
(a)
Interpretation:
How to synthesize a given compound from
Concept introduction:
Answer to Problem 25.76P
The synthesis of a given compound from
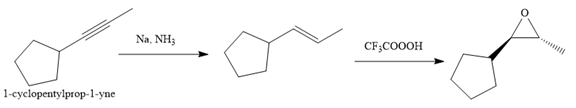
Explanation of Solution
The given synthesis is
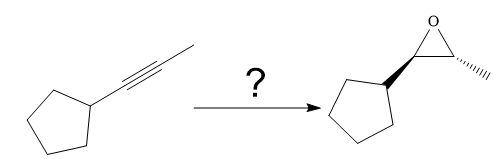
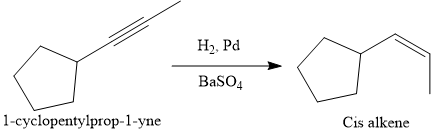
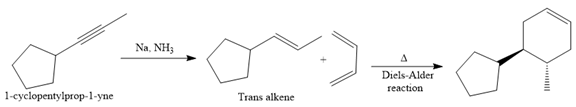
The direct conversion is not known. Therefore, the retrosynthesis analysis is done to know the possible route for the synthesis. It is noticed that the cyclopentyl ring and methyl group are trans to each other, thus the alkene results from
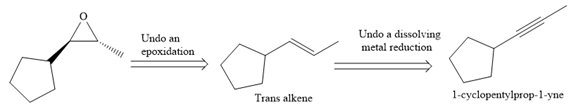
Thus the forward reaction is carried out as below:
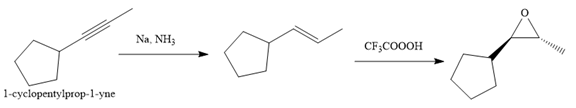
Since the cyclopentyl ring and methyl group are trans to each other, the alkene results from
It is shown how to synthesize a given compound from
(b)
Interpretation:
It is to be shown how to synthesize a given compound from
Concept introduction:
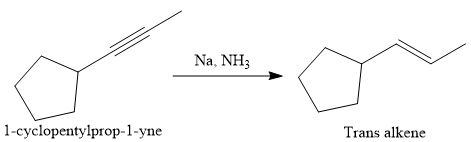
Alkynes can be converted to the corresponding alkene either by the treatment of alkali metal (e.g. Na, K) in ammonia or by catalytic hydrogenation. The advantage of the catalytic hydrogenation is to form the cis-alkene by the syn-addition of molecular hydrogen,
Answer to Problem 25.76P
The synthesis of the given compound from
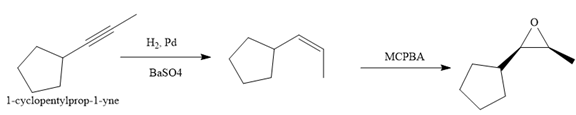
Explanation of Solution
The given synthesis is
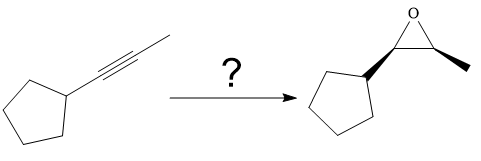
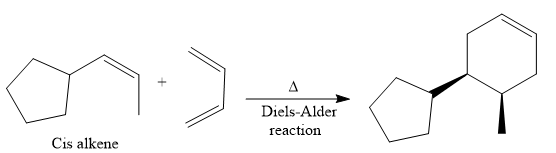
The direct conversion is not known. Therefore, the retrosynthesis analysis is done to know the possible route for the synthesis. It is noticed that the cyclopentyl ring and methyl group are cis to each other, thus the alkene results from
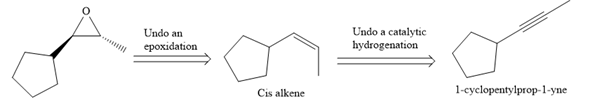
Thus the forward reaction is carried out as below:
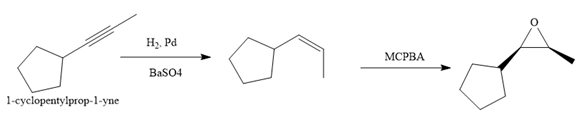
Since the cyclopentyl ring and methyl group are cis to each other, the alkene results from
It is shown how to synthesize a given compound from
(c)
Interpretation:
It is to be shown how to synthesize a given compound from
Concept introduction:
Alkynes can be converted to the corresponding alkene either by the treatment of alkali metal (e.g. Na, K) in ammonia called a dissolving metal reduction or by catalytic hydrogenation. The advantage of the catalytic hydrogenation is to form the cis-alkene by the syn-addition of molecular hydrogen,
Answer to Problem 25.76P
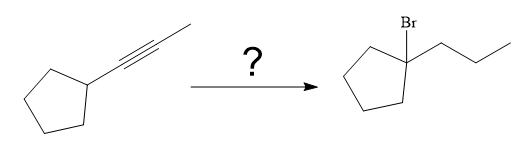
The synthesis of the given compound from
Explanation of Solution
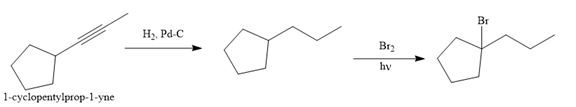
The given synthesis is
The direct conversion is not known. Therefore, the retrosynthesis analysis is done to know the possible route for the synthesis.
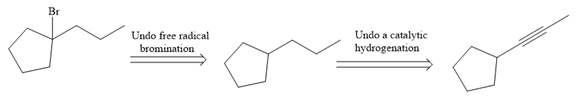
The forward reaction is carried out as:
In the first step the reduction of the alkyne is done to the corresponding alkane by the catalytic hydrogenation as follows:
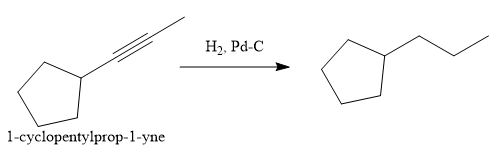
Finally, the proton of the tertiary carbon is replaced by bromine atom by the treatment of molecular bromine in the presence of light. The tertiary carbon is brominated via a free radical mechanism to form the required product.
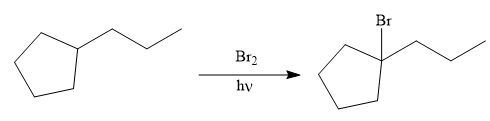
It is to be shown how to synthesize a given compound from
(d)
Interpretation:
How to synthesize a given compound from
Concept introduction:
Alkynes can be converted to the corresponding alkene either by the treatment of alkali metal (e.g. Na, K) in ammonia called a dissolving metal reduction or by catalytic hydrogenation. The advantage of the catalytic hydrogenation is to form the cis-alkene by the syn-addition of molecular hydrogen,
Answer to Problem 25.76P
The synthesis of the given compound from
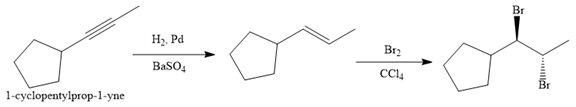
Explanation of Solution
The given synthesis is
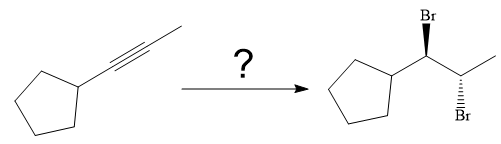
The direct conversion is not known. Therefore, the retrosynthesis analysis is done to know the possible route for the synthesis.
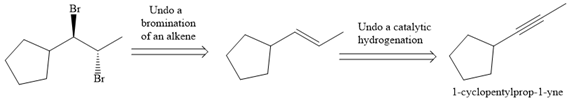
In the first step the reduction of the alkyne is done to the corresponding alkene by the catalytic hydrogenation as follows:
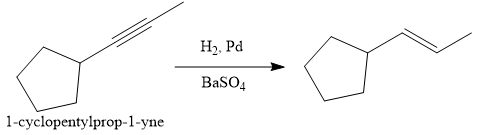
Finally, the addition of the molecular bromine is done to form the final, required product.
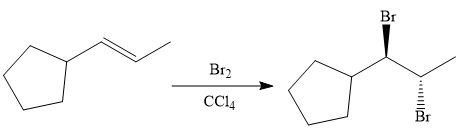
It is shown how to synthesize a given compound from
(e)
Interpretation:
How to synthesize a given compound from
Concept introduction:
Alkynes can be converted to the corresponding alkene either by the treatment of alkali metal (e.g. Na, K) in ammonia called a dissolving metal reduction or by catalytic hydrogenation. The advantage of the catalytic hydrogenation is to form the cis-alkene by the syn-addition of molecular hydrogen,
Answer to Problem 25.76P
The synthesis of the given compound from
Explanation of Solution
The given synthesis is
The direct conversion is not known. Therefore, the retrosynthesis analysis is done to know the possible route for the synthesis.
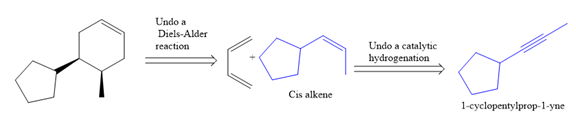
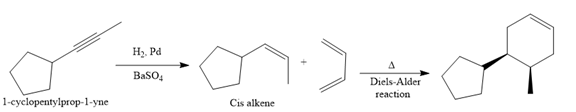
In the first step, the reduction of the alkyne is done to the corresponding alkene by the catalytic hydrogenation as follows:
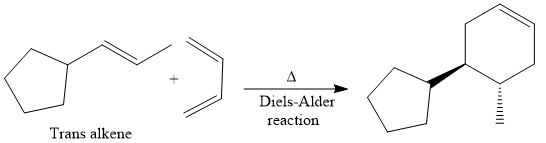
Finally, a Diels-Alder reaction is carried out to produce the required product.
It is shown how to synthesize a given compound from
(f)
Interpretation:
It is to be shown how to synthesize a given compound from
Concept introduction:
Alkynes can be converted to the corresponding alkene either by the treatment of alkali metal (e.g. Na, K) in ammonia called a dissolving metal reduction or by catalytic hydrogenation. The advantage of the catalytic hydrogenation is to form the cis-alkene by the syn-addition of molecular hydrogen,
Answer to Problem 25.76P
The synthesis of the given compound from
Explanation of Solution
The given synthesis is
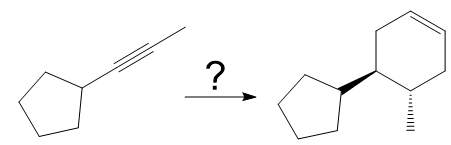
The direct conversion is not known. Therefore, the retrosynthesis analysis is done to know the possible route for the synthesis.
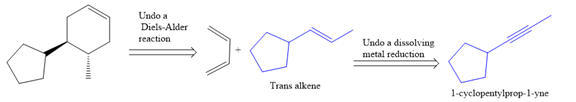
In the first step the reduction of the alkyne is done to the corresponding alkene by the catalytic hydrogenation as follows:
Finally, a Diels-Alder reaction is carried out to produce the required product.
It is shown how to synthesize a given compound from
Want to see more full solutions like this?
Chapter 25 Solutions
EBK ORGANIC CHEMISTRY: PRINCIPLES AND M
- 1. Part 1: Naming Organic Compounds он H₁C-C-CH3 CH3 Br CI CI 2. Br-CH-CH-CH₂ H₂C-CH-C= -CH-CH2-CH3 3. HC-CH-CH-C-OH 5. H₂C-CH-CH₂-OH 7. OH 4. CH CH₂-CH₂ 6. сно CH-CH-CH-CH₂-CH₂ H₁₂C-CH-CH-CH-CH₁₂-CH₁₂ 8. OHarrow_forward11 Organic Chemistry Organic Nomenclature Practice Name/Functional Group n-butane Formula Structural Formula (1) C4tt10 H3C C- (2) CH3CH2CH2 CH 3 H₂ -CH3 Н2 name & functional group (1) and (2) OH H₁₂C Н2 name only (1) and (2) name only (1) and (2) H₁C - = - CH₂ Н2 HC=C-C CH3arrow_forwardUnder aqueous basic conditions, nitriles will react to form a neutral organic intermediate 1 that has an N atom in it first, and then they will continue to react to form the final product 2: NC H₂O он- H₂O 1 2 OH Draw the missing intermediate 1 and the final product 2 in the box below. You can draw the two structures in any arrangement you like. Click and drag to start drawing a structure.arrow_forward
- Assign these COSY Spectrumarrow_forwardAssign these C-NMR and H-NMR Spectrumarrow_forwardPredict the product of this organic reaction: IZ + HO i P+H₂O Specifically, in the drawing area below draw the skeletal ("line") structure of P. If there is no reasonable possibility for P, check the No answer box under the drawing area. No Answer Click and drag to start drawing a structure. ☐ :arrow_forward
- Predict the products of this organic reaction: 0 O ----- A + KOH ? CH3-CH2-C-O-CH2-C-CH3 Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. (If there's more than one product, draw them in any arrangement you like, so long as they aren't touching.) If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No reaction Click anywhere to draw the first atom of your structure. X ⑤ èarrow_forwardPredict the products of this organic reaction: O CH3 + H2O + HCI A A? CH3-CH2-C-N-CH3 Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. If there's more than one product, draw them in any arrangement you like, so long as they aren't touching. If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No Reaction Click anywhere to draw the first atom of your structure.arrow_forwardWhat is the missing reactant in this organic reaction? R+ HO-C-CH2-CH3 0= CH3 CH3 —CH, C−NH—CH CH3 + H₂O Specifically, in the drawing area below draw the condensed structure of R. If there is more than one reasonable answer, you can draw any one of them. If there is no reasonable answer, check the No answer box under the drawing area. Note for advanced students: you may assume no products other than those shown above are formed. No Answer Click anywhere to draw the first atom of your structure. €arrow_forward
- 个 CHEM&131 9267 - $25 - Intro to Mail - Hutchison, Allison (Student x Aktiv Learnin https://app.aktiv.com Draw the product of the reaction shown below. Ignore inorganic byproducts. + Na2Cr2O7 Acetone, H2SO4 Type here to search Dryng OH W Prarrow_forwardPredict the products of this organic reaction: OH + NaOH A? Specifically, in the drawing area below draw the skeletal ("line") structure of the product, or products, of this reaction. (If there's more than one product, draw them in any arrangement you like, so long as they aren't touching.) If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No reaction Click and drag to start drawing a structure. ✓ Sarrow_forwardPredict the products of this organic reaction: CH3-C-O-CH2-CH2-C-CH3 + H₂O ? A Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. (If there's more than one product, draw them in any arrangement you like, so long as they aren't touching.) If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No reaction Click anywhere to draw the first atom of your structure. :☐ darrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





