
ORGANIC CHEMISTRY W/ALEKS
6th Edition
ISBN: 9781264905430
Author: SMITH
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 26, Problem 44P
Draw both pyranose anomers of each aldohexose using a three-dimensional representation with a chair pyranose. Label each anomer as
a. 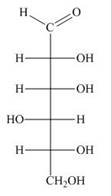 b.
b. 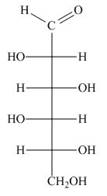
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Label each stereogenic center in the following compounds as R or S.
Labeling a Stereogenic Center as R or S
Label the stereogenic center in each compound as R or S.
Designating a Stereogenic Center as R or S When the Lowest-Priority Group Is Not Drawn Back
Label each stereogenic center as Ror S.
а.
OH
b.
OH
Chapter 26 Solutions
ORGANIC CHEMISTRY W/ALEKS
Ch. 26.2 - Prob. 1PCh. 26.2 - Prob. 2PCh. 26.2 - Label each stereogenic center as R or S. a. b. c....Ch. 26.2 - Convert the ball-and-stick model to a Fischer...Ch. 26.2 - Prob. 5PCh. 26.2 - Prob. 6PCh. 26.3 - Prob. 7PCh. 26.3 - Prob. 8PCh. 26.4 - Prob. 9PCh. 26.4 - Prob. 10P
Ch. 26.6 - Prob. 11PCh. 26.6 - Prob. 12PCh. 26.6 - Prob. 13PCh. 26.6 - Prob. 14PCh. 26.6 - Prob. 15PCh. 26.7 - Prob. 16PCh. 26.7 - Draw a stepwise mechanism for the following...Ch. 26.7 - Prob. 18PCh. 26.8 - Prob. 19PCh. 26.9 - Prob. 20PCh. 26.9 - Prob. 21PCh. 26.9 - Draw the products formed when D-arabinose is...Ch. 26.9 - Prob. 23PCh. 26.10 - Prob. 24PCh. 26.10 - Prob. 25PCh. 26.10 - Prob. 26PCh. 26.10 - Prob. 27PCh. 26.11 - Prob. 28PCh. 26.11 - Prob. 29PCh. 26.12 - Prob. 30PCh. 26.12 - Prob. 31PCh. 26.13 - Prob. 32PCh. 26.13 - Prob. 33PCh. 26.13 - Problem-28.35
Draw the structures of the...Ch. 26.13 - Prob. 35PCh. 26 - 28.37 Convert each ball-and-stick model to a...Ch. 26 - Prob. 37PCh. 26 - Prob. 38PCh. 26 - 28.40 Convert each compound to a Fischer...Ch. 26 - Prob. 40PCh. 26 - Prob. 41PCh. 26 - 28.43 Draw a Haworth projection for each compound...Ch. 26 - Prob. 43PCh. 26 - 28.45 Draw both pyranose anomers of each...Ch. 26 - Prob. 45PCh. 26 - 28.50 Draw the products formed when D-altrose is...Ch. 26 - 28.58 Draw a stepwise mechanism for the following...Ch. 26 - Prob. 62PCh. 26 - Prob. 63PCh. 26 - Prob. 64PCh. 26 - Prob. 65PCh. 26 - Prob. 66PCh. 26 - Prob. 67PCh. 26 - Prob. 68PCh. 26 - Prob. 69PCh. 26 - Prob. 70P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- How many stereoisomers are possible for a. a ketoheptose? b. an aldoheptose? c. a ketotriose?arrow_forwardKk.305.arrow_forwardProline is an unusual amino acid because its N atom on the α carbon is part of a fivemembered ring. a. Draw both enantiomers of proline. b. Draw proline in its zwitterionic form.arrow_forward
- D-Arabinose can exist in both pyranose and furanose forms. a. Draw the α and β anomers of D-arabinofuranose. b. Draw the α and β anomers of D-arabinopyranose.arrow_forwardFor D-arabinose:a. Draw its enantiomer.b. Draw an epimer at C3.c. Draw a diastereomer that is not an epimer.d. Draw a constitutional isomer that still contains a carbonyl group.arrow_forwardLocate the stereogenic centers in telaprevir, a drug used to treat hepatitis C, and label each stereogenic center as R or S.arrow_forward
- Penicillin is well-known anti-bacterial agent discovered by Alexander Fleming. A O. N- H. В Which box contains a carbocyclic unsaturation? IZarrow_forwardProline is an unusual amino acid because its N atom on the α carbon is part of a five-membered ring.a. Draw both enantiomers of proline. b. Draw proline in its zwitterionic form.arrow_forwardLabel each stereogenic center in the attached compounds as R or S.arrow_forward
- В. 1. Draw the Fischer, Haworth & chair (where applicable) representations of the given monosaccharides. Sugar Fischer Haworth Chair a CHO но H- H но OH ČH2OH D-idose (а-anomer) CH2OH FOH H- ČH2OH a ketopentose (B-anomer) b.arrow_forwardCaptopril is a drug used to treat high blood pressure and congestive heart failure. a.Designate each stereogenic center as R or S. b.Draw the enantiomer of captopril. c.What product is formed when captopril is treated with one equivalent of NaH? d.What product is formed when captopril is treated with two equivalents of NaH?arrow_forwardDraw each of the followingarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY