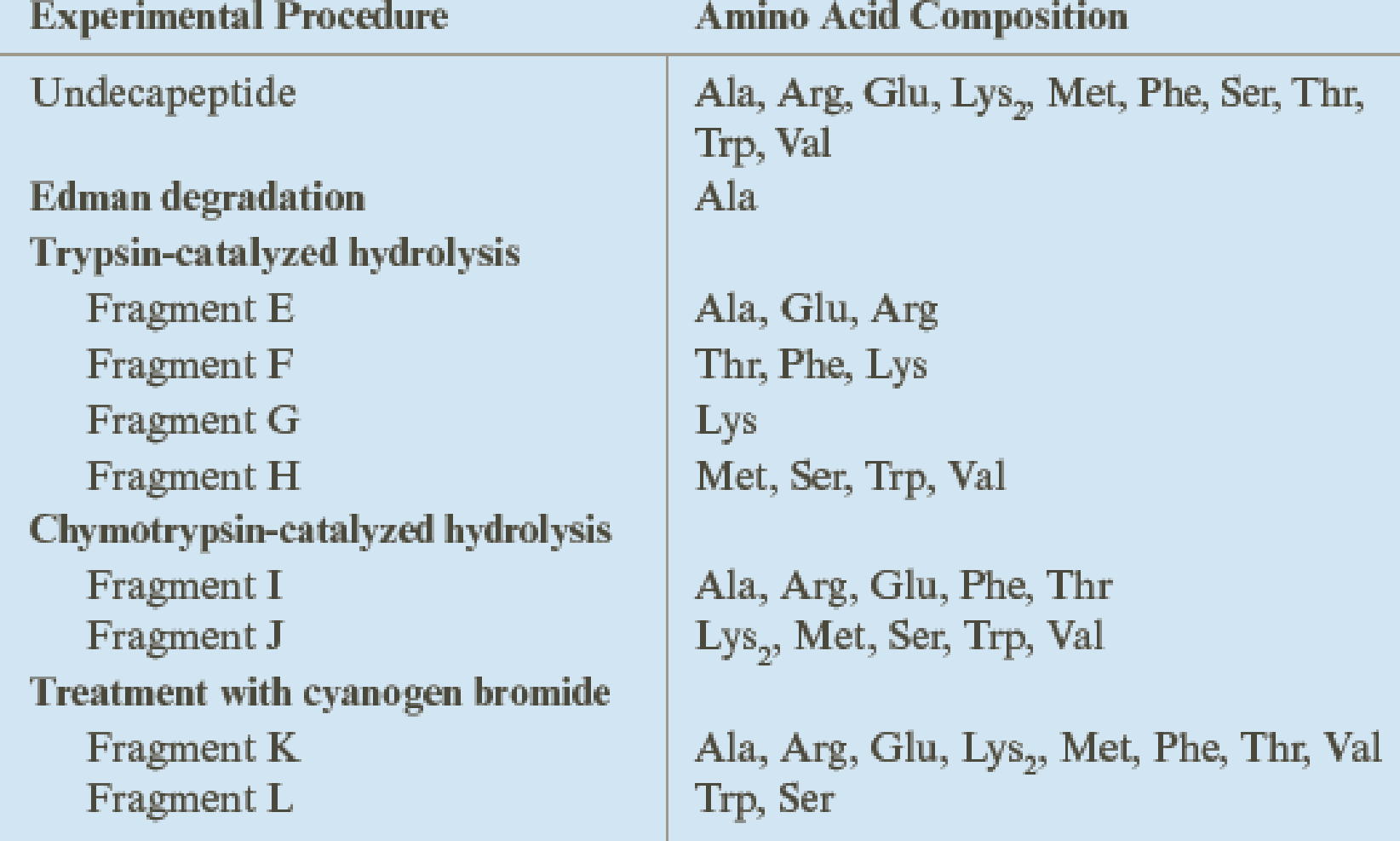
Deduce the amino acid sequence of an undecapeptide (11 amino acids) from the experimental results shown in the table.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Electrochemistry. Briefly describe the Donnan potential.
Indicate what the Luther equation is used for?
Indicate one aspect that benefits and another that makes it difficult to use the hydroquinone electrode to measure pH.
Chapter 27 Solutions
Organic Chemistry
Ch. 27.1 - Of the 20 protein-derived amino acids shown in...Ch. 27.2 - Prob. 27.2PCh. 27.2 - Prob. 27.3PCh. 27.3 - Draw a structural formula for Lys-Phe-Ala. Label...Ch. 27.4 - Which of these tripeptides are hydrolyzed by...Ch. 27.4 - Deduce the amino acid sequence of an undecapeptide...Ch. 27.6 - Prob. 27.7PCh. 27 - What amino acid does each abbreviation stand for?...Ch. 27 - The configuration of the chiral center in -amino...Ch. 27 - Assign an R or S configuration to the chiral...
Ch. 27 - Prob. 27.11PCh. 27 - Prob. 27.12PCh. 27 - Draw zwitterion forms of these amino acids. (a)...Ch. 27 - Prob. 27.14PCh. 27 - Why is Arg often referred to as a basic amino...Ch. 27 - Prob. 27.16PCh. 27 - Prob. 27.17PCh. 27 - Prob. 27.18PCh. 27 - Prob. 27.19PCh. 27 - Prob. 27.20PCh. 27 - Both norepinephrine and epinephrine are...Ch. 27 - Prob. 27.22PCh. 27 - Draw a structural formula for the form of each...Ch. 27 - Prob. 27.24PCh. 27 - Write the zwitterion form of alanine and show its...Ch. 27 - Prob. 27.26PCh. 27 - Write the form of aspartic acid most prevalent at...Ch. 27 - Prob. 27.28PCh. 27 - Prob. 27.29PCh. 27 - For lysine and arginine, the isoelectric point,...Ch. 27 - Prob. 27.31PCh. 27 - Account for the fact that the isoelectric point of...Ch. 27 - Prob. 27.33PCh. 27 - Prob. 27.34PCh. 27 - At pH 7.4, the pH of blood plasma, do the majority...Ch. 27 - Prob. 27.36PCh. 27 - Prob. 27.37PCh. 27 - Prob. 27.38PCh. 27 - A chemically modified guanidino group is present...Ch. 27 - Draw a structural formula for the product formed...Ch. 27 - Prob. 27.41PCh. 27 - Prob. 27.42PCh. 27 - A decapeptide has the following amino acid...Ch. 27 - Following is the primary structure of glucagon, a...Ch. 27 - Prob. 27.45PCh. 27 - Draw a structural formula of these tripeptides....Ch. 27 - Estimate the pI of each tripeptide in Problem...Ch. 27 - Glutathione (G-SH), one of the most common...Ch. 27 - Following are a structural formula and a...Ch. 27 - Prob. 27.50PCh. 27 - Prob. 27.51PCh. 27 - Prob. 27.52PCh. 27 - Prob. 27.53PCh. 27 - Prob. 27.54PCh. 27 - Distinguish between intermolecular and...Ch. 27 - Prob. 27.56PCh. 27 - Prob. 27.57P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- At an electrified interface according to the Gouy-Chapman model, what types of interactions do NOT occur between the ions and the solvent according to this theory?arrow_forwardPlease predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers. Hint: In this case you must choose the best answer to demonstrate the stereochemistry of H2 addition. 1.03 2. (CH3)2S BIZ CH₂OH 2. DMS KMnO4, NaOH ΖΗ Pd or Pt (catalyst) HBr 20 1 HBr ROOR (peroxide) HO H-SO HC 12 11 10 BH, THE 2. H2O2, NaOH Brz cold HI 19 18 17 16 MCPBA 15 14 13 A Br H₂O BH3⚫THF Brz EtOH Pd or Ni (catalyst) D₂ (deuterium) 1. Os04 2. H2O2 CH3CO3H (peroxyacid) 1. MCPBA 2. H₂O* H B + H H H "H C H H Darrow_forwardExplain how Beer’s Law can be used to determine the concentration in a selected food sample. Provide examples.arrow_forward
- Explain the importance of having a sampling plan with respect to food analysis. Explain the importance of having a sampling plan with respect to food analysis. Provide examples.arrow_forwardPlease predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers. cold KMnO4, NaOH 2. DMS 1. 03 CH3OH Br2 1. 03 2. (CH3)2S H₂ Pd or Pt (catalyst) HBr 18 19 20 1 HBr ROOR (peroxide) H₂O H₂SO4 HCI HI 17 16 6 15 MCPBA 1. BH3 THF 2. H₂O2, NaOH 1. OsO4 2. H₂O₂ 110 CH3CO₂H (peroxyacid) 1. MCPBA 2. H₂O* Br2 H₂O BH3 THF B12 EtOH Pd or Ni (catalyst) D₂ (deuterium) Bra A B C D H OH H OH OH H OH α α α OH H OH OH фон d H "Harrow_forwardBriefly indicate the models that describe the structure of the interface: Helmholtz-Perrin, Gouy-Chapman, Stern and Grahame models.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHERChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHERChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

World of Chemistry
Chemistry
ISBN:9780618562763
Author:Steven S. Zumdahl
Publisher:Houghton Mifflin College Div

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY