
Conceptual Physical Science (6th Edition)
6th Edition
ISBN: 9780134060491
Author: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 25, Problem 34TAS
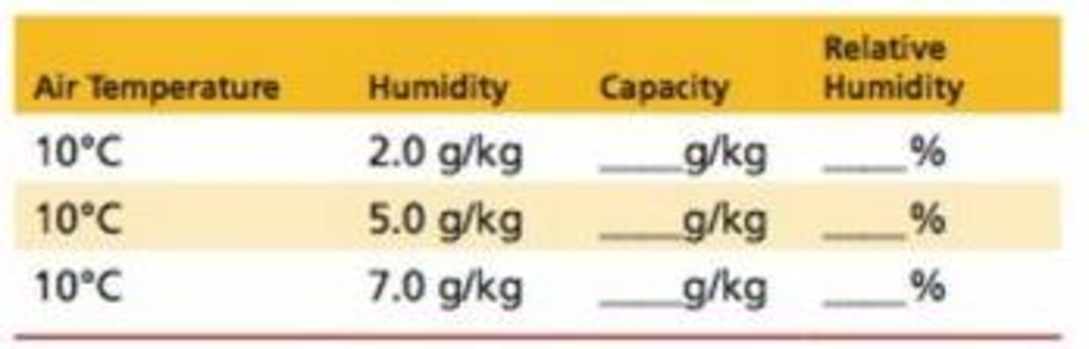
In this problem, temperature is held constant but humidity varies. Use Table 25.1 to find water-vapor capacity, and then use the formula for relative humidity to complete the following chart. What happens to relative humidity when water vapor is added? What happens when water vapor is removed?
Relative humidity = humidity /capacity × 100%
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
No chatgpt pls
No chatgpt pls will upvote
10. Inx 8.817
11.9.30 × 10-6
12.0.00500010
13.331,000,000
14.6.0005
15.pH=-log[H3O+} = 12.1830
Chapter 25 Solutions
Conceptual Physical Science (6th Edition)
Ch. 25 - What is the difference between humidity and...Ch. 25 - Why does relative humidity increase at night?Ch. 25 - As air temperature decreases, does relative...Ch. 25 - What does saturation point have to do with dew...Ch. 25 - What happens to the water vapor in saturated air...Ch. 25 - Prob. 6RCQCh. 25 - Prob. 7RCQCh. 25 - When water vapor condenses to liquid water, is...Ch. 25 - Prob. 9RCQCh. 25 - Explain why warm air rises and cools as it expands
Ch. 25 - When a parcel of air rises, does it become warmer,...Ch. 25 - Prob. 12RCQCh. 25 - Name at least two ways in which thermal energy in...Ch. 25 - Name at least two ways in which thermal energy in...Ch. 25 - Prob. 15RCQCh. 25 - Prob. 16RCQCh. 25 - Prob. 17RCQCh. 25 - Name the cloud form associated with (a) the hazy...Ch. 25 - Prob. 19RCQCh. 25 - Prob. 20RCQCh. 25 - Are clouds that have vertical development...Ch. 25 - Prob. 22RCQCh. 25 - Prob. 23RCQCh. 25 - Explain how a convection cycle is generated.Ch. 25 - Prob. 25RCQCh. 25 - Differentiate between a cold front and a warm...Ch. 25 - What are the three main atmospheric lifting...Ch. 25 - Under what conditions does orographic...Ch. 25 - How do downdrafts form in thunderstorms?Ch. 25 - Prob. 30RCQCh. 25 - What happens when water vapor in a can suddenly...Ch. 25 - Search the sky for passing jets anti look for the...Ch. 25 - At 50F the maximum amount of water vapor in air is...Ch. 25 - In this problem, temperature is held constant but...Ch. 25 - In this problem, humidity is held constant but...Ch. 25 - If the relative humidity of an air mass is 70% and...Ch. 25 - What are the airs temperature and the dew point...Ch. 25 - At sea level, an air parcel at 20C has a relative...Ch. 25 - On a mountain top (elevation 2 km), an air parcel...Ch. 25 - What is the difference between weather and...Ch. 25 - Why does dew form on the ground during dear, calm...Ch. 25 - Why does a July day in the Gulf of Mexico...Ch. 25 - Would you expect a glass of water to evaporate...Ch. 25 - During a summer visit to Cancun, Mexico, you stay...Ch. 25 - After a day of skiing in the Rocky Mountains, you...Ch. 25 - Why are saturation and condensation more likely to...Ch. 25 - Distinguish between dew and frost.Ch. 25 - What is the relationship between capacity and...Ch. 25 - When the condensation rate is greater than the...Ch. 25 - In which atmospheric layer does all our weather...Ch. 25 - Prob. 57ECh. 25 - What is the difference