
Organic Chemistry-Package(Custom)
4th Edition
ISBN: 9781259141089
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 25, Problem 25.73P
Tertiary
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Calculate the variation in the potential of the Pt/MnO4-, Mn2+ pair with pH, indicating the value of the standard potential. Data: E0 = 1.12.
Given the cell: Pt l H2(g) l dis X:KCl (sat) l Hg2Cl2(s) l Hg l Pt. Calculate the emf of the cell as a function of pH.
The decimolar calomel electrode has a potential of 0.3335 V at 25°C compared to the standard hydrogen electrode. If the standard reduction potential of Hg22+ is 0.7973 V and the solubility product of Hg2Cl2 is 1.2x 10-18, find the activity of the chlorine ion at this electrode.Data: R = 8.314 J K-1 mol-1, F = 96485 C mol-1, T = 298.15 K.
Chapter 25 Solutions
Organic Chemistry-Package(Custom)
Ch. 25 - Prob. 25.1PCh. 25 - Draw the structure of a compound of molecular...Ch. 25 - Prob. 25.3PCh. 25 - Prob. 25.4PCh. 25 - Prob. 25.5PCh. 25 - Arrange the compounds in order of increasing...Ch. 25 - Prob. 25.7PCh. 25 - Prob. 25.8PCh. 25 - Problem 25.7
Draw the product of each...Ch. 25 - Prob. 25.10P
Ch. 25 - Prob. 25.11PCh. 25 - Prob. 25.12PCh. 25 - Prob. 25.13PCh. 25 - Prob. 25.14PCh. 25 - Prob. 25.15PCh. 25 - Prob. 25.16PCh. 25 - Prob. 25.17PCh. 25 - Prob. 25.18PCh. 25 - Draw the products of each acidbase reaction....Ch. 25 - Prob. 25.20PCh. 25 - Prob. 25.21PCh. 25 - Prob. 25.22PCh. 25 - Prob. 25.23PCh. 25 - Problem 25.22
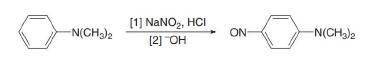
Which nitrogen atom in each compound...Ch. 25 - Which N atom in each drug is more basic? a. b. c.Ch. 25 - Prob. 25.26PCh. 25 - Prob. 25.27PCh. 25 - Prob. 25.28PCh. 25 - Prob. 25.29PCh. 25 - Prob. 25.30PCh. 25 - Problem 25.28
Draw the major product formed in...Ch. 25 - Prob. 25.32PCh. 25 - Prob. 25.33PCh. 25 - Problem 25.31
Devise a synthesis of each compound...Ch. 25 - Prob. 25.35PCh. 25 - Problem 25.33
What starting materials are needed...Ch. 25 - Problem 25.34
(a) What two components are needed...Ch. 25 - Prob. 25.38PCh. 25 - Prob. 25.39PCh. 25 - Prob. 25.40PCh. 25 - 25.37 Varenicline (trade name Chantix) is a drug...Ch. 25 - Prob. 25.42PCh. 25 - Prob. 25.43PCh. 25 - Prob. 25.44PCh. 25 - Prob. 25.45PCh. 25 - Prob. 25.46PCh. 25 - Prob. 25.47PCh. 25 - Prob. 25.48PCh. 25 - Decide which N atom in each molecule is most basic...Ch. 25 - 25.44 Rank the nitrogen atoms in each compound in...Ch. 25 - 25.45 Explain why nitroaniline is a stronger base...Ch. 25 - 25.46 Explain the observed difference in the...Ch. 25 - 25.47 Why is pyrrole more acidic than...Ch. 25 - How would you prepare 3-phenyl-1-propanamine...Ch. 25 - Prob. 25.55PCh. 25 - Prob. 25.56PCh. 25 - Prob. 25.57PCh. 25 - Prob. 25.58PCh. 25 - 25.51 How would you separate toluene , benzoic...Ch. 25 - 25.52 Draw the products formed when methylaniline ...Ch. 25 - Prob. 25.61PCh. 25 - Prob. 25.62PCh. 25 - Prob. 25.63PCh. 25 - Prob. 25.64PCh. 25 - Prob. 25.65PCh. 25 - Prob. 25.66PCh. 25 - Prob. 25.67PCh. 25 - Prob. 25.68PCh. 25 - 25.60 A chiral amine A having the configuration...Ch. 25 - 25.61 Draw a stepwise mechanism for each...Ch. 25 - 25.62 Draw a stepwise mechanism for the following...Ch. 25 - Prob. 25.72PCh. 25 - Tertiary (3) aromatic amines react with NaNO2 and...Ch. 25 - Prob. 25.74PCh. 25 - Devise a synthesis of each compound from benzene....Ch. 25 - Prob. 25.76PCh. 25 - Prob. 25.77PCh. 25 - Prob. 25.78PCh. 25 - Prob. 25.79PCh. 25 - Synthesize each compound from benzene. Use a...Ch. 25 - Prob. 25.81PCh. 25 - Devise a synthesis of each compound from benzene,...Ch. 25 - Prob. 25.83PCh. 25 - 25.72 Three isomeric compounds A, B, and C, all...Ch. 25 - 25.73 Treatment of compound D with LiAlH4 followed...Ch. 25 - Prob. 25.86PCh. 25 - 25.75 Rank the following compounds in order of...Ch. 25 - Prob. 25.88PCh. 25 - Prob. 25.89PCh. 25 - Prob. 25.90P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2. Add the following group of numbers using the correct number of significant figures for the answer. Show work to earn full credit such as rounding off the answer to the correct number of significant figures. Replace the question marks with the calculated answers or write the calculated answers near the question marks. 10916.345 37.40832 5.4043 3.94 + 0.0426 ? (7 significant figures)arrow_forwardThe emf at 25°C of the cell: Pt l H2(g) l dis X:KCl (sat) l Hg2Cl2(s) l Hg l Pt was 612 mV. When solution X was replaced by normal phosphate buffer solution with a pH of 6.86, the emf was 741 mV. Calculate the pH of solution X.arrow_forwardIndicate how to calculate the potential E of the reaction Hg2Cl2(s) + 2e ⇄ 2Hg + 2Cl- as a function of the concentration of Cl- ions. Data: the solubility product of Hg2Cl2.arrow_forward
- How can Beer’s Law be used to determine the concentration in a selected food sample. Provide an in-depth discussion and examples of this.arrow_forwardb) H3C- H3C Me CH 3 I HN Me H+arrow_forwardUsing Luther's rule, determine the reference potentials of the electrodes corresponding to the low stability systems Co³+/Co and Cr²+/Cr from the data in the table. Electrodo ΕΝ Co²+/Co Co3+/Co²+ -0,28 +1,808 Cr³+ / Cr -0,508 Cr3+ / Cr²+ -0,41arrow_forward
- The molecule PYRIDINE, 6tt electrons and is there pore aromuntre and is Assigned the Following structure contenus Since aromatk moleculey undergo electrophilic allomatic substitution, Pyridine should undergo The Following reaction + HNO3 12504 a. write all of the possible Mononitration Products that could Result From this roaction Based upon the reaction the reaction mechanism determine which of these producty would be the major Product of the hegetionarrow_forwardUsing Benzene as starting materia Show how each of the Following molecules could Ve synthesked 9. CHI d. 10450 b 0 -50311 ८ City -5034 1-0-650 e NO2arrow_forwardBA HBr of the fol 1)=MgCI 2) H₂O major NaOEt Ts Cl Py (pyridine) 1) 03 2) Me2S 1arrow_forward
- 4. Provide a clear arrow-pushing mechanism for the following reactions. Do not skip proton transfers, do not combine steps, and make sure your arrows are clear enough to be interpreted without ambiguity. a) NHBoc ⚫OBn HO. H3C CO2CH3 -OBn H3C H3C. H3C. NHBOC CI CO2CH3arrow_forwardDraw structures of the following compounds and identify their role: mCPBA (MCPBA) DMS Py 9-BBN LAH Sia₂BH TsCI PCC t-BuOK LDA MeLi n-BuLi DMSO DMF Sodium Borohydride Lithium DiisopropylAmide 2arrow_forwardUsing Luther's rule, calculate the reference potential of the Hg2+/Hg redox electrode. DATA: Electrode potentials E° = 0,854 V y E 0,788 V Hg2+/Hg 2+ Hg2/Hgarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License