
(a)
Interpretation:
How to synthesize a given compound from
Concept introduction:
Answer to Problem 25.76P
The synthesis of a given compound from
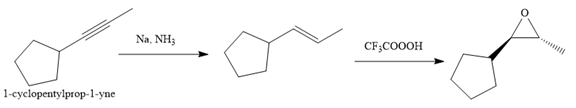
Explanation of Solution
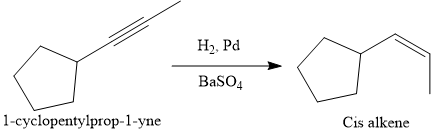
The given synthesis is
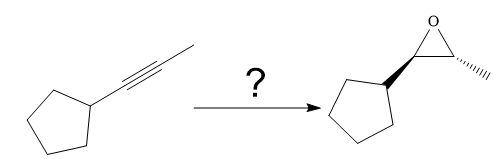
The direct conversion is not known. Therefore, the retrosynthesis analysis is done to know the possible route for the synthesis. It is noticed that the cyclopentyl ring and methyl group are trans to each other, thus the alkene results from
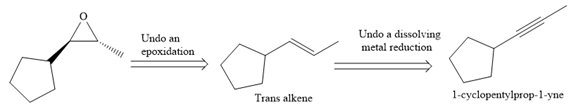
Thus the forward reaction is carried out as below:
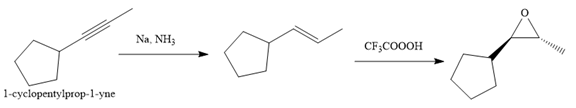
Since the cyclopentyl ring and methyl group are trans to each other, the alkene results from
It is shown how to synthesize a given compound from
(b)
Interpretation:
It is to be shown how to synthesize a given compound from
Concept introduction:
Alkynes can be converted to the corresponding alkene either by the treatment of alkali metal (e.g. Na, K) in ammonia or by catalytic hydrogenation. The advantage of the catalytic hydrogenation is to form the cis-alkene by the syn-addition of molecular hydrogen,
Answer to Problem 25.76P
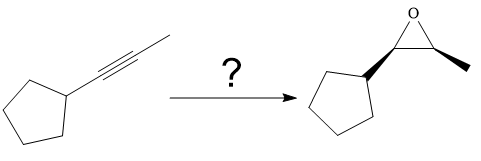
The synthesis of the given compound from
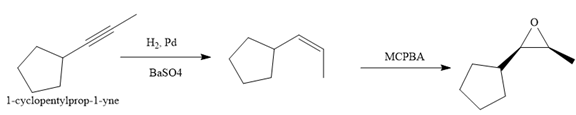
Explanation of Solution
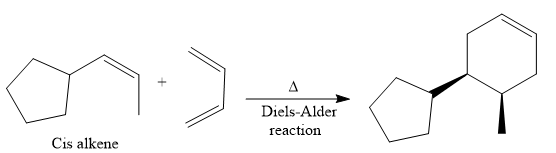
The given synthesis is
The direct conversion is not known. Therefore, the retrosynthesis analysis is done to know the possible route for the synthesis. It is noticed that the cyclopentyl ring and methyl group are cis to each other, thus the alkene results from
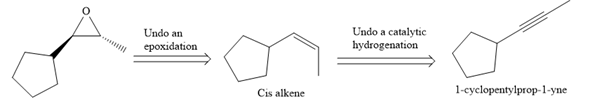
Thus the forward reaction is carried out as below:
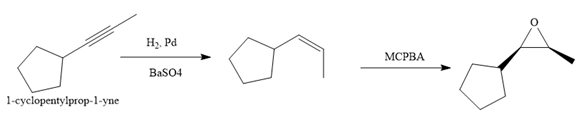
Since the cyclopentyl ring and methyl group are cis to each other, the alkene results from
It is shown how to synthesize a given compound from
(c)
Interpretation:
It is to be shown how to synthesize a given compound from
Concept introduction:
Alkynes can be converted to the corresponding alkene either by the treatment of alkali metal (e.g. Na, K) in ammonia called a dissolving metal reduction or by catalytic hydrogenation. The advantage of the catalytic hydrogenation is to form the cis-alkene by the syn-addition of molecular hydrogen,
Answer to Problem 25.76P
The synthesis of the given compound from
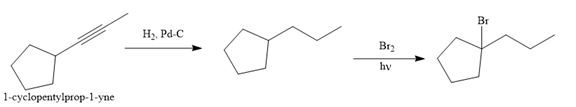
Explanation of Solution
The given synthesis is
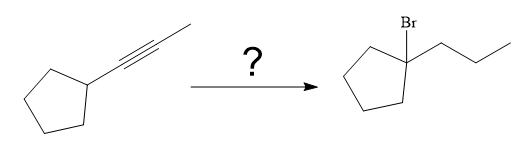
The direct conversion is not known. Therefore, the retrosynthesis analysis is done to know the possible route for the synthesis.
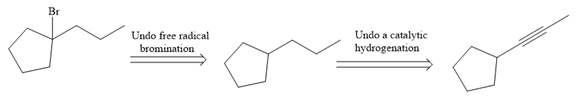
The forward reaction is carried out as:
In the first step the reduction of the alkyne is done to the corresponding alkane by the catalytic hydrogenation as follows:
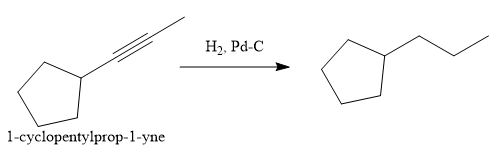
Finally, the proton of the tertiary carbon is replaced by bromine atom by the treatment of molecular bromine in the presence of light. The tertiary carbon is brominated via a free radical mechanism to form the required product.
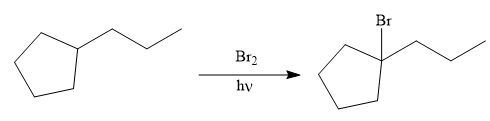
It is to be shown how to synthesize a given compound from
(d)
Interpretation:
How to synthesize a given compound from
Concept introduction:
Alkynes can be converted to the corresponding alkene either by the treatment of alkali metal (e.g. Na, K) in ammonia called a dissolving metal reduction or by catalytic hydrogenation. The advantage of the catalytic hydrogenation is to form the cis-alkene by the syn-addition of molecular hydrogen,
Answer to Problem 25.76P
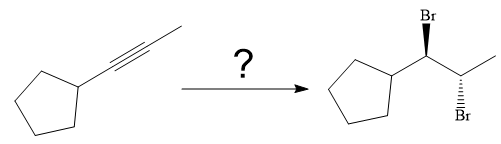
The synthesis of the given compound from
Explanation of Solution
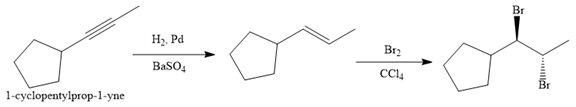
The given synthesis is
The direct conversion is not known. Therefore, the retrosynthesis analysis is done to know the possible route for the synthesis.
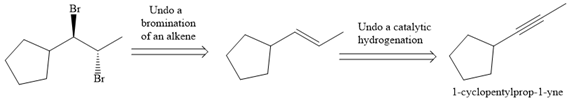
In the first step the reduction of the alkyne is done to the corresponding alkene by the catalytic hydrogenation as follows:
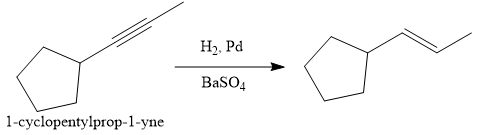
Finally, the addition of the molecular bromine is done to form the final, required product.
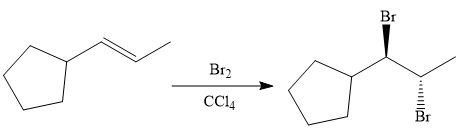
It is shown how to synthesize a given compound from
(e)
Interpretation:
How to synthesize a given compound from
Concept introduction:
Alkynes can be converted to the corresponding alkene either by the treatment of alkali metal (e.g. Na, K) in ammonia called a dissolving metal reduction or by catalytic hydrogenation. The advantage of the catalytic hydrogenation is to form the cis-alkene by the syn-addition of molecular hydrogen,
Answer to Problem 25.76P
The synthesis of the given compound from
Explanation of Solution
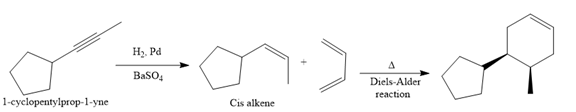
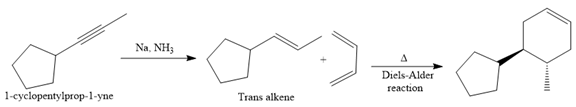
The given synthesis is
The direct conversion is not known. Therefore, the retrosynthesis analysis is done to know the possible route for the synthesis.
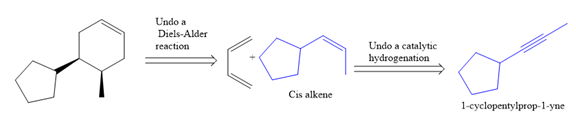
In the first step, the reduction of the alkyne is done to the corresponding alkene by the catalytic hydrogenation as follows:
Finally, a Diels-Alder reaction is carried out to produce the required product.
It is shown how to synthesize a given compound from
(f)
Interpretation:
It is to be shown how to synthesize a given compound from
Concept introduction:
Alkynes can be converted to the corresponding alkene either by the treatment of alkali metal (e.g. Na, K) in ammonia called a dissolving metal reduction or by catalytic hydrogenation. The advantage of the catalytic hydrogenation is to form the cis-alkene by the syn-addition of molecular hydrogen,
Answer to Problem 25.76P
The synthesis of the given compound from
Explanation of Solution
The given synthesis is
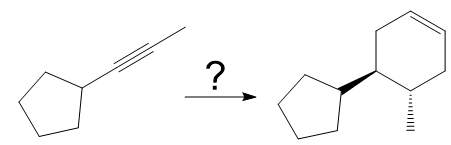
The direct conversion is not known. Therefore, the retrosynthesis analysis is done to know the possible route for the synthesis.
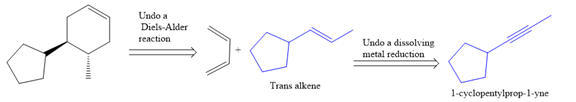
In the first step the reduction of the alkyne is done to the corresponding alkene by the catalytic hydrogenation as follows:
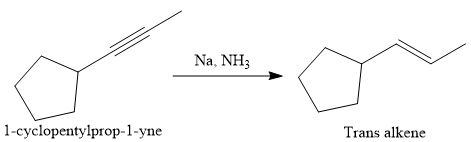
Finally, a Diels-Alder reaction is carried out to produce the required product.
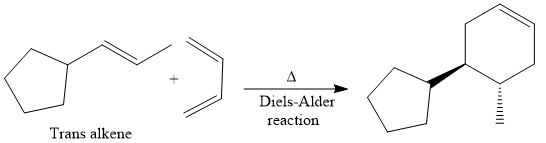
It is shown how to synthesize a given compound from
Want to see more full solutions like this?
Chapter 25 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- Predict the major organic product for this reaction.arrow_forwardWhat are the missing reagents for the spots labeled 1 and 3? Please give a detailed explanation and include the drawings and show how the synthesis proceeds with the reagents.arrow_forwardPlease provide the complete mechanism for the reaction below and include all appropriate arrows, formal charges, and intermediates. Please draw out the answerarrow_forward
- 3) The following molecule, chloral is a common precursor to chloral hydrate, an acetal type molecule that was a first-generation anesthetic. Draw a mechanism that accounts for tis formation and speculate why it does not require the use of an acid catalyst, like most hemiacetal and acetal reaction: (10 pts) H H₂Oarrow_forwardYou are a Quality Manager for a very well-known food ingredient company that produces umami powder, and you are responsible for setting specification limits. The net weight (in grams) of bags of unami powder is monitored by taking samples of six bags on an hourly basis during production. The label on every bag reports a contents of 1KG umami powder. The process mean is μ = 1012 g, and when the process is properly adjusted, it varies with σ = 11 g. QUESTION: Your organisation strives to ensure that >99.97% of bags of umami powder produced conforms to specification. What performance process index value is required to achieve this process yield? Calculate PPK using the following formula: Ppk = (USL – mean)/3 σ Ppk = (mean -LSL)/ 3 σarrow_forwardYou are a Quality Manager for a very well-known food ingredient company that produces umami powder, and you are responsible for setting specification limits. The net weight (in grams) of bags of unami powder is monitored by taking samples of six bags on an hourly basis during production. The label on every bag reports a contents of 1KG umami powder. The process mean is μ = 1012 g, and when the process is properly adjusted, it varies with σ = 11 g. QUESTION: Provide a valid and full justification as to whether you would advise your manager that the process is satisfactory when it is properly adjusted, or would you seek their approval to improve the process?arrow_forward
- You are a Quality Manager for a very well-known food ingredient company that produces umami powder, and you are responsible for setting specification limits. The net weight (in grams) of bags of unami powder is monitored by taking samples of six bags on an hourly basis during production. The label on every bag reports a contents of 1KG umami powder. The process mean is μ = 1012 g, and when the process is properly adjusted, it varies with σ = 11 g. QUESTION: Using all the available information, set the upper and lower specification limits.arrow_forward43) 10.00 ml of vinegar (active ingredient is acetic acid) is titrated to the endpoint using 19.32 ml of 0.250 M sodium hydroxide. What is the molarity of acetic acid in the vinegar? YOU MUST SHOW YOUR WORK. NOTE: MA x VA = MB x VBarrow_forward424 Repon Sheet Rates of Chemical Reactions : Rate and Order of 1,0, Deception B. Effect of Temperature BATH TEMPERATURE 35'c Yol of Oh نام Time 485 Buret rend ing(n) 12 194 16. 6 18 20 10 22 24 14 115 95 14738 2158235 8:26 CMS 40148 Total volume of 0, collected Barometric pressure 770-572 ml mm Hg Vapor pressure of water at bath temperature (see Appendix L) 42.2 Slope Compared with the rate found for solution 1, there is Using the ideal gas law, calculate the moles of O; collected (show calculations) times faster 10 Based on the moles of O, evolved, calculate the molar concentration of the original 3% 1,0, solution (sho calculations)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





