
Concept explainers
Interpretation:
Using octadecanoic (stearic) acid and any necessary organic and inorganic reagents, an efficient synthesis for each compound is to be described.
Concept introduction:
The reduction of
The primary alcohol on oxidation with pyridinium dichromate
In Clemmensen reduction, the carbonyl group (aldehyde or
The esters can be synthesized by acid catalyzed condensation of carboxylic acid with alcohol.
The ester on reaction with one molar equivalent of Grignard’s reagent in diethyl ether gives ketone by carbon-carbon bond formation.
The ester on reaction with two molar equivalents of Grignard’s reagent in diethyl ether gives tertiary alcohol.
The dehydration of alcohol is the loss of
The alcohol on acid catalyzed dehydration gives corresponding alkene.
The alkene on hydrogenation with the catalyst undergoes addition of hydrogen across the double bond and forms an alkane.
The primary amine can be prepared by the acylation of ammonia.
The secondary amide can be prepared by the nucleophilic substitution of acyl chloride by amine. The two moles amines used with one mole of acyl chloride, because one amine molecule acts as a nucleophile and second acts as a Brønsted base.
The carboxylic acids on reaction with thionyl chloride forms acyl chloride by replacing the hydroxyl group of carboxylic acid with chlorine atom.
The primary amide on reduction with lithium aluminum hydride
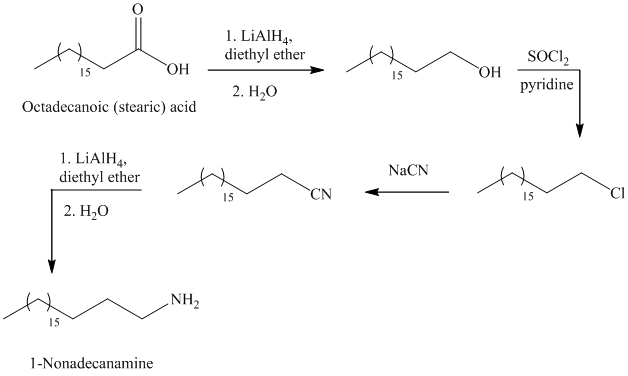
The reaction of thionyl chloride with alcohol gives alkyl halide.
The reaction of alkyl halide with sodium cyanide gives alkyl cyanide.
The cyanide (nitrile) can be reduced to primary amine using lithium aluminum hydride
The alkyl bromide can be prepared by the reaction of alcohol with phosphorus tribromide
Grignard reagents are prepared by the reaction of the magnesium metal with an alkyl or aryl halide usually in diethyl ether as the solvent.
The potassium or sodium dichromate in presence of strong acid forms chromic acid which is a good oxidizing agent, in hydrous medium oxidizes primary alcohol to carboxylic acid.
The epoxide on treatment with Grignard reagent undergoes epoxide ring opening by forming a corresponding alcohol.
Answer to Problem 28P
Solution:
a)
b)
c)
d) 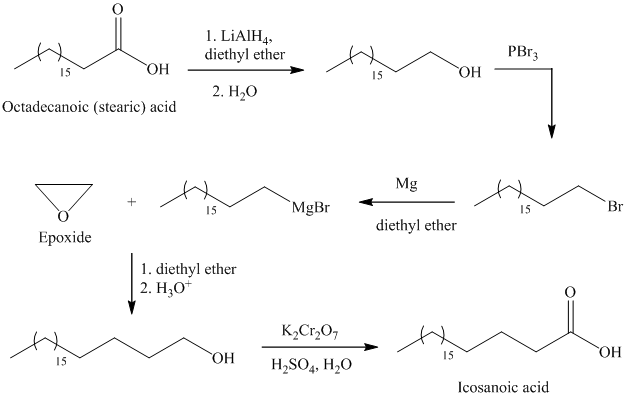
e) 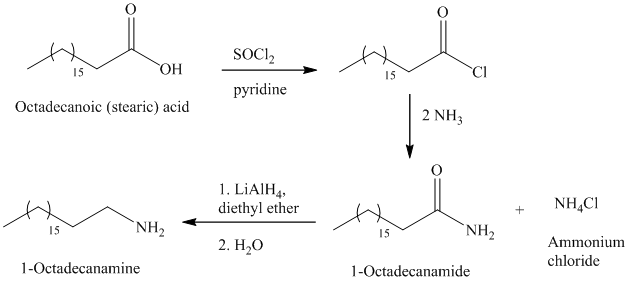
f)
Explanation of Solution
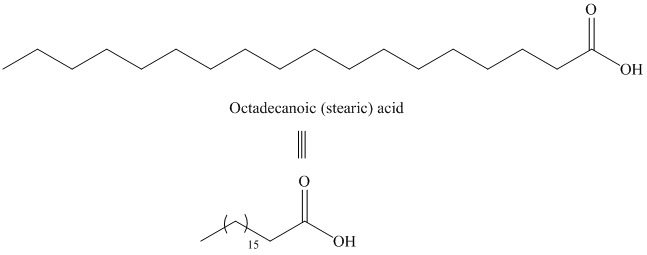
The structure of octadecanoic (stearic) acid is shown below:
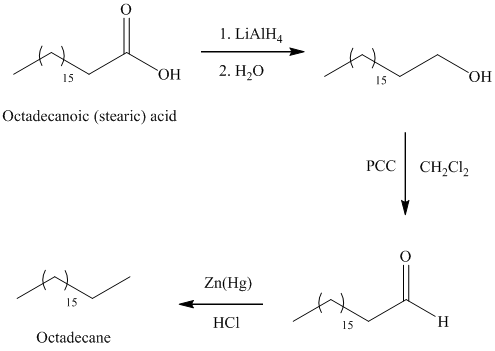
a) Octadecane
The synthesis of octadecane from octadecanoic acid can be done by following reactions sequence:
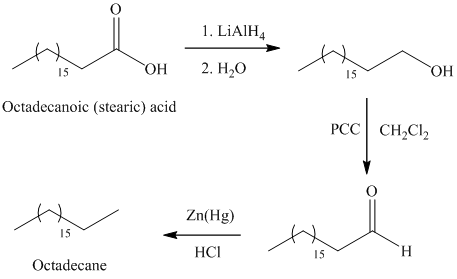
The octadecanoic acid on reaction with lithium aluminum hydride in aqueous medium reduced to octadecanol which is further on oxidation with pyridinium dichromate
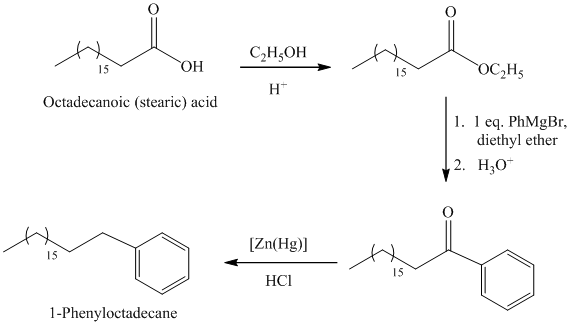
b)
The synthesis of
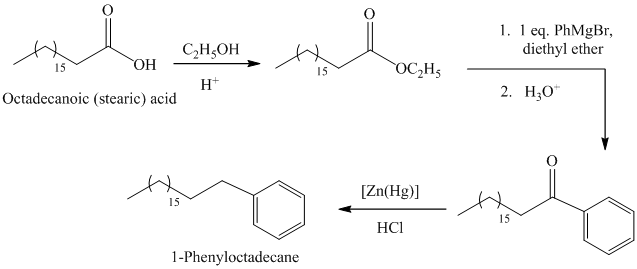
The octadecanoic acid first converted to an ester by reacting it with ethanol in acidic condition. The ester formed is then reacted with a Grignard’s reagent phenylmagnesium bromide
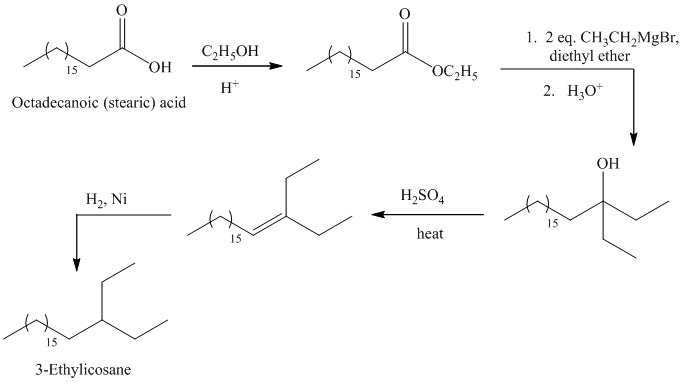
c)
The synthesis of
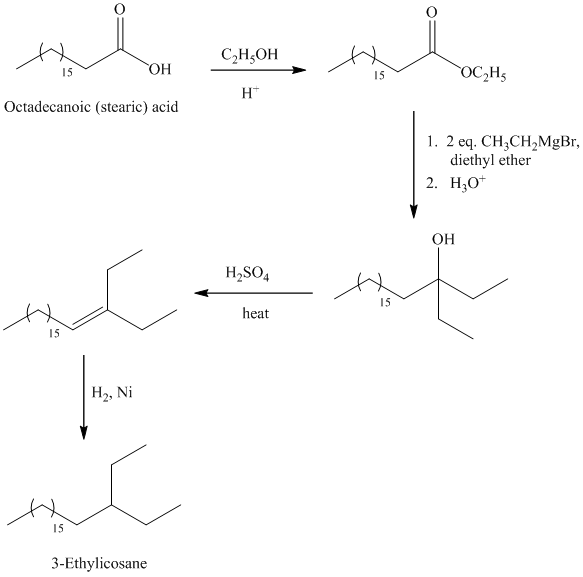
The octadecanoic acid is first converted to an ester by reacting it with ethanol in acidic condition. The ester formed is then reacted with a Grignard’s reagent ethyl bromide bromide
d) Icosanoic acid
The synthesis of Icosanoic acid from octadecanoic acid can be done by following reactions sequence:
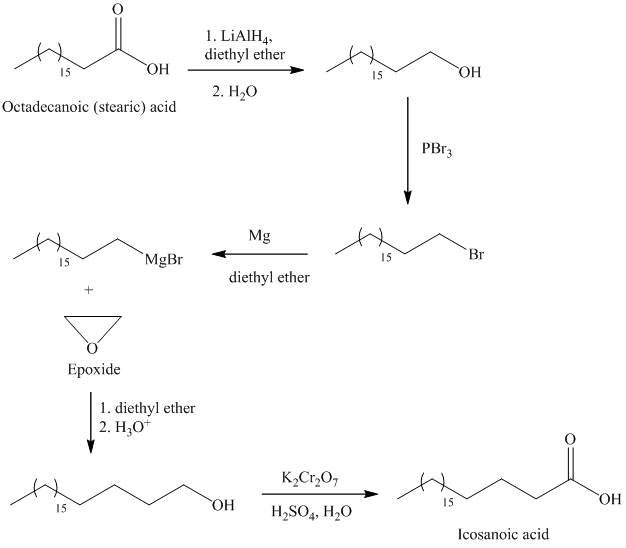
In the first step, the octadecanoic acid is reduced to primary alcohol by reducing agent lithium aluminum hydride
e)
The synthesis of
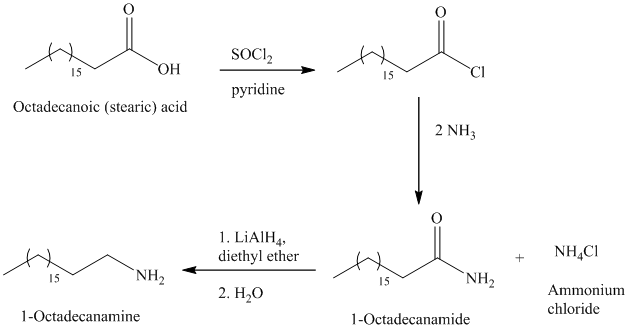
The octadecanoic acid on reaction with thonyl chloride in presence of pyridine gave the product of acyl chloride. The acyl chloride is converted to
f)
The synthesis of
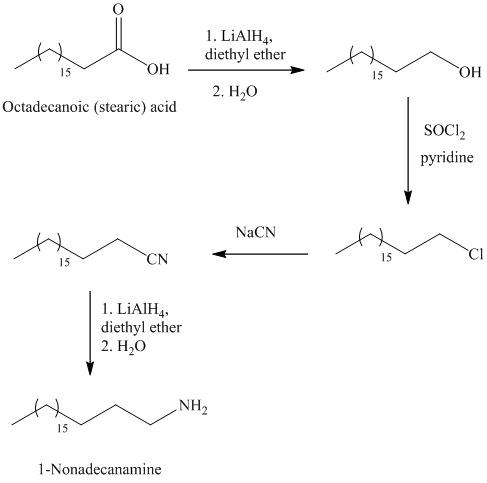
In the first step, the octadecanoic acid is reduced to primary alcohol by reducing agent lithium aluminum hydride
Want to see more full solutions like this?
Chapter 25 Solutions
ORGANIC CHEMISTRY-W/STUD.SOLN.MAN.
- Show the mechanism steps to obtain the lowerenergy intermediate: *see imagearrow_forwardSoap is made by the previous reaction *see image. The main difference between one soap and another soap isthe length (number of carbons) of the carboxylic acid. However, if a soap irritates your skin, they mostlikely used too much lye.Detergents have the same chemical structure as soaps except for the functional group. Detergentshave sulfate (R-SO4H) and phosphate (R-PO4H2) functional groups. Draw the above carboxylic acidcarbon chain but as the two variants of detergents. *see imagearrow_forwardWhat are the reactions or reagents used? *see imagearrow_forward
- The two pKa values of oxalic acid are 1.25 and 3.81. Why are they not the same value? Show the protontransfer as part of your explanation. *see imagearrow_forwardасть Identify all the bonds that gauche interact with C-OMe in the most stable conformation of the above compound.arrow_forwardPredict the reactants used in the formation of the following compounds using Acid-Catalyzed dehydration reactionarrow_forward
- Can I please get help with this?arrow_forward.. Give the major organic product(s) for each of the following reactions or sequences of reactions. Show ll relevant stereochemistry [3 ONLY]. A H Br 1. NaCN 2 NaOH, H₂O, heat 3. H3O+ B. CH₂COOH 19000 1. LiAlH4 THF, heat 2 H₂O* C. CH Br 1. NaCN, acetone 2 H3O+, heat D. Br 1. Mg. ether 3. H₂O+ 2 CO₂ E. CN 1. (CH) CHMgBr, ether 2 H₂O+arrow_forwardAssign this COSY spectrumarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