between specific humidity...Ch. 25 - What happens to relative humidity when temperature...Ch. 25 - Why does warm, moist air blowing over cold water...Ch. 25 - When compared to calm and clear nights, why are...Ch. 25 - Can the temperature of an air mass change if heat...Ch. 25 - When does an adiabatic process happen in the...Ch. 25 - Why do clouds tend to form above mountain peaks?Ch. 25 - Which air parcel will have the greater temperature...Ch. 25 - The density of air is generally given as mass per...Ch. 25 - Two air parcels with the same temperature (25C)...Ch. 25 - Prob. 68ECh. 25 - Give three ways in which heat can be added to an...Ch. 25 - Prob. 70ECh. 25 - Prob. 71ECh. 25 - Prob. 72ECh. 25 - What accounts for the large spaces of blue sky...Ch. 25 - Why dont cumulus clouds form over cool water?Ch. 25 - Prob. 75ECh. 25 - Prob. 76ECh. 25 - How can altostratus clouds change into altocumulus...Ch. 25 - Prob. 78ECh. 25 - Prob. 79ECh. 25 - Prob. 80ECh. 25 - Prob. 81ECh. 25 - Prob. 82ECh. 25 - Prob. 83ECh. 25 - Prob. 84ECh. 25 - In simplest terms, what is an occluded front?Ch. 25 - Prob. 86ECh. 25 - Prob. 87ECh. 25 - What is the name of the cold, dry air mass that...Ch. 25 - In the spring months, what happens when cold, dry...Ch. 25 - Sinking air warms, and yet the downdrafts in a...Ch. 25 - Prob. 91ECh. 25 - Prob. 92ECh. 25 - What is the source of the enormous amount of...Ch. 25 - In what part of the United States do tornadoes...Ch. 25 - On a broadcast news report, you hear that a...Ch. 25 - Prob. 96ECh. 25 - The accuracy of weather forecasts depends on great...Ch. 25 - Prob. 98ECh. 25 - What is an occluded front, and how is it indicated...Ch. 25 - Prob. 100DQCh. 25 - Clouds can act like a blanket around Earththey can...Ch. 25 - Prob. 102DQCh. 25 - Prob. 103DQCh. 25 - Prob. 104DQCh. 25 - Air that contains the maximum amount of water...Ch. 25 - In most midlatitude cyclones, the warm front (a)...Ch. 25 - Prob. 3RATCh. 25 - When air sinks, it (a) compresses and warms. (b)...Ch. 25 - When upper regions of the atmosphere are warmer...Ch. 25 - A key factor needed for precipitation to occur is...Ch. 25 - For clouds to form, air must be lifted. The...Ch. 25 - As air temperature decreases, relative humidity...Ch. 25 - In the Northern Hemisphere, tornadoes and...Ch. 25 - When air is saturated, the condensation rate (a)...
Additional Science Textbook Solutions
Find more solutions based on key concepts
The number of named species is about __________, but the actual number of species on Earth is estimated to be a...
Biology: Life on Earth (11th Edition)
Calculate the molarity of each solution. a. 22.6 g of C12H22O11 in 0.442 L of solution b. 42.6 g of NaCl in 1.5...
Introductory Chemistry (6th Edition)
Based on your answers to Questions 2 and 3, which part of the Atlantic basin appears to have opened first?
Applications and Investigations in Earth Science (9th Edition)
10.71 Identify each of the following as an acid or a base: (10.1)
H2SO4
RbOH
Ca(OH)2
HI
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
a. Draw the mechanism for the following reaction if it a involves specific-base catalysis. b. Draw the mechanis...
Organic Chemistry (8th Edition)
Where are skeletal cartilages located?
Human Anatomy & Physiology (2nd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Required information In a standard tensile test, a steel rod of 1 3 -in. diameter is subjected to a tension force of P = 21 kips. It is given that v= 0.30 and E= 29 × 106 psi. 1-in. diameter P P -8 in. Determine the change in diameter of the rod. (Round the final answer to six decimal places.) The change in diameter of the rod is - in.arrow_forward5.84 ... If the coefficient of static friction between a table and a uni- form, massive rope is μs, what fraction of the rope can hang over the edge of the table without the rope sliding? 5.97 Block A, with weight Figure P5.97 3w, slides down an inclined plane S of slope angle 36.9° at a constant speed while plank B, with weight w, rests on top of A. The plank is attached by a cord to the wall (Fig. P5.97). (a) Draw a diagram of all the forces acting on block A. (b) If the coefficient of kinetic friction is the same between A and B and between S and A, determine its value. B 36.9°arrow_forward5.60 An adventurous archaeologist crosses between two rock cliffs by slowly going hand over hand along a rope stretched between the cliffs. He stops to rest at the middle of the rope (Fig. P5.60). The rope will break if the tension in it exceeds 2.50 X 104 N, and our hero's mass is 90.0 kg. (a) If the angle is 10.0°, what is the tension in the rope? (b) What is the smallest value can have if the rope is not to break? Figure P5.60arrow_forward
- please answer the question thanks!arrow_forward5.48 ⚫ A flat (unbanked) curve on a highway has a radius of 170.0 m. A car rounds the curve at a speed of 25.0 m/s. (a) What is the minimum coefficient of static friction that will prevent sliding? (b) Suppose that the highway is icy and the coefficient of static friction between the tires and pavement is only one-third of what you found in part (a). What should be the maximum speed of the car so that it can round the curve safely?arrow_forward5.77 A block with mass m₁ is placed on an inclined plane with slope angle a and is connected to a hanging block with mass m₂ by a cord passing over a small, frictionless pulley (Fig. P5.74). The coef- ficient of static friction is μs, and the coefficient of kinetic friction is Mk. (a) Find the value of m₂ for which the block of mass m₁ moves up the plane at constant speed once it is set in motion. (b) Find the value of m2 for which the block of mass m₁ moves down the plane at constant speed once it is set in motion. (c) For what range of values of m₂ will the blocks remain at rest if they are released from rest?arrow_forward
- 5.78 .. DATA BIO The Flying Leap of a Flea. High-speed motion pictures (3500 frames/second) of a jumping 210 μg flea yielded the data to plot the flea's acceleration as a function of time, as shown in Fig. P5.78. (See "The Flying Leap of the Flea," by M. Rothschild et al., Scientific American, November 1973.) This flea was about 2 mm long and jumped at a nearly vertical takeoff angle. Using the graph, (a) find the initial net external force on the flea. How does it compare to the flea's weight? (b) Find the maximum net external force on this jump- ing flea. When does this maximum force occur? (c) Use the graph to find the flea's maximum speed. Figure P5.78 150 a/g 100 50 1.0 1.5 0.5 Time (ms)arrow_forward5.4 ⚫ BIO Injuries to the Spinal Column. In the treatment of spine injuries, it is often necessary to provide tension along the spi- nal column to stretch the backbone. One device for doing this is the Stryker frame (Fig. E5.4a, next page). A weight W is attached to the patient (sometimes around a neck collar, Fig. E5.4b), and fric- tion between the person's body and the bed prevents sliding. (a) If the coefficient of static friction between a 78.5 kg patient's body and the bed is 0.75, what is the maximum traction force along the spi- nal column that W can provide without causing the patient to slide? (b) Under the conditions of maximum traction, what is the tension in each cable attached to the neck collar? Figure E5.4 (a) (b) W 65° 65°arrow_forwardThe correct answers are a) 367 hours, b) 7.42*10^9 Bq, c) 1.10*10^10 Bq, and d) 7.42*10^9 Bq. Yes I am positve they are correct. Please dont make any math errors to force it to fit. Please dont act like other solutiosn where you vaugley state soemthing and then go thus, *correct answer*. I really want to learn how to properly solve this please.arrow_forward
- I. How many significant figures are in the following: 1. 493 = 3 2. .0005 = | 3. 1,000,101 4. 5.00 5. 2.1 × 106 6. 1,000 7. 52.098 8. 0.00008550 9. 21 10.1nx=8.817arrow_forwardplease solve and answer the question correctly please. Thank you!! (Hint in second photo)arrow_forwardplease solve and answer the question correctly please. Thank you!!arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
 Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Glencoe Physics: Principles and Problems, Student...
Physics
ISBN:9780078807213
Author:Paul W. Zitzewitz
Publisher:Glencoe/McGraw-Hill


Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY