
EBK ORGANIC CHEMISTRY
7th Edition
ISBN: 9780133556186
Author: Bruice
Publisher: VST
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 25, Problem 67P
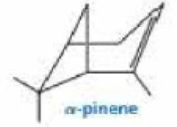
Propose a mechanism for the biosynthesis of α-pinene from farnesyl pyrophosphate.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the total energy cost associated with the compound below adopting the shown conformation?
CH3
HH
DH
CH3
ΗΝ,
Draw Final Product
C
cyclohexanone
pH 4-5
Edit Enamine
H3O+
CH3CH2Br
THF, reflux
H
Edit Iminium Ion
How many hydrogen atoms are connected to the indicated carbon atom?
Chapter 25 Solutions
EBK ORGANIC CHEMISTRY
Ch. 25.6 - Prob. 2PCh. 25.6 - Prob. 3PCh. 25.6 - Why does the OH group add to the -carbon rather...Ch. 25.6 - Prob. 5PCh. 25.6 - How many molecules of NADH are formed from the...Ch. 25.7 - Prob. 7PCh. 25.7 - Prob. 8PCh. 25.7 - The oxidation of glyceraldehyde-3-phosphate to...Ch. 25.7 - Prob. 10PCh. 25.8 - Prob. 11P
Ch. 25.8 - Prob. 12PCh. 25.8 - Prob. 13PCh. 25.8 - Propose a mechanism for the reduction of...Ch. 25.9 - Prob. 15PCh. 25.9 - Prob. 16PCh. 25.10 - Acid-catalyzed dehydration reactions are normally...Ch. 25.10 - Prob. 18PCh. 25.10 - Prob. 19PCh. 25.10 - Acid-catalyzed dehydration reactions are normally...Ch. 25.10 - Prob. 21PCh. 25.10 - Prob. 22PCh. 25.11 - Prob. 23PCh. 25.12 - a. What is the name of the enzyme that converts...Ch. 25.15 - Prob. 25PCh. 25.16 - Prob. 26PCh. 25.16 - Prob. 27PCh. 25.17 - Propose mechanisms for the Claisen condensation...Ch. 25.17 - Prob. 29PCh. 25.17 - Propose a mechanism for the conversion of...Ch. 25.17 - Propose a mechanism for the biosynthesis of...Ch. 25.17 - Propose a mechanism for the conversion of the E...Ch. 25.17 - The fluoro-substitued geranyl pyrophosphate shown...Ch. 25.17 - Prob. 35PCh. 25.18 - Draw the individual 1,2-hydride and 1,2-methyl...Ch. 25 - Prob. 38PCh. 25 - Prob. 39PCh. 25 - Prob. 40PCh. 25 - Prob. 41PCh. 25 - Prob. 42PCh. 25 - Prob. 43PCh. 25 - Prob. 44PCh. 25 - Prob. 45PCh. 25 - Prob. 46PCh. 25 - Prob. 47PCh. 25 - Prob. 48PCh. 25 - Prob. 49PCh. 25 - Prob. 50PCh. 25 - Prob. 51PCh. 25 - Prob. 52PCh. 25 - Prob. 53PCh. 25 - Prob. 54PCh. 25 - Prob. 55PCh. 25 - Prob. 56PCh. 25 - Prob. 57PCh. 25 - Prob. 58PCh. 25 - Prob. 59PCh. 25 - Prob. 60PCh. 25 - Prob. 61PCh. 25 - Prob. 62PCh. 25 - UDP-galactose-4-epimerase converts UDP-galactose...Ch. 25 - A student is trying to determine the mechanism for...Ch. 25 - What would be the results of the experiment in...Ch. 25 - Prob. 66PCh. 25 - Propose a mechanism for the biosynthesis of...Ch. 25 - Eudesmol is a sesquiterpene found in eucalyptus....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Identify the compound with the longest carbon - nitrogen bond. O CH3CH2CH=NH O CH3CH2NH2 CH3CH2C=N CH3CH=NCH 3 The length of all the carbon-nitrogen bonds are the samearrow_forwardIdentify any polar covalent bonds in epichlorohydrin with S+ and 8- symbols in the appropriate locations. Choose the correct answer below. Η H's+ 6Η Η Η Η Η Ηδ Η Ο Ο HH +Η Η +Η Η Η -8+ CIarrow_forwardH H:O::::H H H HH H::O:D:D:H HH HH H:O:D:D:H .. HH H:O:D:D:H H H Select the correct Lewis dot structure for the following compound: CH3CH2OHarrow_forward
- Rank the following compounds in order of decreasing boiling point. ннннн -С-С-Н . н-с- ННННН H ΗΤΗ НННН TTTĪ н-с-с-с-с-о-н НННН НН C' Н н-с-с-с-с-н НН || Ш НННН H-C-C-C-C-N-H ННННН IVarrow_forwardRank the following compounds in order of decreasing dipole moment. |>||>||| ||>|||>| |>|||>|| |||>||>| O ||>>||| H F H F H c=c || H c=c F F IIIarrow_forwardchoose the description that best describes the geometry for the following charged species ch3-arrow_forward
- Why isn't the ketone in this compound converted to an acetal or hemiacetal by the alcohol and acid?arrow_forwardWhat is the approximate bond angle around the nitrogen atom? HNH H Harrow_forwardOH 1. NaOCH2CH3 Q 2. CH3CH2Br (1 equiv) H3O+ Select to Draw 1. NaOCH2 CH3 2. CH3Br (1 equiv) heat Select to Edit Select to Drawarrow_forward
- Complete and balance the following half-reaction in acidic solution. Be sure to include the proper phases for all species within the reaction. S₂O₃²⁻(aq) → S₄O₆²⁻(aq)arrow_forwardQ Select to Edit NH3 (CH3)2CHCI (1 equiv) AICI 3 Select to Draw cat. H2SO4 SO3 (1 equiv) HO SOCl2 pyridine Select to Edit >arrow_forwardComplete and balance the following half-reaction in basic solution. Be sure to include the proper phases for all species within the reaction. Zn(s) → Zn(OH)₄²⁻(aq)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY