
CHEMISTRY+CHEM...HYBRID ED.(LL)>CUSTOM<
9th Edition
ISBN: 9781305020788
Author: John C.Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: CENGAGE C
expand_more
expand_more
format_list_bulleted
Question
Chapter 24.3, Problem 3Q
Interpretation Introduction
Interpretation:
The molecular geometry around the phosphorous atom in a phosphorothioate group has to be identified.
Concept introduction: In phosphorothioate group phosphrous atom bonded with surrounding atoms oxygen and sulphur as shown below.
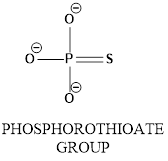
In the diagram shows
Tetrahedral geometry: A tetrahedral has a central atom which is surrounded by four other atoms. In tetrahedral geometry the bond angle is
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
<
Done
19:22
www-awu.aleks.com
Chapter 12 HW
Question 4 of 39 (2 points) | Question Attempt: 5 of Unlimited
:
.. LTE
סוי
1
✓ 2
✓ 3
= 4
✓ 5
✓ 6
✓ 7
✓ 8
✓ 9
= 10
11
✓ 12
Consider the molecule (CH3)2CHCH2CHCн for the following questions.
Part 1 of 2
Which of the following molecules is/are constitutional isomer(s) to
(CH3)2CHCH2CH2CH3? Check all that apply.
Part 2 of 2
(CH3),C(CH2)2CH3
CH3
H,C-CH-CH-CH,
CH 3
None of the above.
☑
Which of the following molecules is/are identical molecules to
(CH3)2CHCH2CH2CH₁₂? Check all that apply.
CH3
H,C-CH-CH₂-CH2-CH,
CH3(CH2)2CH(CH3)2
CH2-CH2-CH3
HỌC-CH=CH,
乂
☑
а
None of the above
Check
Save For Later
Submit Assignment
© 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center Accessibility
18:11
LTE ا...
US$50 off hotels is waiting for you
Book now, hotels in Nashville are going fast
QUTSLIVII 25 61 69 points) | QuestIVIT ALLēm...
now
Give the IUPAC name for each compound.
Part 1 of 3
Part 2 of 3
X
☑
Х
Check
Save For Later
Submit
© 2025 McGraw Hill LLC. All Rights Reserved.
TOMS CT US
...vacy Center | Accessibility
Done
19:17
www-awu.aleks.com
Chapter 12 HW
Question 29 of 39 (6 points) | Question Attempt: 1 of Unlimited
.III LTE
סוי
27
28
= 29
30
31
32
= 33
34
35
Consider this structure.
CH3CH2CH2
Part 1 of 3
3
CH2 CH2CH3
-
C-CH2CH
3
H
CH₂
Give the IUPAC name of this structure.
3-ethyl-3,4-dimethylheptane
Part: 1/3
Part 2 of 3
Draw the skeletal structure.
Skip Part
<
Check
Click and drag to start drawing a
structure.
Save For Later
Submit
© 2025 McGraw Hill LLC. All Rights Reserved.
Terms of Use | Privacy Center | Accessibility
Х
Chapter 24 Solutions
CHEMISTRY+CHEM...HYBRID ED.(LL)>CUSTOM<
Ch. 24.1 - Draw the Lewis structure for the tripeptide...Ch. 24.1 - Prob. 1RCCh. 24.1 - Prob. 2RCCh. 24.1 - Prob. 3RCCh. 24.2 - Prob. 1RCCh. 24.2 - Prob. 2RCCh. 24.2 - Prob. 3RCCh. 24.3 - What is the sequence of the strand of DNA...Ch. 24.3 - Prob. 2CYUCh. 24.3 - 1. Which breaks down more quickly in an aqueous...
Ch. 24.3 - Prob. 2RCCh. 24.3 - 3. Which amino acid is selected by the mRNA codon...Ch. 24.3 - Kynamro has the hydrogen bonding sequence:...Ch. 24.3 - The formula of Kynamro is...Ch. 24.3 - Prob. 3QCh. 24.4 - 1. Which of the following is not an example of a...Ch. 24.4 - Prob. 2RCCh. 24.5 - Prob. 1RCCh. 24.5 - Prob. 2RCCh. 24.5 - Prob. 1QCh. 24.5 - Prob. 2QCh. 24.5 - Prob. 3QCh. 24 - (a) Draw the Lewis structure for the amino acid...Ch. 24 - (a) Draw the Lewis structure for the amino acid...Ch. 24 - Prob. 3PSCh. 24 - Prob. 4PSCh. 24 - Draw Lewis structures for the two dipeptides that...Ch. 24 - Do the amino acid sequences: valine-asparagine and...Ch. 24 - Draw the Lewis structure for the tripeptide...Ch. 24 - Prob. 8PSCh. 24 - Prob. 9PSCh. 24 - Prob. 10PSCh. 24 - Prob. 11PSCh. 24 - Prob. 12PSCh. 24 - (a) Draw the structural formula for the sugar...Ch. 24 - (a) Draw the structural formula for the sugar -D-2...Ch. 24 - Prob. 15PSCh. 24 - Prob. 16PSCh. 24 - Given the following nucleotide sequence in DNA:...Ch. 24 - Given the following nucleotide sequence in DNA: 5'...Ch. 24 - Prob. 19PSCh. 24 - If a drop of oleic acid is added to a dish of...Ch. 24 - What structure do all steroids have in common?Ch. 24 - Prob. 22PSCh. 24 - Prob. 23PSCh. 24 - The chemical equation for the fermentation of...Ch. 24 - Prob. 25PSCh. 24 - Prob. 26PSCh. 24 - Prob. 27GQCh. 24 - Prob. 28GQCh. 24 - Prob. 29GQCh. 24 - Prob. 30GQCh. 24 - Prob. 31GQCh. 24 - There are 41 = 4 mononucleotides of DNA, there are...Ch. 24 - Prob. 33GQCh. 24 - The first step of the metabolic process known as...Ch. 24 - Prob. 35ILCh. 24 - Insulin is a protein important in the metabolism...Ch. 24 - Prob. 37SCQCh. 24 - Prob. 38SCQCh. 24 - Do the DNA sequences ATGC and CGTA represent the...Ch. 24 - Prob. 41SCQCh. 24 - Which of the following statements is/are true? (a)...
Knowledge Booster
Similar questions
- 18:57 .III LTE www-awu.aleks.com Chapter 12 HW Question 31 of 39 (8 points) | Question Attem... Give the IUPAC name of each compound. Part 1 of 4 Part 2 of 4 Х Х Check Save For Later Submit © 2025 McGraw Hill LLC. All Rights Reserved. TOMS OF US vacy Center | Accessibilityarrow_forwardWhat is the missing reactant in this organic reaction? CH3-C-CH2-NH2 + R - CH3 O: 0 CH3-N-CH2-C-NH-CH2-C-CH3 + H2O Specifically, in the drawing area below draw the condensed structure of R. If there is more than one reasonable answer, you can draw any one of them. If there is no reasonable answer, check the No answer box under the drawing area. Note for advanced students: you may assume no products other than those shown above are formed. Explanation Check Click anywhere to draw the first atom of your structure. C © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center Accesarrow_forwardDone 18:17 • www-awu.aleks.com Chapter 12 HW Question 24 of 39 (4 points) | Question Attempt: 1 of Unlimited ▼ 20 ✓ 21 × 22 23 24 25 26 raw the structure corresponding to each IUPAC name. Part 1 of 2 .III LTE 22 27 28 סוי 29 29 3 A skeletal structure corresponding to the IUPAC name 3-ethyl-4-methylhexane. Part 2 of 2 Click and drag to start drawing a structure. A condensed structure corresponding to the IUPAC name 2,2,4- trimethylpentane. Click anywhere to draw the first atom of your structure. Check Save For Later Submit < Х ப: G © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility : Garrow_forward
- Done 18:25 www-awu.aleks.com .III LTE Chapter 12 HW Question 29 of 39 (6 points) | Question Attempt: 1 of Unlimi... Oli 23 24 25 26 27 28 29 30 Consider this structure. CH2 CH2CH2 CH2CH2CH₂ C -C. -CH2CH3 H CH Part: 0 / 3 Part 1 of 3 Give the IUPAC name of this structure. Skip Part < Check ☑ Save For Later © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility ....................arrow_forwardCalculate Ecell at 25.0 oC using the following line notation. Zn(s)|Zn+2(aq, 0.900 M)||Cu+2(aq, 0.000200 M)|Cu(s)arrow_forwardPredict the product of this organic reaction: O OH + H + OH A P + H2O Specifically, in the drawing area below draw the skeletal ("line") structure of P. If there isn't any P because this reaction won't happen, check the No reaction box under the drawing area. Explanation Check Click and drag to start drawing a structure. X G ☐ :arrow_forward
- 0.0994 g of oxalic acid dihydrate is titrated with 10.2 mL of potassium permanganate. Calculate the potassium permanganate concentration. Group of answer choices 0.0433 M 0.135 M 0.0309 M 0.193 Marrow_forwardExperts...can any one help me solve these problems?arrow_forwardAccording to standard reduction potential data in Lecture 4-1, which of the following species is the most difficult to reduce? Group of answer choices Zn2+ AgCl(s) Al3+ Ce4+arrow_forward
- Which Group 1 metal reacts with O2(g) to form a metal peroxide (M2O2)? Group of answer choices Li K Rb Naarrow_forwardWhich of the following statements is true regarding the reaction between Group 1 metals and water? Group of answer choices These reactions result in a basic solution. The metals do not actually react easily with water due to the metals' lack of conductivity. These reaction result in an acidic solution. The metals need their outer coatings of metal oxides to react.arrow_forwardWhich element cannot interact with hydrogen through hydrogen bonds? Group of answer choices O S Br Narrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning