
CHEMISTRY 1111 LAB MANUAL >C<
1st Edition
ISBN: 9781307092097
Author: Chang
Publisher: MCG/CREATE
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 25, Problem 25.11QP
(a)
Interpretation Introduction
Interpretation:
Plausible monomer for the given
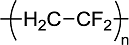
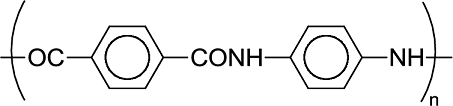
(b)
Interpretation Introduction
Interpretation:
Plausible monomer for the given polymer should be identified.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Can I get some help drawing my arrows. I included what the final needs to look like
please help
(a)
(e)
O₂N.
(h)
21.8 Name the following compounds.
Br
(f)
Ph.
(c)
(d)
Br
(g)
NO₂
H
NH2
Br
mo. 0-0.
OMe
(i)
Chapter 25 Solutions
CHEMISTRY 1111 LAB MANUAL >C<
Ch. 25 - Prob. 25.1QPCh. 25 - Prob. 25.2QPCh. 25 - Prob. 25.3QPCh. 25 - Prob. 25.4QPCh. 25 - Prob. 25.5QPCh. 25 - Prob. 25.6QPCh. 25 - Prob. 25.7QPCh. 25 - Prob. 25.8QPCh. 25 - Prob. 25.9QPCh. 25 - Prob. 25.10QP
Ch. 25 - Prob. 25.11QPCh. 25 - Prob. 25.12QPCh. 25 - Prob. 25.13QPCh. 25 - Prob. 25.14QPCh. 25 - Prob. 25.15QPCh. 25 - Prob. 25.16QPCh. 25 - Prob. 25.17QPCh. 25 - Prob. 25.18QPCh. 25 - Prob. 25.19QPCh. 25 - Prob. 25.20QPCh. 25 - Prob. 25.21QPCh. 25 - Prob. 25.22QPCh. 25 - Prob. 25.23QPCh. 25 - Prob. 25.24QPCh. 25 - Prob. 25.25QPCh. 25 - Prob. 25.26QPCh. 25 - Discuss the importance of hydrogen bonding in...Ch. 25 - Proteins vary widely in structure, whereas nucleic...Ch. 25 - Prob. 25.29QPCh. 25 - Prob. 25.30QPCh. 25 - Prob. 25.31QPCh. 25 - Prob. 25.32QPCh. 25 - Prob. 25.33QPCh. 25 - Prob. 25.34QPCh. 25 - Prob. 25.35QPCh. 25 - Chemical analysis shows that hemoglobin contains...Ch. 25 - Prob. 25.37QPCh. 25 - What kind of intermolecular forces are responsible...Ch. 25 - Draw structures of the nucleotides containing the...Ch. 25 - Prob. 25.40QPCh. 25 - Prob. 25.41QPCh. 25 - Prob. 25.42QPCh. 25 - Prob. 25.43QPCh. 25 - Prob. 25.44QPCh. 25 - When deoxyhemoglobin crystals are exposed to...Ch. 25 - Prob. 25.46QPCh. 25 - Prob. 25.47QPCh. 25 - Prob. 25.48QPCh. 25 - Prob. 25.49QPCh. 25 - Prob. 25.50QPCh. 25 - Prob. 25.51QPCh. 25 - Assume the energy of hydrogen bonds per base pair...Ch. 25 - Prob. 25.53IME
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Can I get helpp drawing my arrowsarrow_forwardWhich of the m/z values corresponds to the base peak in the mass spectrum shown? 100 80 A. 45 B. 44 C. 29 D. 15 Intensity 20 0 10 20 30 40 B- m/z -8 50 E. 30 Which of the m/z values correspond to the molecular ion for the compound shown? A. 18 B. 82 OH C. 100 D. 102 E. 103arrow_forwardCan someone help me with drawing my arrows.arrow_forward
- I'm having trouble with converting lewis diagrams into VSEPR diagrams. I currently have this example of C2BrCl3 which I want to turn into a lewis structure, but I'm not sure what steps I need to do in order to do so. I have the table written down, however, there's two central atoms so what would I do? There seems to be 4 electron domains on the carbon atom and no lone pairs so it would seem like this shape would be tetrahedral. Here's what I have now. Thanks!arrow_forwardWe discussed the solid phase resin using in peptide synthesis. Provide a mechanism, for its formation. DRAW THE MECHANISM.arrow_forwardPlease help. Every time I've asked an expert in the past, it's been wrong :(arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning