
Concept explainers
(a)
Interpretation:
The lipogenesis C4-ACP monoacid intermediate counterpart for the citric acid cycle C4-diacid intermediate succinate has to be determined.
Concept introduction:
Lipogenesis is the process employed for the synthesis of fatty acid. The starting precursor for the synthesis is acetyl CoA. The enzyme employed for the process is fatty acid synthase. It is a multienzyme complex that ties the reaction responsible for the synthesis of fatty acid. This process is the reverse of the degradation of fatty acid.
The Citric acid cycle is a series of biochemical reactions that use acetyl CoA (produced by oxidation of pyruvate) to produce carbon dioxide, NADH and FADH2 in a series of
(a)
Answer to Problem 25.106EP
The lipogenesis C4-ACP monoacid intermediate counterpart for the citric acid cycle C4-diacid intermediate succinate is butyrate.
Explanation of Solution
Succinic acid is a dicarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the citric acid cycle is a C4 derivative of dicarboxylic acid. The structure of succinic acid is,
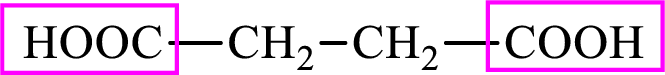
Intermediates involved in the lipogenesis are derivatives of C4 molecule butyric acid. Butyric acid is a monocarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the lipogenesis is a C4 derivative of monocarboxylic acid. The structure of butyric acid is,
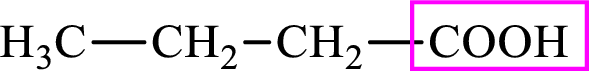
The structure of succinate is,
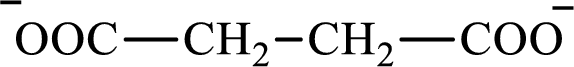
The structure of butyrate is,
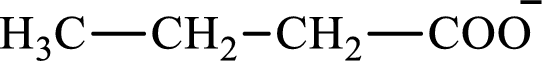
Butyrate and succinate are saturated acid with four carbon atoms in each molecule. butyrate is a monoacid formed as intermediate in lipogenesis while succinate is a diacid formed as intermediate in the citric acid cycle. Therefore, the lipogenesis C4-ACP monoacid intermediate counterpart for the citric acid cycle C4-diacid intermediate succinate is butyrate.
(b)
Interpretation:
The lipogenesis C4-ACP monoacid intermediate counterpart for the citric acid cycle C4-diacid intermediate malate has to be determined.
Concept introduction:
Lipogenesis is the process employed for the synthesis of fatty acid. The starting precursor for the synthesis is acetyl CoA. The enzyme employed for the process is fatty acid synthase. It is a multienzyme complex that ties the reaction responsible for the synthesis of fatty acid. This process is the reverse of the degradation of fatty acid.
The Citric acid cycle is a series of biochemical reactions that use acetyl CoA (produced by oxidation of pyruvate) to produce carbon dioxide, NADH and FADH2 in a series of redox reactions.
Intermediates involved in the lipogenesis are derivative of C4 molecule butyric acid. Butyric acid is a monocarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the lipogenesis is a C4 derivative of monocarboxylic acid. The structure of butyric acid is,
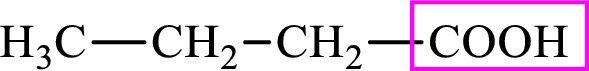
Succinic acid is a dicarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the citric acid cycle is a
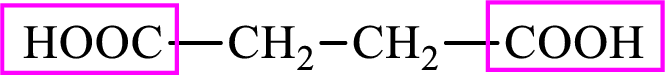
(b)
Answer to Problem 25.106EP
The lipogenesis C4-ACP monoacid intermediate counterpart for the citric acid cycle C4-diacid intermediate malate is β-hydroxybutyrate.
Explanation of Solution
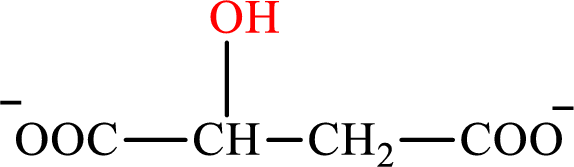
Malate is the intermediate in the citric acid cycle. The structure of malate is,
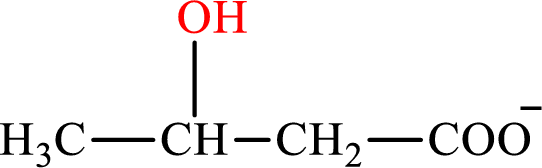
β-Hydroxybutyrate is the intermediate in the lipogenesis. The structure of β-hydroxybutyrate is,
Malate and β-hydroxybutyrate are the hydroxy derivatives of saturated acid with four carbon atoms in each molecule. Malate is a hydroxy derivative of dicarboxylic acid that is formed as the intermediate in the citric acid cycle whileβ-hydroxybutyrate is a hydroxy derivative of monocarboxylic acid that is formed as the intermediate in the lipogenesis. The lipogenesis C4-ACP monoacid intermediate counterpart for the citric acid cycle C4-diacid intermediate malate is β-hydroxybutyrate.
(c)
Interpretation:
The lipogenesis C4-ACP monoacid intermediate counterpart for the citric acid cycle C4-diacid intermediate oxaloacetate has to be determined.
Concept introduction:
Lipogenesis is the process employed for the synthesis of fatty acid. The starting precursor for the synthesis is acetyl CoA. The enzyme employed for the process is fatty acid synthase. It is a multienzyme complex that ties the reaction responsible for the synthesis of fatty acid. This process is the reverse of the degradation of fatty acid.
The Citric acid cycle is a series of biochemical reactions that use acetyl CoA (produced by oxidation of pyruvate) to produce carbon dioxide, NADH and FADH2 in a series of redox reactions.
Intermediates involved in the lipogenesis are derivatives of C4 molecule butyric acid. Butyric acid is a monocarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the lipogenesis is a C4 derivative of monocarboxylic acid. The structure of butyric acid is,
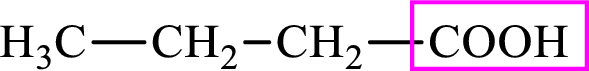
Succinic acid is a dicarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the citric acid cycle is a C4 derivative of dicarboxylic acid. The structure of succinic acid is,
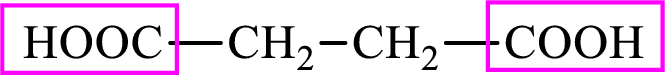
(c)
Answer to Problem 25.106EP
The lipogenesis C4-ACP monoacid intermediate counterpart for the citric acid cycle C4-diacid intermediate oxaloacetate is acetoacetate.
Explanation of Solution
Oxaloacetate is the intermediate in the citric acid cycle. The structure of oxaloacetate is,
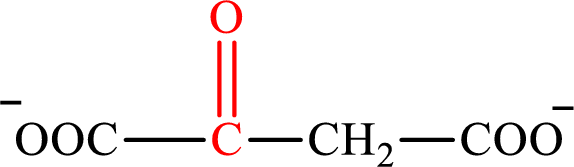
Acetoacetate is the intermediate in the lipogenesis. The structure of acetoacetate is,
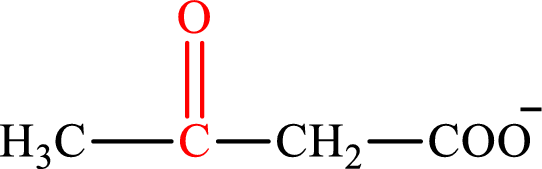
Oxaloacetate and acetoacetate are the keto derivatives of saturated acid with four carbon atoms in each molecule. Oxaloacetate is a keto derivative of dicarboxylic acid that is formed as the intermediate in the citric acid cycle while acetoacetate is a keto derivative of monocarboxylic acid that is formed as the intermediate in the lipogenesis. Therefore, the lipogenesis C4-ACP monoacid intermediate counterpart for the citric acid cycle C4-diacid intermediate oxaloacetate is acetoacetate.
(d)
Interpretation:
The lipogenesis C4-ACP monoacid intermediate counterpart for the citric acid cycle C4-diacid intermediate fumarate has to be determined.
Concept introduction:
Lipogenesis is the process employed for the synthesis of fatty acid. The starting precursor for the synthesis is acetyl CoA. The enzyme employed for the process is fatty acid synthase. It is a multienzyme complex that ties the reaction responsible for the synthesis of fatty acid. This process is the reverse of the degradation of fatty acid.
The Citric acid cycle is a series of biochemical reactions that use acetyl CoA (produced by oxidation of pyruvate) to produce carbon dioxide, NADH and FADH2 in a series of redox reactions.
Intermediates involved in the lipogenesis are derivatives of C4 molecule butyric acid. Butyric acid is a monocarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the lipogenesis is a C4 derivative of monocarboxylic acid. The structure of butyric acid is,
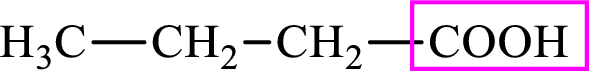
Succinic acid is a dicarboxylic acid and has 4 carbon atoms. Thus, each intermediate of the citric acid cycle is a C4 derivative of dicarboxylic acid. The structure of succinic acid is,
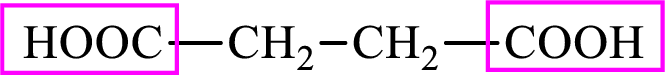
(d)
Answer to Problem 25.106EP
The lipogenesis C4-ACP monoacid intermediate counterpart for the citric acid cycle C4-diacid intermediate fumarate is crotonate.
Explanation of Solution
Fumarate is the intermediate in the citric acid cycle. The structure of fumarate is,
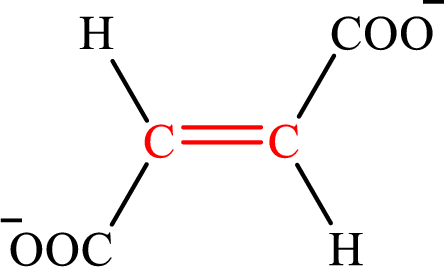
Crotonate is the intermediate in the lipogenesis. The structure of crotonate is,
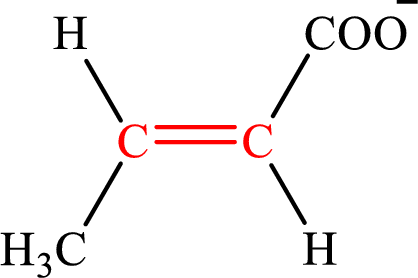
Fumarate and crotonate are the unsaturated derivatives of the
Want to see more full solutions like this?
Chapter 25 Solutions
General, Organic, And Biological Chemistry, Hybrid (with Owlv2 Quick Prep For General Chemistry Printed Access Card)
- in the scope of the SCH4U course! please show all steps as im still learning how to format my answers in the format given, thank you!arrow_forwardhelp me solve this HWarrow_forwardMolecules of the form AH2 can exist in two potential geometries: linear or bent. Construct molecular orbital diagrams for linear and bent CH2. Identify the relevant point group, include all of the appropriate symmetry labels and pictures, and fill in the electrons. Which geometry would you predict to be more stable, and why? (Please draw out the diagram and explain)arrow_forward
- Indicate the variation in conductivity with concentration in solutions of strong electrolytes and weak electrolytes.arrow_forwardThe molar conductivity of a very dilute solution of NaCl has been determined. If it is diluted to one-fourth of the initial concentration, qualitatively explain how the molar conductivity of the new solution will compare with the first.arrow_forwardWhat does the phrase mean, if instead of 1 Faraday of electricity, Q coulombs (Q/F Faradays) pass through?arrow_forward
- What characteristics should an interface that forms an electrode have?arrow_forwardFor a weak acid AcH, calculate the dissociated fraction (alpha), if its concentration is 1.540 mol L-1 and the concentration [H+] is 5.01x10-4 mol L-1.arrow_forwardIf the molar conductivity at infinite dilution of HAC is A0 = 390.5 S cm² mol¹. Calculate the Arrhenius conductivity of a 9.3% by weight solution of HAc with a pH of 3.3. Data: molecular weight of HAC is 60.05 g/mol and the density of the solution is 1 g/cm³.arrow_forward
- If the molar conductivity at infinite dilution of HAC is A0 = 390.5 S cm² mol¹. Calculate the Arrhenius conductivity of a 9.3% by weight solution of HAc with a pH of 3.3. Data: molecular weight of HAC is 60.05 g/mol and the density of the solution is 1 g/cm³.arrow_forwardIf the molar conductivity at infinite dilution of HAC is A0 = 390.5 S cm² mol¹. Calculate the Arrhenius conductivity of a 9.3% by weight solution of HAc with a pH of 3.3. Data: molecular weight of HAC is 60.05 g/mol and the density of the solution is 1 g/cm³.arrow_forwardDetermine the distance between the metal and the OHP layer using the Helm- holtz model when the electrode's differential capacitance is 145 μF cm². DATA: dielectric constant of the medium for the interfacial zone &r= lectric constant of the vacuum &0 = 8.85-10-12 F m-1 = 50, die-arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning




